Morphometry and expression of immunoglobulins-containing plasma cells in the Harderian gland of Birds
Abstract
Johann Jacob Harder first described the Harderian gland in 1694 in deer. It is found in most terrestrial animals and is located within the variable aspects of the orbit. It is believed that this gland is involved in diverse functions. Among these, it has been held to be a site of immune response, a source of thermoregulatory lipids and pheromones, act as photoprotective organ as well as part of a retinal-pineal axis. In birds, this glad was reported first in sparrow in 1918. The Harderian gland is covered by capsule and the connective tissue septa that divide the gland into numerous unequal-sized lobes and lobules. Plasma cells are found in the interacinar space and the lumina of lobules. The recent studies suggest that the Harderian gland act as an immunopotent organ in birds, and that the gland in scavenging birds contains more immunoglobulin-containing plasma cells due to their scavenging nature. Moreover, this gland shows considerable species/strain differences in terms of macro anatomy, microanatomy as well as in the dynamics of immunoglobulin-containing plasma cells among different birds. In this review, these species and strain differences are discussed based on recent studies and several goals of future research are identified.
INTRODUCTION
Birds are continuously exposed to a wide spectrum of potential environmental immunomodulators including physical and chemical factors as well as various microorganisms and antigenic particles, whether occurring naturally or deliberately introduced. In addition, temperature, housing, air quality, diet, environment contaminants, feed additives, therapeutics and vaccines are significant categories of immunomodulators. To cope with the diversity of potentially harmful agents on one hand, and to mount a protective immune response on the other, birds possess an array of humoral and cell mediated immune mechanisms that destroy infected cells or pathogens and mount an adaptive immune response. For this, immunocompetent defense system is the decisive prerequisite [1, 2]. The lymphoid tissue of the chicken is divided into “central” and ” peripheral” ones. The central lymphoid tissue includes bursa of Fabricius and thymus. The peripheral lymphoid tissue includes the spleen, cecal tonsils and all the mucosa-associated lymphoid tissues including respiratory tract, genitourinary tract, alimentary tract and head associated lymphoid tissues that consists of Harderian gland [2, 3, 4, 5, 6, 7]
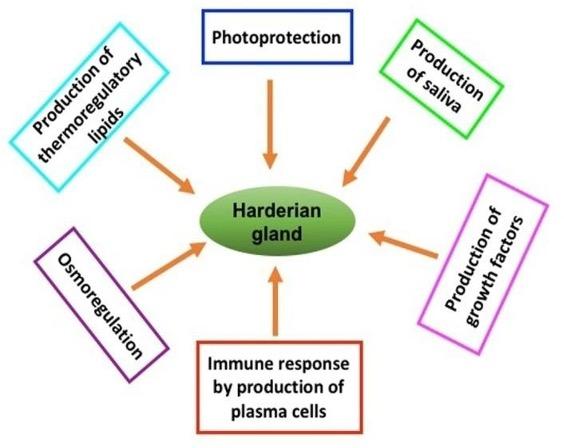
The Harderian gland was first described by Johann Jacob Harder in deer on 1694 and is found in most of the terrestrial vertebrates, amphibians, reptiles, birds and mammals [8]. The Harderian gland forms a unitary structure, which is firmly attached to the medial part of the orbit, and the duct of this gland opens usually on the surface of the nictitating membrane, which is a characteristic feature of this gland and distinguishes from lacrimal gland and the other ocular glands. The later ones are normally classified as a cluster of glandular tissue actually within the nictitating membrane, whereas, Harderian gland is often surprisingly large, in some cases larger that eye itself [9], with diverse functions (Figure 1).
Avian Harderian gland was first described in sparrow (Passer domesticus) in 1918, which was located at the variable aspects of orbit [3, 9, 10]. The Harderian gland is relatively larger in the fowl, much larger than the lacrimal gland. In birds, the usual function of this gland is to lubricate the surface of the eyeball and nictitating membrane [11]. In addition, the Harderian gland possesses numerous large plasma cells specific for anti-immunoglobulin (Ig) A, -IgG or -IgM marker in the interstitial stroma of the chicken that plays a part in immunological defense of the para ocular region [10, 12, 13]. Many other authors also suggest that the Harderian gland is responsible for the local immunity of the eye orbit [14, 15]. At the same time, it is accepted that the gland is a peripheral lymphoepithelial organ, which together with the spleen, the bursa of Fabricius and the caecal tonsils form a system of avian organs that determines both the general and the local immunity [16, 17, 18]. The major classes of Igs produced in chicken Harderian gland are probably related to the secretory feature of this gland [19, 20]
Till to date, Harderian gland of birds has been studied in different species or strains of birds such as chicken, duck, goose, ostrich and Guinea Fowl. The present review is focused on the species or strain differences on macro anatomical, micro anatomical and immunohistochemical studies particularly the differences in the frequencies and distribution of Ig-containing plasma cells.
MACROANATOMY OF HARDERIAN GLAND
Location
Harderian gland of bird shows considerable species differences in location. In White Leghorn chickens the Harderian gland is located in the ventro-medail aspect of the eyeball extending rostrally from the optic nerve [23].In contrast, it has been reported that Harderian gland of native chickens of Bangladeshis situated on the dorsal posterior surface of the eyeball occupying the considerable part of the orbit [3, 4, 10]. While in Rook, this gland is located in the ventral and posterior medial to the eyeball [3]. In the Canadian ostrich, the Harderiangland is located ventromedially around the posterior part of the eyeball [24]. In duck, this gland is located in the anteriomedial part of the orbit [25]
Shape
In White leghorns, the harderian gland is hourglass shaped and in Rook it is tongue shaped [3]. In native chickens of Bangladesh, this gland is triangular in shape [3, 4]. Whereas, in Canadian ostrich, the Harderian gland is flattened, oval shaped, irregular in outline and pointed in the dorsal end [24].
Size
The size of the Harderian gland is also varied among the different types of the birds. In native chickens, the Harderian gland of male bird is 9.2 mm in length and 5.1 mm in breadth, whereas, the Harderian gland of female bird is 9.0 mm in length and 3.2 mm in breadth [4]. The mean dimension of the Harderian gland in White leghorn is 17.66 mm in length and 6.2 mm in breadth [23], which is almost double than that of native chickens. Whereas, Harderian gland of rook is bigger than that of White leghorn [3].It is stated that the length and breadth of Harderian gland in rook is 18mm and 8.6 mm respectively. Interestingly, the size of the Harderian gland in Canadian ostrich is much bigger and it is 35.30 mm in length and 15.65 mm in breadth [24]. There are no significant differences in size of the left and right gland, which is similar in the report of native chickens of Bangladesh [4].
Color
The Harderian gland is covered with a thin layer of fat and after removing the fat this gland of native chickens shows brownish in color [4], while in broiler it is reddish in color [3]. The color of Harderian gland of White leghorn bird is pale pink and rook it is pink yellow [23] and in Canadian ostrich, it is light pink in color [24].
MICROANATOMY OF HARDERIAN GLAND
The Harderian gland of birds is surrounded with a thick connective tissue capsule. From the capsule, the trabeculae enter into the gland, dividing the gland into different sized lobes and lobules. These inter-lobular trabeculae are thinner compared to the capsule surrounded the whole gland and contain collagen fibers, fibroblasts, blood vessels and nerve fibers. The smallest secretory unit of the Harderian gland is called acini lined by simple columnar epithelial cells [3, 10, 11, 24, 25]. There is a variation in length of the lobules in between broiler male and female, and in between broiler male and native male, however, their breadth does not vary [3]. The lumen of the lobules is irregular in native chicken and regular in broiler. This variation in size of the histological lobule of Harderian gland of chickens might be due to strain differences. The lumen of the acini in broiler is spherical and regular or elongated, and the cell boundaries were distinctly visible. Whereas, in native male, the acini are lined by tall single columnar epithelium and the lumen of the acini in these strains of chickens were elongated, irregular and narrower, and the cell boundaries are not distinctly visible. This luminal variation, in regards to its cellular contents and shape in between broiler and native chicken is also possibly due to strain differences [3, 10].
LYMPHOID CELLS IN THE HARDERIAN GLAND
Different types of lymphoid cells such as macrophages, lymphocytes, plasma cells as well as granulocytes are found in the subepithelial layer of the Harderian gland of birds [26, 27]. Although, these cells are often densely aggregated in the central regions of the lobules, individual cells are also occurred near the periphery of the Harderian gland. It has been reported that pre-plasma cells form a desmosome-like contact with macrophages and junctional complexes with each other. In addition, the lymphoid cells were sometimes embedding in a dense homogeneous material, which is also found in the intercellular spaces of the secretory epithelium [28]. In contrast, other research group described that the lymphoid substance of the chicken’s Harderian gland organized in two histologically distinct compartments [29]. Light and electron microscopical investigations reveal that the lymphoid structure of the Harderian gland of chickens organizes in different histological frameworks. In the head the surface epithelium of the central canal can be classified as a lymphoepithelial tissue, which covers the dense lymphoid substance. It consists of small and medium-sized lymphocytes, dendritic-like cells, and occasional macrophages. High endothelial venules are associated with intense lymphocyte migration and homing that give circumstantial evidence for a T-dependent region, as found in a secondary lymphoid organ. The B-dependent germinal centers are also common structural units of the head region’s lymphoid substance. The body of the gland is loaded with plasma cells of different maturation stages. They immigrate into the epithelium of the central canal and produce IgM and IgA and IgG [28, 30]. This plasmocytic region is accounted for the immunosurveillance on the conjunctiva and in the upper respiratory tract through antibody production against bacterial or parasitic infections. In both the head and body regions of the gland, anti-B-L (anti-Ia) antibody recognizes scattered elongated cells that might represent dendritic cells. The immunological relationship between the two histologically different parts of the Harderian gland is unknown, but it is speculated that the dense lymphoid tissue with high endothelial venule receive the blood-borne, immunologically mature, but uncommitted B cells. By the influence of local antigen stimulus, these B cells transform to plasma cells, which gradually appear in the body of the gland. The lymphoid structures of the head and the body fulfill the function of secondary and tertiary lymphoid organs, respectively [29]. The dynamics of IgA-, IgG- and IgM-containing plasma cells also shows strain variations that discussed below.
IMMUNOGLOBULINS-CONTAINING PLASMA CELLS IN THE HARDERIAN GLAND
The immunoglobulins are secreted by Igs- containing plasma cells that perform local or systemic defenses in the body. It has been shown that in the native chickens the frequency of the population of IgA-positive cells is higher followed by IgG and IgM-positive cells and in the broiler chickens the frequency of IgM-positive cells is more followed by IgA- and IgG-positive cells [10]. These observations are similar to the study made by different research groups in chickens [31, 32] but varied greatly with the observations in the White Leghorn chickens [12]. It has been revealed thatin White Leghorn chicken IgG- is more than IgA- and IgM-positive cells. In addition, the frequency of the population of Ig- containing plasma cells is varied between the sexes in the same strain, and also in between different strain of chickens [10]. In the broiler, IgA- and IgG- positive cells are more in the male, whereas, IgM-containing cells are more in female.In the native chickens the frequency of IgA- and IgM-containing cells are more in the female, in contrast, the IgG –positive cells are higher in the male. The frequencies of Ig-containing plasma cells are also varied between sexes. The population of IgA- and IgG-containing cells is significantly higher in the native male than the broiler male chickens. In addition, the frequency of IgA- and IgG-containing cells is more in native female than the broiler female. It has been speculated that these variations concerning the frequency of occurrences of the plasma cells positive for the different classes of immunoglobulins in the Harderian gland of birds might be related to age, strain, species, health condition and their the condition of their surrounding environment. Interestingly, significant differences in lymphatic cells components are also observed between the Harderian gland of wild pheasants and hybrid pheasants [33]. It has been reported that the common wild pheasant presented a higher number of immunoglobulin-producing plasma cells. The hybrid pheasants have a lower number of plasma cells, but a higher number of lymphocytes in the gland tissue in comparison with the common pheasant [33]. In addition, in domestic duck, the IgM-containing plasma cells are the most abundant than other classes of immunoglobulins into the epithelium of the Harderian gland. In contrast, IgY- or IgA-positive cells are predominant in the lamina propria [25].
SECRETORY MECHAMISM OF PLASMA CELL IN THE HARDERIAN GLAND
Interestingly enough, strain variations are also observed in the mode of release of Ig-containing plasma cells. It is reported that secretory IgA-, IgG-, and IgM-containing plasma cells are released by the disintegration of the acinar cells at the apical part of the lobules of the broiler but by dissolution or holocrine mode of secretion in the native chicken [10]. The secretory mechanism is merocrine in rabbit [33] merocrine to apocrine in the other avian species [25]. In a very recent study in Canadian ostrich it is also observed apocrine in nature [24]. Among the available studies on native chickens of Bangladesh showed holocrine type of secretion for plasma cell in the Harderian gland.
DIRECTIONS FOR FUTURE STUDY
The comparative ontogeny of Harderian gland of birds has not studied yet. The comparative trajectory of different classes of Ig-containing plasma cells in the Harderian gland of different species of birds will also need to be clarified by future study. In addition, the further knowledge, component and function of Harderian gland as a part of immune system will also need to be clarified.
CONCLUSION
Harderian gland of birds, even though it is not a primary lymphoid organ as a whole, but act as an immunopotent organ, and that showed considerable species or strain variations in anatomical or histological structures as well as in the dynamics of Ig-containing plasma cells (Table 1). The wild type or domestic birds contained more Igs-containing plasma cells possibly due to their scavenging in nature. The current review could also serve as a caution that data on morphology or frequencies of Igs-containing plasma cells of Harderian gland from one species should not always be directly applied to others among birds.
Table 1. Species differences in gross and histochemical parameters of Harderian gland of birds [3, 4, 9, 10, 11, 23, 2].
CONFLICT OF INTEREST
The author declares that no conflict of interest exists.
References
- [1]Jeurissen SHM, Claassen E, Janse EM. Histological and functional differentiation of non-lymphoid cells in the chickens spleen. Immunology.1992; 77: 75-80.
- [2]Jeurissen SHM, Vervelde L, Janse EM. Structure and function of lymphoid tissues of the chicken. Poultry Sci. Rev.1994; 5: 183-207.
- [3]Jahan MR, Khan MZI, Islam MN, Akter SH.Morphometrical analysis of Harderian gland of broiler and native chickens of Bangladesh. J Bangladesh Agril Univ. 2006; 4(2): 273-278
- [4]Jahan MR, Islam MN, Khan MZI. Gross and biometrical study of the Harderian gland in adult native chickens of Bangladesh.Int J Biol Res. 2009; 7: 31-34.
- [5]Islam MN, Khan MZI, Jahan MR, Karim MR, Kon Y. Comparative studies of mucosa and immunoglobulin-containing plasma cells in the gastrointestinal tract of broiler and native chickens of Bangladesh. J. Poult. Sci. 2008; 45: 125-131.
- [6]Islam MN, Khan MZI, Jahan MR, Fujinaga R, Yanai A, Kokubu K, Shinoda K. Histomorphological study on prenatal development of the lymphoid organs of native chickens of Bangladesh. Pak. Vet. J. 2012; 32: 175–178.
- [7]Islam MN, Khan MZI, Jahan MR, Shinoda K. Developmental trajectory of the prenatal lymphoid organs in native chickens:a macro anatomical study. Asian J. Med. Biol. Res. 2017; 3(4): 432–436.
- [8]vanGinkel FW, Tang DC, Gulley SL, Toro H. Induction of mucosal immunity in the avian Harderian gland with a replication-deficient Ad5 vector expressing avian influenza H5 hemagglutinin. Dev Comp Immunol. 2009; 33: 28-34
- [9]Payne AP. The Harderian gland: a tercentennial review. J. Anat. 1994;185: 1-49.
- [10]Khan MZI, Jahan MR, Islam MN, Haque Z, Islam MR, Kon Y. Immunoglobulin (Ig)-containing plasma cells in the Harderian gland in broiler and native chickens of Bangladesh. Tissue and cell. 2007; 39: 141–149.
- [11]Mobini B. Histological and histochemical studies on the Harderian gland in native chickens. Vet Med. 2012; 8: 404-409.
- [12]Nasrin M, Khan MZI, Siddiqi MNH, Masum MA. Mobilization of immunoglobulin (Ig)-containing plasma cells in Harderian gland, cecal tonsil and trachea of broilers vaccinated with Newcastle Diseases Vaccine. Tissue and Cell. 2013; 45: 191-197.
- [13]Bejdic P, Avdic R. Amidzic L, Cutahija V, Hadziomerovic N, Katica A, Mlaco N. Ultrastuctural of plasma cells in harderian gland of laying hen. AntatHistolEmbryol. 2017; 47: 46-50.
- [14]Baba T, kawata T, Masumoto K, Kajikawa T. Role of the Harderian gland in immunoglobulin A production in chicken lacrimal fluid. ResVet Sci. 1990; 49: 20-26.
- [15]Ohshima K, Hiramatsu K. Immunohistochemical localization of three different immunoglobulin classes in the Harderian gland of young chicken. Tissue cell. 2002; 34 (2): 129-133.
- [16]Shirama K, SatohT, Kitamura T, Yamade J.The avian Harderian gland: Morphology and Immunology. MicroscRes and Tech. 1996; 34: 16-27.
- [17]Korbel R, Schaffer E, RavelhoferK, Kosters J. (1997). Ocular Manifestation of mycobacterium infections in bird.TierartzlParxAusg K. 1997; 25: 552-558.
- [18]Fix AS, Arp LH. Morphologic characterization of conjunctiva- associated lymphoid tissue in chicken. Am J Vet Res. 1998; 52 (11): 1852-1859.
- [19]Albini B, Wick G, Rose E. Immunoglobulin production in chicken Harderian gland. Int Arch Allergy.1974; 47: 23-34.
- [20]Baba T, MasumotoK, Nishida S, kajikawa T, Mitsui. Harderian gland dependency of immunoglobulin A production in the lacrimal fluid of chicken. Immunology.1988; 65(1): 67-71.
- [21]Rahman ML, Islam MR, Asaduzzaman M, Khan MZI. Lymphoid tissues in the digestive tract of Deshi Chicken (Gallus domesticus) in Bangladesh. Pak J Biol Sci. 2003; 6: 1145-1150.
- [22]Islam MN, Khan MZI, Jahan MR, Fujinaga R, Shinoda K. Ontogenic development of immunoglobulins (Igs)-positive lymphocytes in the lymphoid organs of native chickens of Bangladesh. Int. J Vet Sci Med. 2013; 1: 96-101.
- [23]Ashok P, Venkatkrishnan A, Vijayaragavan C,Ramkrishna V. Gross morphology and biometrical study of the Harderian gland in White Leghorn birds. Indian J Anim Sci. 2000; 15(2): 217-219.
- [24]Frahmand S, Mohammadpour AA. Harderian gland in Canadian Ostrich (Struthiocamelus): A morphological and histochemical study.Antat Histol Embryol. 2015; 44: 178-185.
- [25]Oliveria CA, Telles FL, Oliveria AG, Kalapothakis E, Goncalves-Dornelas H, Mahecha GAB. Expression of different classes of immunoglobulin in intraepithelial plasma cells of the Harderian gland of domestic ducks Anasplatyrhynchos. Vet ImmunolImmunopathol. 2006; 113: 257-266.
- [26]Bejdic P, Avdic R. Amidzic L, Cutahija V, Tandir F, Hadziomerovic N. Developmental changes of lymphoid tissue in the Harderian gland of laying hens. Mac Vet Rev. 2014; 37: 83-88.
- [27]Scott TR. Our current understanding of humoral immunity of poultry. Poultry Sci. 2004; 83: 574-579.
- [28]Khan MZI, Akter SH, Islam MN, Karim MR, Islam MR, Kon Y. The Effect of Selenium and Vitamin E on the Lymphocytes and Immunoglobulincontaining Plasma cells in the Lymphoid organ and Mucosa-Associated Lymphatic Tissues of Broiler Chicken. AnatHistolEmbryol. 2008; 37: 52-59.
- [29]Olah I, Kupper A, Kittner Z. The lymphoid substance of the chicken’s Harderian gland is organized in two histologically distinct compartments.Microsc Res Tech.1996; 34(2):166-76.
- [30]Masum MA, Khan MZI, Nasrin M, Siddiqi MNH, Khan MZI, Islam MN, Detection of immunoglobulins containing plasma cells in the thymus, bursa of Fabricius and spleen of vaccinated broiler chickens with Newcastle disease virus vaccine, Int J Vet Sci Med. 2014; 2 (2): 103-108.
- [31]Bienenstock J, Gauldie J. Perey DYE.Synthesis of IgG, IgA and IgM by chicken tissues.J Immunol. 1973; 111: 1112-1118.
- [32]Klećkowska-Nawrot J, Goździewska- arłajczuk K, Kowalczyk A, Łukaszewicz E, Nowaczyk R. Structural Differences of the Harderian Gland between Common Pheasants (PhasianusColchicusTalischensis) and Hybrids of Italian Amber and Common Pheasants.Rev. Bras. Cienc. Avic. 2016; 18 (2): 309-318
- [32]Kuhnel W. Structural and cytochemical study of Harderian gland of Druse von kaninchen, Z. Zelloforsch.Mikrosk. Amt.1971; 119: 384-404.