Isolation of stem cell populations from wharton’s jelly sections of umbilical cord and comparison analysis with cord blood stem cells
Abstract
Wharton’s jelly (WJ) is a gelatinous tissue exists in the umbilical cord contains stromal cells, termed as mesenchymal stromal cells (MSCs). Current study designed to characterize the stem cell population both within the cord blood and it’s wharton’s jelly tissue and it’s correlation to neonatal parameters. Both wharton’s jelly and cord blood and their CD34+ and CD90+ expression was enumerated by flow-cytometry. Neonatal parameters along with the mode of delivery were analyzed to correlate with CD34 and CD90 markers. From a total of 20 deliveries, CD34 and CD90 expression pattern showed varied in both samples. Gender and the expression of CD34 and CD90 markers doesn’t show any significant change. Whereas, correlation analysis of weight with CD34+ and CD90+ cell counts showed significant difference and it may have the effect on both cell counts. Neonatal parameters especially birth weight may have the effect on cell count of CD34 and CD90 in both CB and WJ. Current study provide a fast method for mesenchymal cell isolation from Wharton’s jelly tissue using the total length of the cord, which may be easily applied along with the UC blood, for the banking of UC.
INTRODUCTION
In the recent years, there seems to be an unbounded interest regarding mesenchymal stem cells. MSC from the bone marrow (BM) is still considered as the gold standard for various clinical applications and stem cell engineering. But the bone marrow has limitations as MSCs source, like mesenchymal stem cell low frequency, painful procedure and the decline in MSC characteristics with donor’s age [1]. Kobayashi et al., [2] demonstrated that umbilical cord which may be discarded during delivery can be provided as an inexhaustible source for stem cells and they shows the similar characteristics like MSCs from bone marrow. Wharton’s jelly derived MSCs have advantageous characteristics like plenty source, rapidly available donor, non-invasive collection and painless procedure, ethically noncontroversial and doesn’t shows any teratoma [3]and this may have immune privileged and immunomodulatory phenotype capable of suppressing the immune response in vitro. Whereas bone marrow MSC (bMSC) may shows notable changes with increasing patient’s age, reduction in the available cell number, longer doubling times and lower differentiation potential in vitro. Wharton’s jelly stem cells shown to be differentiated into osteoblast [4], chondrocyte [5], cardiomyocyte [6], skeletal myoblast [7], hepatocyte cells [8], endothelial cell [9], neural cell & adipocyte [10], dopaminergic cell [11] andlens fiber cell [12]. Wharton’s jelly stem cells shows positive forCD44, CD105 [7, 13], CD68 [14], CD13 and CD95 markers, but negative for CD34, CD45, CD38 and CD71 markers. Wharton’s jelly MSCs are primitive MSCs than other tissue sources and theydon’t express the major histocompatibility complex (MHC) class II HLA-DR [15]. The important graft-vs-host disease (GVHD) markers like CD86, CD80 and CD40 were not noticeable or weakly expressed in WJMSCs [16]. CD34+ and CD90+ marker analysis was useful for the examination of the quality of cord blood for future any hematological and non-hematological disease therapeutics. Still there was little or no knowledge with regard to neonatal characteristics and their effect on CD34 and CD90 cell population in WJ and CB. Hence current study designed to analyze the cell population from complete section of umbilical cord wharton’s jelly and it’s comparison to the stem cells population derived from cord blood. Also we evaluated the cell counts of each of the marker of fresh cord blood samples and WJ sample and were correlated between their expressions with neonatal parameters.
MATERIALS AND METHODS
Materials
Fetal bovine serum, streptomycin, trypan blue and phosphate-buffered saline pH 7.4 (Gibco, USA). Collagenase type II, Bovine serum albumin, DMEM-F12 Medium (Sigma-Aldrich, USA).CD133, CD90 Antibodies conjugated microbeads kits and miniMACS magnetic column with separation system from Miltenyi Biotech, Germany. All chemicals & Reagents are of molecular biology grade.
Collection of Wharton’s jelly tissue material in sterile condition
Normally, cords and it’s blood will be thrown after the delivery in many settings, very few centers will train the parents to store these sources. By information, we used the clinical material which is normally going to wastage. Preparation of Human umbilical cord samples was aseptically collected after delivery full-term by normal labor/C-section at Dept of obstetrics & gynaecology, Narayana Medical College Hospital and the protocol is approved by the Institutional Ethics Committee. After the delivery of the baby, the cord is collected in a sterile flask containing PBS supplemented with 100 U/mL penicillin, 0.1 mg/mL streptomycin, and 0.25 mg/mL amphotericin B. Cords were processed within 12 hr of birth in aseptic condition under Bio Safety Cabinet. Immediately, cords transferred to the laboratory and washed twice with PBS to remove remnant blood. The net weight of each cord was measured. Length of the cord is estimated. Cord is processed in a sterile 10 cm petridish; cord is divided into four sections according to its length from placenta to baby side. Each sectioned cord was spliced into 3–5 cm long pieces. Horizontal sections of each piece are engraved then cut open lengthwise with scissors and the outer sheath of umbilical cord sections peeled to extract its inner surface with WJ. The exposed vein and arteries removed by pulling with forceps and peeled away from the WJ. This tissue was chopped into small pieces placed into 50 ml centrifuge tube and digested with collagenase enzyme solution for 2 hrs at 37° C with constant agitation. The cord pieces are crushed using cell scrappers to disintegrate tissue into cell suspension. This cell suspension is moved to a new sterile 50ml centrifuge tube containing trypsin enzyme solution with constant agitation for 5minutes and stopped by adding Bovine serum albumin. After the second enzymatic digestion is complete, the cord pieces are scrapped with cell scrapper through 40micron filter to remove as many cells from Wharton’s jelly as possible. Cell suspension was centrifuged at 1000 rpm for 5 min and 10 ml of DMEM-F12 medium is added to the cell pellet. Cells were counted using a hemocytometer.
Collection of cord blood before placental delivery (in utero)
The cord was clamped immediately after the delivery and iodine swab done. The umbilical cord blood was collected from the vein by gravity in a sterile 100 ml collection tube containing layered with heparin. This process was performed by trained obstetric staffs. Cord blood was stored at 4°C until processing.
After placental delivery (ex utero) collection
Immediately after the placental delivery, the placenta is placed in a sterile tray with a central hole that allows the umbilical cord to hang down. Ubilical cord cleaned with an iodine swab, cord blood was collected from the umbilical vein in a sterile collection 100 ml tube layered with heparin and stored at 4°C until processing. For both methods, obstetric and clinical maternal chart contains route of delivery, gestation number, placental weight, sex and weight were recorded.
Mononuclear cell (MNC) separation
A range of 55-80 ml of cord blood was collected in 100ml tubes with heparin. Cord blood subjected to density gradient centrifugation procedure using Histopaque 1077. Briefly, cord blood was diluted equal volume of PBS and layered on Histopaque in 50 ml. centrifuge tube and centrifuged at 850xg for 20 minutes. The mononuclear cell layer was separated, washed twice with PBS and resuspended in 1ml of PBS. The cells were counted using haemocytometer. The MNCs were subjected to Magnetic sorting.
Flow cytometry
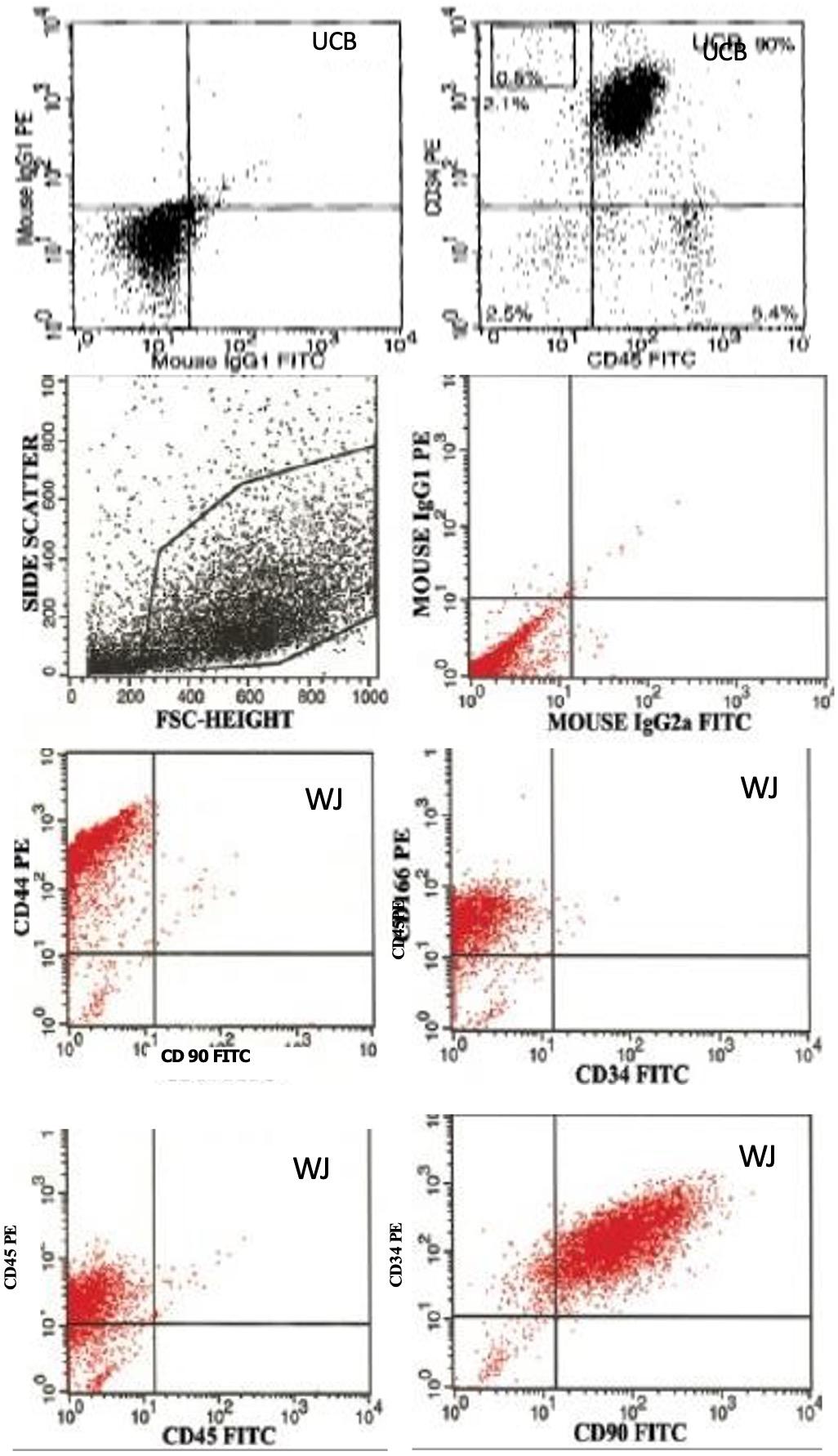
Cord blood and WJ tissue samples were analyzed for the expression of CD34 and CD90 makers. The MNCs were stained with anti-human monoclonal antibodies i.e., FITC conjugated CD34 and PE-conjugated CD90 (BD Biosciences), after which they were kept in refrigerator at 4 °C for 20–30 minutes by flow cytometry analysis by BD FACS Canto II with reference to mouse isotypic Immunoglobulin as control. WJ samples were also analyzed as above procedure and the data was analyzed using BD FACS DIVA Software.
Immunomagnetic separation
MNCs collected from cord blood subjected to Magnetic activated cell sorting (MACS) by using positive selection to CD34 phenotypic markers. According to the manufacturer’s instructions, 50 to 200 x 106 mononuclear cells in PBS and 0.5% BSA were incubated for 15 min at 4°C with a blocking reagent (human IgG) and simultaneously with anti-human CD34 antibody. After incubation, the cells were washed to remove unbound antibody and loaded in LS+ column separation. Labelled cells by positive selection were collected by flushing LS+ column with 5 ml DMEM medium. The cell viability was checked using Trypan blue dye exclusion assay and cell count performed by using a hemocytometer. CD90 positive cells were also collected using the same procedure.
Cell viability and enumeration
The stock (0.4 %) was diluted with PBS to 0.2 %. Cell suspension and 0.2 % trypan blue solutions were mixed at 1:1. Dilution factor is 5. The tip of the pipette is placedin the V-shaped groove on the hemacytometer to load the sample into the chamber (about 15 microliters) Capillary action will draw the fluid into the chamber. The sample is allowed to settle for 1- 2 minutes so that the cells stop drifting around the chamber and most will be in the same plane of focus. To avoid drying, the hemacytometer was placed in a petri dish containing a moistened filter paper. Total and viable cells were observed and counted in nine squares of hemacytometer. Live cell count divided by total cell count is Cell viability. Viable cell count / Quadrants counted x Dilution factor x Hemocytometer factor x Current volume (mL) = Viable cell yield. Here, hemocytometer factor is 10000.The correlaion between stem cell markers (CD34+ and CD90+) with neonatal parameters. Statistical test was performed by statistical software using SPSS 20.0 version. P value < 0.05 was considered significant.
RESULTS
Clinical characteristics
Data about umbilical cord (UC), and its cord blood were obtained from 12 vaginal and 8 caesarean sections. Eight cases were excluded from CB units before processing in both vaginal and caesarean sections. The cord blood collected ranged from 55-80 ml from 12 umbilical veins. Placental weight observed to be ranged from 418-540 grams. A total of 12 cords were collected and the mean weight per length of cords were of 1.2 g/cm (range 0.7–1.75 grams/cm). The clinical characteristics of Umbilical cord samples were mentioned in Table 1.
CD34+ cells in Cord Blood and WJ tissue
After density separation, Total MNCs obtained ranged from umbilical cord blood were 3.5 x 106 to 4.1 x 108 perml. The mononuclear cell purity observed as 68% from umbilical cord blood sample. After magnetic separation, the purity of cord blood CD34+ cells shows ~85%. The concentration of CD34+ cells sorted cells from umbilical cord blood mononuclear cells ranged from 2.42 x106 to 3.65×106. Cells show a mean of 94% viability after Histopaque density gradient separation of umbilical cord MNCs and WJ of umbilical cord blood.
Table 1. Characteristics of UC samples that generated MSCs
CD90+ population from WJ
CD90 cells were isolated from 12 numbers of cords and counted on a hemocytometer. Cell yield was calculated at 27×104 viable cells per centimeter length of cord (range 80- 469×103 cm–1 cord) the total mean cord length is 48 cm. Viability of CD90+ was observed as 92±3% (mean± SD (Table 2). The purity observed CD90+ cells were 96.4±1.4% after MACs sorting.
Table 2. CD90 phenotype marker sorting levels in various sources.
CD34 and CD45 markers in cord blood and WJ
The mean percentage CD34+cell count was 1.02 ± 0.72 in cord blood in recovery. Hematopoetic stem cell [CD34+/CD45–] cell number observed as 35 – 250 cells in cord blood. Whereas there was negligible or nil expression of CD34+CD45 cell number observed in WJ samples i.e. 0.3 ± 0.3.
CD34 and CD90 in cord blood and WJ
The mean percentage CD90+ cell count was 13.5 ± 2.72 in WJ tissue in recovery. These cells showed 70-80% positivity with CD90 not with CD34 and CD45. There was CD90+CD45–CD34– cell number 30-110 in observed cord blood. The mean percentage of CD90+ was 0.23 ±0.17 in cord blood [Figure 1]. Correlation of CD34 and CD90 with neonatal variables in c-section delivery, there was an increased expression of CD34 and CD90 when compared to the vaginal delivery without significant difference in both cell counts. Baby’s gender doesn’t shown association with number of CD34 and CD90. The mean CD34+ and CD90+ cell counts between the two delivery groups of birth weight was compared using post hoc analysis, which showed that increased cell counts with increasing birth weight. Birth weight with CD34+ cell count showing correlation coefficient of 0.909 with p value 0.0001. Birth weight with CD90+ cell count showing correlation coefficient of 0.890 with p value 0.0001. Hence birth weight may be act as significant predictors of both CD34+ and CD90+ cell yield of the cord blood and WJ tissue.
Correlation of CD34 and CD90 with neonatal variables
In c-section delivery, there was an increased expression of CD34 and CD90 when compared to the vaginal delivery without significant difference in both cell counts. Baby’s gender doesn’t shown association with number of CD34 and CD90. The mean CD34+ and CD90+ cell counts between the two delivery groups of birth weight was compared using post hoc analysis, which showed that increased cell counts with increasing birth weight. Birth weight with CD34+ cell count showing correlation coefficient of 0.909 with p value 0.0001. Birth weight with CD90+ cell count showing correlation coefficient of 0.890 with p value 0.0001. Hence birth weight may be act as significant predictors of both CD34+ and CD90+ cell yield of the cord blood and WJ tissue.
DISCUSSION
The human umbilical cord contains distinct anatomical regions, two umbilical arteries, umbilical vein, cord lining and Wharton’s jelly. Each of these regions comprises fibroblast like MSCs. Cells isolated from arteries, vein, cord lining and wharton’s jelly have the capacity of plastic adherence, multipotent and differentiating ability to cell types such as osteoblasts, adipocytes, chondrocytes, hepatocytes, and neural and cardiac cells. They express markers like bone marrow derived MSC, negative for haematopoietic and macrophage markers. In the current study, WJ tissue derived cells shown positive to CD90 and negative to CD34 and CD45 markers respectively. It indicates WJ derived cells fulfill the mesenchymal stem cell lineage. Whereas, cord blood sample shown an opposite pattern of expression. CB shown positive for CD34 and negative or less expression of CD45. It indicates that CB derived cells are of hematipoetic lineage.
Previous studies have succeeded in the establishment of correlation of the hematopoietic stem cell content with the neonatal, obstetric and other factors. The present study designed to characterize the CD34+ cell content (hematopoetic) and CD90+ (mesenchymal) cell content in cord blood and wharton’s jelly tissue of the same pregnant with respect to the major neonatal parameters. In this study, there was significant impact of birth weight on CD34+/CD45- hematopoetic stem cell content of the cord blood, which also been reported by other study was found to be supportive to the present observation [17]. Though birth weight was correlated significantly with CD34+/CD90+ cell counts, the increasing gestational age doesn’t showing significant increase in CD34+/CD90+ cell counts in the current study. Babies with higher weight shown higher CD34 expression in cord blood and also higher CD90 in WJ derived cell counts. Mode of delivery doesn’t shown any influence on hematopoetic and mesenchymal stem cell yield in our study, which also been supported by the previous study [18]. Whereas, another study observed more progenitor cell content especially CD34+ cell concentration in case of cesarean mode of delivery than the vaginal delivery mode [19]. In the current study, it was an attempt to made or formulate the guidelines related to maternal and neonatal parameters to store the cord blood sample or tissues before collecting the cord blood or tissue in India. Limitation of this study was small sample size, hence the observations should be validated in long term prospective studies in the country.
Unlike embryonic stem cells, adult stem cell usage in research and therapeutics doesn’t have any controversy because it does not require the creation/destruction of an embryo for the production of adult stem cells. In recent years, there seems to be an unbounded interest concerning mesenchymal stem cells (MSCs). Reason due to this is there are some exciting characteristics of MSC, which includes long-term ex vivo proliferation, multi-lineage potential and immunomodulatory properties. Bone marrow has some limitations as MSCs sources, including mesenchymal stem cell low frequency, isolation procedure is painful and the decline in MSC characteristics with donor’s age [20]. Kobayashi et al [2] demonstrated that umbilical cord which is discarded during the birth may have an inexhaustible stem cells source and showed similar characteristics with the MSCs from bone marrow. MSCs from Wharton’s jelly tissue may serve as large, rapidly available and ethically noncontroversial and doesn’t induce teratomas [3]. WJ tissue derived MSCs are more primitive MSC than other tissue sources and doesn’t express MHC class II (HLA-DR) antigens [15]. During embryonic development, wharton’s jelly is derived from extra-embryonic mesoderm and partly from the embryonic mesoderm.
The isolation of MSCs from individual cord regions including cord vein and the perivascular region of Wharton’s jelly and cord blood has been reported by many groups. However, very few/none of the studies or indeed any other studies have compared all cord regions of the same pregnant individual. We described the characterisation of MSCs obtained from each of the distinct regions carefully dissected from the same umbilical cord, providing a direct and relevant comparison of cells with the potential for tissue engineering and repair.
CONCLUSION
In this study MSCs from four distinct regions of the same cord (artery, vein, Wharton’s jelly, and cord lining), in addition to cord blood cells from same individual from the whole cord, have been isolated and compared. The MSCs, which express CD90 from the richest site of the umbilical cord (Wharton’s jelly) can only be isolated using enzymatic digestion. Cells from whole cord and cells isolated from individual cord regions have potential as a useful source for obtaining promising cell populations for further study. The present study also suggests positive association between hematopoietic stem cell content of the cord blood and the neonatal factor especially birth weight. Mesenchymal stem cell content also has shown positive correlation with birth weight.
ACKNOWLEDGEMENT
This research does not receive any fund from public or private organization. Authors acknowledge to Narayana Medical College & Hospitals for providing reagents and infrastructure.
CONFLICT OF INTEREST
The author declares that no conflict of interest exists.
References
- [1]D’Ippolito G, Schiller PC, Ricordi C, Roos BA, Howard GA. Age‐related osteogenic potential of mesenchymal stromal stem cells from human vertebral bone marrow. Journal of Bone and Mineral Research. 1999;14:1115-22.
- [2]Kobayashi K, Kubota T, Aso T. Study on myofibroblast differentiation in the stromal cells of Wharton’s jelly: expression and localization of alpha-smooth muscle actin. Early Hum Dev. 1998;51:223-33.
- [3]Fong CY, Chak LL, Biswas A, Tan JH, Gauthaman K, Chan WK, et al. Human Wharton’s jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Reviews and Reports. 2011;7:1-16.
- [4]Sarugaser R, Lickorish D, Baksh D, Hosseini MM, Davies JE. Human umbilical cord perivascular (HUCPV) cells: a source of mesenchymal progenitors. Stem cells. 2005;23:220-9.
- [5]Wang L, Detamore MS. Insulin‐like growth factor‐I improves chondrogenesis of predifferentiated human umbilical cord mesenchymal stromal cells. Journal of Orthopaedic Research. 2009;27:1109-15.
- [6]Pereira WC, Khushnooma I, Madkaikar M, Ghosh K. Reproducible methodology for the isolation of mesenchymal stem cells from human umbilical cord and its potential for cardiomyocyte generation. Journal of tissue engineering and regenerative medicine. 2008;2:394-9.
- [7]Conconi MT, Burra P, Di Liddo R, Calore C, Turetta M, Bellini S, et al. CD105 (+) cells from Wharton’s jelly show in vitro and in vivo myogenic differentiative potential. International journal of molecular medicine. 2006;18:1089-96.
- [8]Campard D, Lysy PA, Najimi M, Sokal EM. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology. 2008;134:833-48.
- [9]Chen M-Y, Lie P-C, Li Z-L, Wei X. Endothelial differentiation of Wharton’s jelly–derived mesenchymal stem cells in comparison with bone marrow–derived mesenchymal stem cells. Experimental hematology. 2009;37:629-40.
- [10]Karahuseyinoglu S, Cinar O, Kilic E, Kara F, Akay GG, Demiralp DÖ, et al. Biology of stem cells in human umbilical cord stroma: in situ and in vitro surveys. Stem cells. 2007;25:319-31.
- [11]Fu YS, Cheng YC, Lin MYA, Cheng H, Chu PM, Chou SC, et al. Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: potential therapeutic application for Parkinsonism. Stem cells. 2006;24:115-24.
- [12]Maleki M, Parivar K, Nabiyouni M, Yaghmaei P, Naji M. Induction of alpha-crystallins expression in umbilical cord mesenchymal stem cells. Iranian Journal of Ophthalmology. 2010;22:67-71.
- [13]Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, et al. Mesenchymal stem cells in the Wharton’s jelly of the human umbilical cord. Stem cells. 2004;22:1330-7.
- [14]La Rocca G, Anzalone R, Farina F. The Expression of CD68 in Human Umbilical Cord Mesenchymal Stem Cells: New Evidences of Presence in Non‐Myeloid Cell Types. Scandinavian journal of immunology. 2009;70:161-2.
- [15]Troyer DL, Weiss ML. Concise Review: Wharton’s Jelly‐Derived Cells Are a Primitive Stromal Cell Population. Stem cells. 2008;26:591-9.
- [16]Ma L, Feng X, Cui B, Law F, Jiang X, Yang L, et al. Human umbilical cord Wharton’s Jelly-derived mesenchymal stem cells differentiation into nerve-like cells. CHINESE MEDICAL JOURNAL-BEIJING-ENGLISH EDITION-. 2005;118:1987.
- [17]Wen S-H, Zhao W-L, Lin P-Y, Yang K-L. Associations among birth weight, placental weight, gestational period and product quality indicators of umbilical cord blood units. Transfusion and Apheresis Science. 2012;46:39-45.
- [18]Lim FT, Van Winsen L, Willemze R, Kanhai HH, Falkenburg JH. Influence of delivery on numbers of leukocytes, leukocyte subpopulations, and hematopoietic progenitor cells in human umbilical cord blood. Blood Cells. 1994;20:547-58.
- [19]Aroviita P, Teramo K, Hiilesmaa V, Kekomäki R. Cord blood hematopoietic progenitor cell concentration and infant sex. Transfusion. 2005;45:613-21.
- [20]Chen MY, Lie PC, Li ZL, Wei X. Endothelial differentiation of Wharton’s jelly-derived mesenchymal stem cells in comparison with bone marrow-derived mesenchymal stem cells. Experimental hematology. 2009;37:629-40.