DNA fingerprinting and molecular diversity analysis for the improvement of brinjal (Solanum melongena L.) cultivars
Abstract
An investigation was conducted to assess genetic diversity of Bangladesh Agricultural Research Institute (BARI) released 11 varieties of brinjal (BARI begun 1, BARI begun 4-10, BARI hybrid begun 1, 3 and 4) using PCR-based randomly amplified polymorphic DNA (RAPD) markers. A total of 44 distinct DNA amplified bands were observed for five primers (OPB-04, OPB-08, OPD-02, OPP-13 and OPW-08) with an average of 5.6 polymorphic bands per primer. The overall gene diversity was detected 0.216 and level of polymorphism was found 63.64%. The pair wise inter-variety similarity indices ranged from 67.03% to 97.61%. The UPGMA dendrogram segregated 11 genotypes of brinjal into two main clusters. The first major cluster had only one genotype (BARI begun 6) and the second major cluster had rest of ten genotypes. BARI begun 6 vs BARI begun 1 showed the highest Nei’s genetic distance (0.5261) as they are released from different parental origin. On the other hand, BARI begun 9 vs BARI begun 7 varietal pair showed lowest genetic distance (0.0465) as they are released from same parental origin. The experiment reveals genetic diversity and relatedness among 11 brinjal genotypes which may be informative for the future varietal identification and genetic improvement of this vegetable crop.
INTRODUCTION
Brinjal (Solanum melongena L.) is an economically important Solanaceous vegetable, widely consumed in Asia, Europe, Africa and America [1; 2; 3]. It has the early European name ‘eggplant” locally known as “Begun” in Bangladesh. It is a self-pollinated, diploid (2n=24) annual crop belongs to the family Solanaceae. Brinjal is largely cultivated in almost all districts of Bangladesh. The total area of eggplant cultivation is 76370 acres with total annual production of 310354 metric tons with an average yield of 4064 kg per acre in 2014-2015 [4]. Globally, the eggplant is ranked as third most important crop from Solanaceae family after potato and tomato with an annual production of more than 41×106 tons [5].
Brinjal is nutritious vegetable and has got multifarious use as a dish item. It is a valuable source of minerals (particularly iron) and vitamins. The fruit and other parts of the plant are used in traditional medicine [6]. Eggplant is ranked as one of the top ten vegetables due to its high oxygen radical scavenging capacity [7]. Research studies have shown that phenolic compounds of eggplant have antioxidant properties preventing oxidation and diabetes complications [8; 9]. Additionally, the eggplant peel is rich in anthocyanins having therapeutic potential against hyperlipidemia and cardiovascular diseases by inhibiting lipid peroxidation [9; 10].
Genetic resources management of brinjal cultivars is co-related with the sustainable agricultural and economic development of the country. The yield potential of this eggplant in Bangladesh is very low compared to other countries due to the incidence of insect pests and diseases [11; 12]. Molecular characterization of this eggplant may provide an avenue to screen the germplasm resources for any selective breeding and genetic improvement. Molecular markers are reliable tools to characterize the DNA profile of plant genotypes to study the genetic diversity. In Bangladesh, genetic data on brinjal is not rich enough. So genetic status of this important crop is needed to be established and documented by using DNA markers which may provide valuable information for further breeding programme. Randomly amplified polymorphic DNA (RAPD) markers are effective for the evaluation of genetic diversity due to easy application and relatively low expensive compared to other molecular markers [13; 14]. Molecular characterization by RAPD markers is easy and rapid. RAPD markers are being used for the identification of genetic relationship among cultivars [15; 16].
The present investigation was formulated with the objective of studying genetic variation among the different released brinjal cultivars through RAPD markers, molecular characterization by DNA profiling and to reveal phylogenetic relatedness and genetic distance among the brinjal genotypes.
MATERIALS AND METHODS
Experimental plant materials and their sources
The plant materials for this study consisted of 11 brinjal varieties namely BARI begun 1, BARI begun 4-10, F1 hybrids (BARI hybrid begun 1, 3 and 4). All the varieties were collected from Horticulture Research Centre (HRC) of Bangladesh Agricultural Research Institute (BARI), Joydebpur, Gazipur. Seedlings were grown at the Laboratory of Department of Biotechnology at Sher-e-Bangla Agricultural University, Dhaka in earthen tubs containing soils collected from nursery.
Genomic DNA isolation
Genomic DNA of the brinjal seedlings was isolated following protocol described by Saghai-Maroof and co-workers [17] as well as Islam and co-workers [18; 19] with some modifications. Approximately 200 mg of young leaves of 30 days old plants were cut into minuscule pieces, homogenized and digested with extraction buffer (pH= 8.0): 50 mM Tris-HCl, 25 mM EDTA (Ethylenediaminetetraacetic acid), 300 mM NaCl and 1% SDS (Sodium Dodecyl Sulfate). Following incubation for 20 minutes at 65°C with intermittent shaking, the mixture was vortexed with an equal volume of phenol: chloroform: isoamyl alcohol (25:24:1, v/v/v) for 20 seconds. The emulsified mixture was centrifuged at 13000 rpm for 10 minutes. The supernatant was used for precipitation with 2/3 volume of 100% alcohol in presence of 0.3 M sodium acetate and DNA was pelleted by centrifugation. The DNA pellets were then washed with 70% ethanol. The air dried DNA pellets were resuspended in an appropriate volume of TE buffer (10 mM Tris-HCl, 1 mM EDTA, pH=8.0) and finally treated with RNAse and stored at -20°C. The quality and quantity of DNA were checked by electrophoresis and spectrophotometer, respectively.
Screening and selection of RAPD primers
Initially, nine decamer primers namely OPA-18, OPB-04, OPB-06, OPB-08, OPD-02, OPF-08, OPG-19, OPP-13 and OPW-08 (Operon Technologies, Inc. Alameda, California, USA) were selected and screened by polymerase chain reaction (PCR) for their ability to produce polymorphic patterns in brinjal varieties and five primers (OPB-04, OPB-08, OPD-02, OPP-13 and OPW-08) which gave reproducible and distinct polymorphic amplified products were selected finally for RAPD analysis.
Polymerase chain reaction
PCR reactions were performed for each DNA sample in a 25 µL reaction mix containing 12.5 µL 2X Taq Master mix (GeneON, Germany), 2.5 µL of RAPD primer (10 pM), 7.5 µL of sterile de-ionized water and 2.5 µL of genomic DNA (25ng/ µL). DNA amplification was performed in an oil-free thermal cycler by setting a thermal profile as pre-heating at 95o C for 5 min followed by 33 cycles of 45 s denaturation at 95o C, 30 s annealing at 30o C and extension at 72o C for 1 min. After the last cycle, a final step of 5 min at 72o C was added to allow complete extension of all amplified fragments.
Visualization of PCR products
PCR products were confirmed by electrophoresis on 1.5 % agarose gel containing 1 µL ethidium bromide (10 mg/L) in 1X TBE buffer at 85 V for 50 min. After electrophoresis, the gel was taken out carefully from the gel chamber and was placed on high-performance ultraviolet light box (UV trans-illuminator) of gel documentation for checking the RAPD bands and photographed by a Gel Cam Polaroid camera.
RAPD data analysis
Following electrophoresis, the sizes of the PCR products were estimated by comparisons of distance travelled by each fragment with distance travelled by known size fragments of the DNA molecular weight markers (100 bp DNA ladder, Genei, India or 1 Kb DNA ladder, BRIC, Korea). All distinct bands or fragments (RAPD markers) were thereby given identification numbers according to size and scored visually on the basis of their presence (1) or absence (0), separately for each variety for each primer. The scores recorded for all RAPD primers were then pooled to create a single data matrix. This was used to compare the frequencies of all polymorphic RAPD markers among populations with 1000 simulated samples using POPGENE (version 1.31) [20] computer program. The size of the RAPD markers were estimated by using the software DNAfrag, version 3.03 [21]. Nei’s [22] genetic distance values were calculated based on the frequencies of polymorphic markers using the POPGENE (Version 1.31) computer package [20]. For each population group, Nei’s gene diversity (h) was calculated for each locus and then averaged over all loci.
These parameters were performed using POPGENE software [20] in the formula: h=1-∑pi2, I= ∑pi log pi, where pi = the frequency of the allele i in the population. A value indicates small genetic distances, 0.10-0.15 indicates moderate genetic distances, 0.15-0.2 indicates high genetic distances and >0.2 indicates very large genetic distances [23]. The dendrogram was constructed using POPGENE (Version 1.31) [20]. Genetic similarity values were calculated manually based on the formula, Similarity Index (SI) = 2NXY/(NX + NY); where, NXY is the number of RAPD bands shared by individuals X and Y, respectively, and NX and NY are the number of bands in individuals X and Y, respectively [24; 25].
RESULTS
Profiling of RAPD bands and their polymorphisms
A total of 44 distinct and different PCR based RAPD bands were produced with 8.8 per primer. The size of the bands ranges from 198 to 2898 bp (Table 1). The highest number of bands (12) was generated by primer OPD-02. Besides, the primer OPB-04, OPB-08, OPP-13 and OPW-08 generated 8 scorable bands each. The RAPD banding profile is shown in Figure 1. The highest proportion of polymorphic bands (87.5%) was detected by primer OPW-08 while primer OPB-04 detected the least proportion (37.5%) and overall level of polymorphism was 63.64% (Table 1). The average values of Nei’s gene diversity for the RAPD primers ranged from 0.083 to 0.388 with an overall value of 0.216 (Table 1).
Table 1. RAPD primers with corresponding number and size range of bands together with a percentage of polymorphic loci and Nei‟s gene diversity values observed in 11 brinjal varieties.
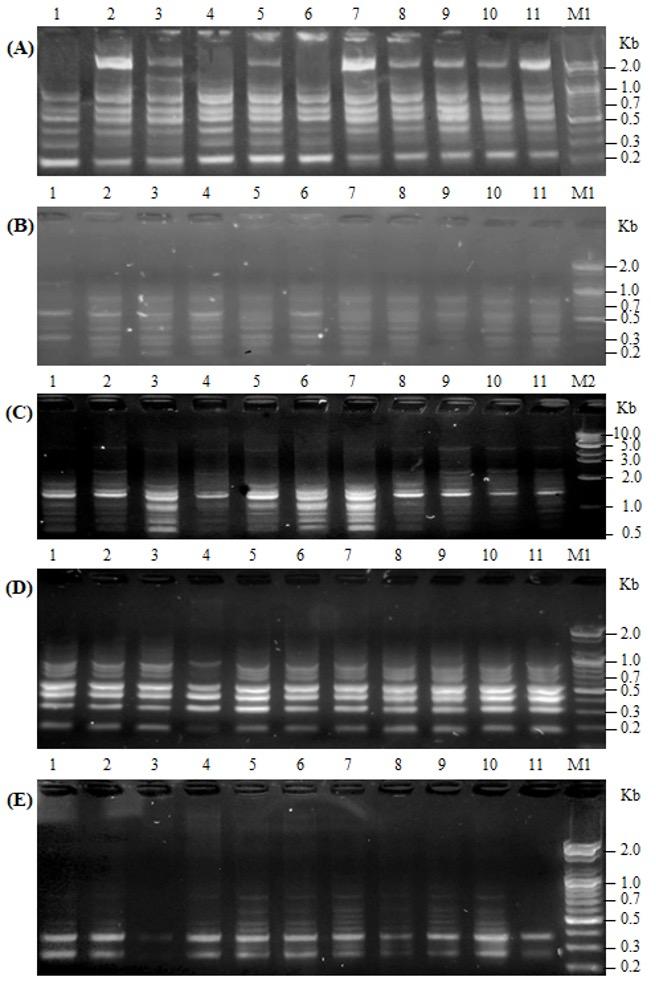
Inter-variety similarity indices
Inter-varietal similarity indices (Sij) ranged from 67.03% to 97.61% (Data are not shown due to large sized table). The highest similarity indices (97.61%) were found between BARI begun 7 and BARI begun 9. BARI begun 6 and BARI begun 10 showed least inter-variety similarity indices 67.03%. All the 55 varietal pairs were not homogenous at different number of loci.
Genetic identity and genetic distance
The highest Nei’s genetic identity (0.9545) was observed in BARI begun 9 vs BARI begun 7 varietal pairs whereas the lowest genetic identity (0.6136) was estimated in BARI begun 6 vs BARI begun 5 varietal pair (Table 2). The result indicates that the low or high level genetic distance exists between varieties with their same or different origins. BARI began 6 vs BARI begun 1 showed highest Nei’s genetic distance (0.5261) as they are released from different parental origin. On the other hand BARI began 9 vs BARI begun 8 varietal pair showed lowest genetic distance (0.0706) as they are released from same parental origin. This variation might be created due to geographical origin.
Table 2. Neil's genetic identity.
Phylogenetic relatedness and construction of dendrogram
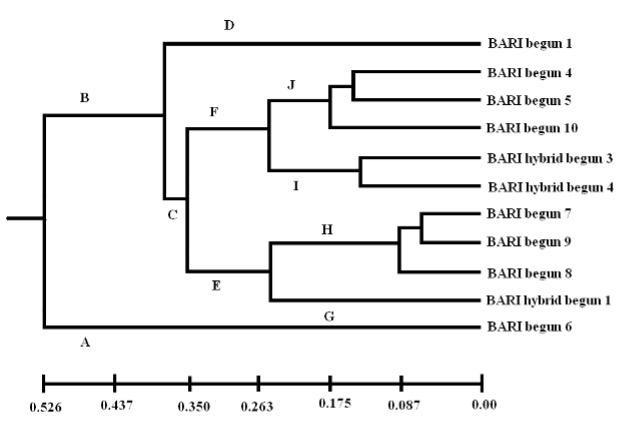
Phylogenetic dendrogram based on Nei’s (1972) genetic distance using Unweighted Pair Group Method of Arithmetic Means (UPGMA) indicated the segregation of 11 genotypes of brinjal into two main clusters, ‘A’ and ‘B’ (Figure 2). The first major cluster ‘A’ had only one genotype (BARI begun 6) and the second major cluster ‘B’ had rest of ten genotypes. The second major cluster subdivided into two minor clusters (C & D) in which one cluster ‘D’ had only one genotype (BARI begun 1) and the other cluster ‘C’ had rest of the nine varieties. This minor cluster further subdivided into two clusters (E & F). In which cluster E divided into two sub clusters (G and H). BARI hybrid begun 1 formed cluster ‘G’ and BARI begun 7, BARI begun 8 and BARI begun 9 grouped in cluster ‘H’. Cluster ‘I’ and ‘J’ were the subdivision of cluster ‘F’. BARI hybrid begun 3 and BARI hybrid begun 4 formed cluster ‘I’ and BARI begun 4, BARI begun 5 and BARI begun 10 were grouped in cluster ‘J’.
DISCUSSION
With advancement of DNA marker technology, molecular characterization plays a vital role in managing and utilizing plant genetic resources [26]. It is immensely helpful in selective breeding from diverse parents to widen the breeding gene pool [27]. Thereby, DNA markers are increasingly adopted as an effective and appropriate tool for basic and applied studies addressing biological components in agricultural production systems [28].
In the present study, the overall level of polymorphism (63.64%) indicated the effectiveness of RAPD technique to investigate genetic diversity among the different genotypes of brinjal. Number of polymorphic bands ranged from 3-9 with an average of 5.6. Verma and co-workers [29] reported 5.58 bands per primer and Islam and co-workers [30] found 5.67 bands per primer in brinjal varieties which are mostly consistent with the present study. Almost similar level of polymorphisms was reported in different experiment with brinjal and other eggplants, for instance 57.89% [31], 57.58% [32]. In contrast, higher level of polymorphism in brinjal germplasm was obtained by Aguoru and co-workers [33] (83.33%) as well as by Islam and coworkers [30] (70.59%). The value of overall gene diversity was 0.216 in this study which is very close to the findings reported by Biswas and co-workers [31], where they observed overall gene diversity value of 0.23 among 10 promising brinjal varieties with RAPD markers.
Karihaloo and co-workers [34] reported the highest similarity (0.947) between S. insanum and S. melongena while analyzing genetic similarity of 52 accessions of Solanum. The present study indicates a high level of genetic variation within some brinjal varieties which were perceived from the data of inter-variety similarity indices and genetic distance value. Phylogenetic clustering patterns based on genetic distance values also indicated diverse relationship that occurred might be due to geographical origin of the parental varieties.
Finally it can be concluded that high degree of diversity of the studied brinjal cultivars may be attributable to genetic improvement programme based on the clustering patterns. It will also provide support for selection of crossing combinations from parental genotypes and for broadening the genetic basis of breeding programs. Due to having some medicinal values particularly against cancer, diabetes and cardiovascular disease, marker assisted breeding and production of this important eggplant may contribute to enrich diets and bring health benefits.
ACKNOWLEDGEMENT
The authors express thanks to the Ministry of Science and Technology, Government of the Peoples’ Republic of Bangladesh for providing NST (National Science and Technology) fellowship with financial support to conduct this fundamental research.
AUTHOR CONTRIBUTIONS
SS performed the experiment; MNI and MEH conceived the study; SS and MNI analyzed the data; SS and MNI wrote the paper.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
References
- [1]Harish DK, Agasimani AK, Imamsaheb SJ, Patil SS. Growth and yield parameters in brinjal as influenced by organic nutrient management and plant protection conditions. Res J Agril Sci 2011; 2(2): 221-25.
- [2]Ali Z, Xu ZL, Zhang DY, He XL, Bahadur S, Yi JX. Molecular diversity analysis of eggplant (Solanum melongena) genetic resources. Genet Mol Res 2011; 10(2): 1141-55.
- [3]Kumar G, Meena BL, Kar R, Tiwari SK. Morphological diversity in brinjal (Solanum melongena L.) germplasm accessions. Plant Genet Resour 2008; 6(3): 232-36.
- [4]BBS. Yearbook of Agricultural Statistics-2015 (27th Series), Bangladesh Bureau of Statistics, Ministry of Planning, Government of the People’s Republic of Bangladesh, Dhaka, Bangladesh, 2016.
- [5]Food and Agriculture Organization (FAO) of the United Nations, Agriculture database, 2010.
- [6]Kashyap V, Kumar S, Collonier C, Fusari F, Haicour R, Rotino GL, Sihachak RD, Rajam MV. Biotechnology of eggplant. Sci Hort 2003; 97(1): 1-25.
- [7]Jung EJ, Bae MS, Jo EK, Jo YH, Lee SC. Antioxidant activity of different parts of eggplant. J Med Plants Res 2011; 5(18): 4610-15.
- [8]Kwon YI, Apostolidis E, Shetty K. In vitro studies of eggplant (Solanum melongena) phenolics as inhibitors of key enzymes relevant for type 2 diabetes and hypertension. Bioresour Technol 2008; 99(8): 2981-88.
- [9]Scorsatto M, Pimentel AC, Silva AJR, Sabally K, Rosa G, Oliveira GMM. Assessment of bioactive compounds, physicochemical composition, and in vitro antioxidant activity of eggplant flour. Int J Cardiovasc Sci 2017; 30(3): 235-42.
- [10]Basuny AM, Arafat SM, El-Marzooq MA. Antioxidant and antihyperlipidemic activities of anthocyanins from eggplant peels. J Pharma Res Rev 2012; 2(3): 50-57.
- [11]Das AN, Singh BR. Field reaction of brinjal varieties against shoot and fruit borer, Leucinodes orbonalis. Environ Eco 2000; 8(2): 761-62.
- [12]Rashid MM. A Guidebook of Plant Pathology. Department of Plant Pathology, Hajee Mohammad Danesh Science and Technology University, Bangladesh, 2000, p58.
- [13]Williams JGK, Hanafey MK, Rafalski JA, Tingey SV. Genetic analysis using random amplified polymorphic DNA markers. Methods Enzymol 1990; 1218: 704-40.
- [14]Rafalski JA, Tingey SV. Genetic diagnostics in plant breeding: RAPDs, microsatellites and machines. Trends Genet 1999; 9: 275-79.
- [15]Afzal MA, Haque MM, Shanmugasundaram S. Random Amplified Polymorphic DNA (RAPD) analysis of selected mungbean (Vigna radiata L. Wilczek) cultivars. Asian J Plant Sci 2004; 3(1): 20-24.
- [16]Tosti N, Negri V. Effiency of three PCR-based markers in assessing genetic variation among cowpea (vigna ungiculata spp unguiculata) landraces. Genome 2002; 45: 268-75.
- [17]Saghai-Maroof MA, Soliman KM, Jonsensan RA, Allard RW. Ribosomal spacer length polymorphism in barley: Mendelian inheritance, chromosomal location and population dynamics. Proc Natl Acad Sci USA, 1984; 81: 8014-19.
- [18]Islam MN, Molla MR and Rahman L. Genetic characterization of sesame varieties through RAPD fingerprinting technique in Bangladesh. J Sher-e-Bangla Agric Univ 2013; 7(1): 1-5.
- [19]Islam MN, Molla MR, Rohman MM, Hasanuzzaman M, Islam SMN and Rahman L. DNA fingerprinting and genotyping of cotton varieties using SSR Markers. Not. Bot. Horti Agrobot. Cluj-Napoca 2012; 40: 261-65.
- [20]Yeh FC, Yang RC, Boyle T. POPGENE VERSION 1.31: Microsoft Window-based free software for population genetic analysis. ftp://ftp.microsoft.com/Softlib/ HPGL.EXE, 1999.
- [21]Nash JHE. DNAfrag, Version 3.03. Institute for Biological Sciences, National Research Council of Canada, Ottawa, Ontario, Canada, 1991.
- [22]Nei M. Genetic distance between populations. Am Nat 1972; 106: 283-92.
- [23]Nei M. Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 1978; 89: 583-90.
- [24]Chapco W, Ashton NW, Martel RKB, Antonishyn N. A feasibility study of the use of random amplified polymorphic DNA in the population genetics and systematics of grasshoppers. Genome 1992; 35: 569-74.
- [25]Lynch M. The similarity index and DNA fingerprinting. Mol Biol Evol 1990; 7:478-84.
- [26]Karp A. The new genetic era: will it help us in managing genetic diversity? 43-56 p. In: Engels JMM, Rao VR, Brown AHD, Jackson MT (Eds.). Managing Plant Genetic diversity. International Plant Genetic Resources Institute, Rome, Italy, 2002.
- [27]Fu YB. Genetics redundancy and distinctness of flax germplasm as revealed by RAPD dissimilarity. Plant Genet Resour 2006; 4:177-84.
- [28]Jones N, Ougham H, Thomas H. Genome mapping, molecular markers and marker–assisted selection in crop plants. New phytol 1997; 137:165-77.
- [29]Verma M, Rathi S, Munshi AD, Kumar A, Arya L, Bhat KV, Kumar R. Genetic diversity of Indian brinjal revealed by RAPD and SSR markers. Indian J Horticult 2012; 69 (4): 517-22.
- [30]Islam Z, Siddiqua MK, Hasan MM, Nahar MA, Islam MA, Shamsuzzaman SM, Mondol MMA, Puteh AB. Assessment of genetic diversity of brinjal (Solanummelongena L.) germplasm by RAPD markers. Res Crops 2014; 15(2): 416-22.
- [31]Biswas MS, AkhondMA,Yousuf M, Al‐Amin, Mahmuda K, Muhammed RK. Genetic relationship among ten promising eggplant varieties using RAPD markers. Plant Tissue Cult Biotech 2009; 19(2): 119-26.
- [32]Sharmin D, Khalil MI, Begum SN, Meah MB. Molecular characterization of eggplant crosses by using RAPD analysis. Int J Sustainability Crop Prod 2011; 6: 22- 28.
- [33]Aguoru CU, Omoigui LO, Olasan JO. Population genetic study of eggplants (Solanum) species in Nigeria, Tropical West Africa, using molecular markers.Int J Plant Res 2015; 5: 7-12.
- [34]Karihaloo JL, Brauner S, Gottlieb LD. Random amplified polymorphic DNA variation in the eggplant, Solanummelongena L. (Solanaceae). Theor Applied Genet. 1995; 90(6): 767-70.