Role of auxin and nitric oxide on growth and development of lateral root of plants: possible involvement of exogenously induced Phot1
Abstract
Development of root architecture including lateral root formation is a critical event for successful growth of plants. Auxin promotes the production of nitric oxide (NO) in roots, which is required for auxin-mediated root organogenesis. Inhibition of auxin transport reduced phototropin1 (Phot1)-GFP expression at the basal and central part of LRs, indicating role of auxin in Phot1 expression in lateral roots. However, the involvement of auxin-mediated NO on Phot1 expression in LR growth and development yet to be explored. This review gives an insight that auxin-mediated NO may lead to LR growth and development through interacting with Phot1.
Keywords
LATERAL ROOT IN PLANT GROWTH AND DEVELOPMENT
Lateral root (LR) development in Arabidopsis can be used as a model for the study of factors that regulate organogenesis of plants. Development of root architecture including LR formation is a critical event for successful growth of plants [1]. Whereas the structure of the LR itself is highly predictable, the number, placement and direction of growth of LRs in the system are all highly variable. Expansion of LRs in root system help to increase the surface area for absorption of water/nutrient from environment and enrich the capability to anchorage [2] ;[3]. Generally, LR primordia and the youngest LRs are located close to the root tip as well as older LRs are come upon higher in the root [4]. In most of the plants, LRs grow horizontally and then start to grow at vertically and however, this phenomenon is not because of gravitropic response in Arabidopsis plants [5].
PHOTOTROPIN1 IN PLANT GROWTH AND DEVELOPMENT
Plant growth is regulated by many kinds of factor including environmental and climatic. Generally, plants respond to stimuli or signals from their environment in order to live successfully. These include light, wind and gravity. Among them, light is the most important environmental signal that is involved in various responses. After receiving signals by photoreceptor, the plant converts them into different physiological responses. The nature of the photoreceptor depends upon the stimulus and the receptor is a pigment/molecule that absorbs light.
Phototropism is a process through which plant organs can respond to changes in light direction to maximize photosynthetic potential, and support root growth for water/nutrient acquisition [6]. The photoreceptor kinase phototropin 1 (Phot1) was the first of two phototropin photoreceptors mediating phototropism and other blue light responses described [7]. The A. thalianaPhot1-5 mutant was characterized as defective in both shoot and root phototropism [8]. Galen and co-workers showed that the abundance of blue light receptor Phot1 in roots is correlated with enhanced root growth efficiency [9]. As in the case for positive shoot phototropism in etiolated Arabidopsis hypocotyls, negative root phototropism is mediated by Phot1[8]. Another group reported that the endogenous Phot1 has negative effect on LR formation and root growth [10]. Inhibition of auxin transport by NPA reduced Phot1-GFP expression at the basal and central part of LRs, indicating role of auxin in Phot1 expression in LRs [11].
AUXIN IN PLANT GROWTH AND DEVELOPMENT
In multicellular organisms, growth and development, including proper pattern formation and organogenesis, must be tightly regulated. In plants, the phytohormone auxin plays a prominent role in controlling nearly every step in growth and development [12]. Additionally, auxin is an important mediator of LR development [13]; [14]. Considerable progress has recently been made describing the role of auxin during LR formation in A. thaliana [15]; [16]. In Arabidopsis, mutation in auxin regulatory genes are shown to arrest LR development at various stages [17].
The light environment has been known to regulate polar auxin transport that drives developmental programs of plants [18]. Pre-initiation, initiation, and post-initiation are considered as three major steps of LR formation [16]. The precise role of auxin deposition has been observed in LR initiation as well as in the production of a new root [19], and also measured in both pre- and post initiation actions including emergence [16]. LR initiation is regulated by auxin originating from the root tip [20] ; [21] , whereas emergence depends exclusively on auxin derived from the shoot [22]. In addition, after emergence LRs can synthesize their own auxin [23]. Further, inhibition of emergence is involved with elimination of the leaves and cotyledons [24]. Additionally (Utsuno et al.[25] and Marchant et al. [26] demonstrated that auxin transport plays an important function in root growth and development. Further, application of auxin transporter, IAA to plants enhance LR development and LR elongation [25]; [26]; [27]. On the other hand, use of auxin-transport inhibitors, NPA reduces the number of LR in tomato grown on agar [27]. Further, development of LR may be affected by variation in auxin transports and inhibitors. Normally, blue light induces Phot1 expression which ultimately inhibits LR formation through decreasing the effects of auxin. Phot1 and phot2 both are functional serine/threonine protein kinase photoreceptors [28]. The phototropins absorb blue light and activate the kinase domain [7]. Though perception of directional blue light leads to phototropic curvature through differential accumulation of auxin [29], the mechanism by which phototropin activation initiates signaling leading to this signal output remains elusive. However, recent report suggest that exogenous auxin and Phot1 directly helps in formation of blue light through binding auxin to Phot1 promoter region and mediates transcription of various effects on blue light formation [11]. Additionally, inhibition of auxin transport by NPA reduces Phot1-GFP expression at the basal and central part of LRs, indicating role of auxin in Phot1 expression in LRs [11].
In addition, fungal contamination is a common problem in culture of Arabidopsis seedling in laboratory condition and many phytopathogenic fungi produce most common natural auxin (indole-3-acetic acid, IAA) in culture [30] ; [31]. Therefore, it is suggested that the effects of fungal contamination on expression of 1 in LR at different developmental stages may occur. Surprisingly, we found that fungal contamination significantly decreased Phot1 expression with the progress of days (our preliminary data).
NITRIC OXIDE IN PLANT GROWTH AND DEVELOPMENT
Nitric oxide (NO) is a well-known stress signaling molecule that plays a crucialrole during plant defense against pathogens [14]. Recently, a more fundamental role in basic growth processes has been discussed. Rather surprisingly, NO has been reported to function as a downstream signaling molecule of auxin-induced lateral and adventitious root formation [32] ; [33]. Moreover, gravistimulation of roots not only induces auxin accumulation at the lower root flank, but also of NO [34] ; [35] ; [36], and a reduction in the NO level inhibits gravitropic bending of gravistimulated root apices [35].
Development of root architecture including LR formation is a critical event for successful growth of plants [1]. Auxin promotes the production of nitric oxide (NO) in roots, which is required for auxin-mediated root organogenesis [37] ; [38]. NO inhibits blue light-induced stomatal opening by regulating the K+ influx in guard cells. Inhibition of auxin transport by NPA reduced 1-GFP expression at the basal and central part of LRs, indicating role of auxin in Phot1 expression in LRs [11]. However, roles of auxin-mediated NO on Phot1 expression in LR growth and development need to be explored.
PROSPECTS AND FUTURE DIRECTIONS
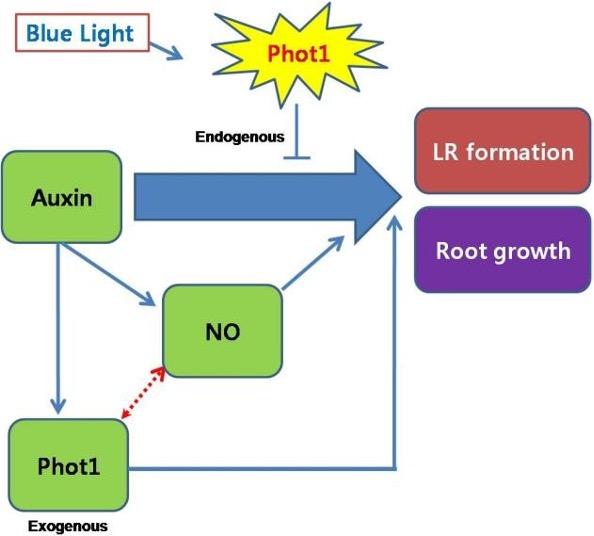
Throughout this review we highlighted the diverse mechanisms by which Phot1 has been involved in LR growth and development in plants. Blue light induces Phot1 expression which ultimately inhibits LR formation through decreasing the effects of auxin. On the other hand, exogenous auxin and NO directly helps in formation of LR. Auxin binds to Phot1 promoter region and mediates transcription of various effects on LR formation. These evidences suggest that auxin-mediated LR formation may be regulated by interact between Phot1 and NO (Figure 1). To advance our understanding on the mechanisms of LR formation through determining the role of NO to influence the regulation of Phot1 would be of matter of interest in order to develop strategy to enhance plant growth and development.
ACKNOWLEDGEMENT
This work did not need any financial support.
CONFLICTS OF INTERESTS
None of the authors declared any competing interest.
References
- [1]Woodward A.W. BB. Auxin: Regulation, Action, and Interaction. Ann Bot 2005;95:707–735. doi:doi:10.1093/aob/mci083.
- [2]Zhang H, Forde1 BG. Regulation of Arabidopsis root development by nitrate availability. J Exp Bot MP Spec Issue 2000. doi:10.1093/jexbot/51.342.51.
- [3]Beeckman T, Burssens S, Inzé D. The peri-cell-cycle in Arabidopsis. J Exp Bot 2001. doi:10.1093/jexbot/52.suppl_1.403.
- [4]Fahn A. Plant Anatomy. Plant Anat., 1982. doi:10.2134/agronj1953.00021962004500070024x.
- [5]Mullen JL, Hangarter RP. Genetic analysis of the gravitropic set-point angle in lateral roots of Arabidopsis. Adv Sp Res 2003. doi:10.1016/S0273-1177(03)00249-7.
- [6]Iino M. Chapter 23 Phototropism in higher plants. Compr Ser Photosciences 2001;1:659–811. doi:10.1016/S1568-461X(01)80027-2.
- [7]Christie JM. Phototropin Blue-Light Receptors. Annu Rev Plant Biol 2007;58:21–45. doi:10.1146/annurev.arplant.58.032806.103951.
- [8]Liscum E. Mutations in the NPH1 Locus of Arabidopsis Disrupt the Perception of Phototropic Stimuli. Plant Cell Online 1995;7:473–85. doi:10.1105/tpc.7.4.473.
- [9]Galen C, Rabenold JJ, Liscum E. Functional ecology of a blue light photoreceptor: Effects of phototropin-1 on root growth enhance drought tolerance in Arabidopsis thaliana. New Phytol 2007;173:91–9. doi:10.1111/j.1469-8137.2006.01893.x.
- [10]Wan YL, Eisinger W, Ehrhardt D, Kubitscheck U, Baluska F, Briggs W. The subcellular localization and blue-light-induced movement of phototropin 1-GFP in etiolated seedlings of Arabidopsis thaliana. Mol Plant 2008. doi:10.1093/mp/ssm011.
- [11]Moni A, Lee AY, Briggs WR, Han IS. The blue light receptor Phototropin 1 suppresses lateral root growth by controlling cell elongation. Plant Biol 2015;17:34–40. doi:10.1111/plb.12187.
- [12]Vanneste S, Friml J. Auxin: A Trigger for Change in Plant Development. Cell 2009. doi:10.1016/j.cell.2009.03.001.
- [13]Benková E, Hejátko J. Hormone interactions at the root apical meristem. Plant Mol Biol 2009;69:383–96. doi:10.1007/s11103-008-9393-6.
- [14]Fukaki H, Tasaka M. Hormone interactions during lateral root formation. Plant Mol Biol 2009;69:437–49. doi:10.1007/s11103-008-9417-2.
- [15]Galen C, Rabenold JJ, Liscum E. Light-sensing in roots. Plant Signal Behav 2007;2:106–8. doi:10.4161/psb.2.2.3638.
- [16]Péret B, De Rybel B, Casimiro I, Benková E, Swarup R, Laplaze L, et al. Arabidopsis lateral root development: an emerging story. Trends Plant Sci 2009;14:399–408. doi:10.1016/j.tplants.2009.05.002.
- [17]Celenza JL, Grisafi PL, Fink GR. A pathway for lateral root formation in Arabidopsis thaliana. Genes Dev 1995. doi:10.1101/gad.9.17.2131.
- [18]Sassi M, Wang J, Ruberti I, Vernoux T, Xu J. Shedding light on auxin movement: Light-regulation of polar auxin transport in the photocontrol of plant development. Plant Signal Behav 2013. doi:10.4161/psb.23355.
- [19]Benková E, Michniewicz M, Sauer M, Teichmann T, Seifertová D, Jürgens G, et al. Local, Efflux-Dependent Auxin Gradients as a Common Module for Plant Organ Formation. Cell 2003. doi:10.1016/S0092-8674(03)00924-3.
- [20]Casimiro I, Marchant A, Bhalerao RP, Beeckman T, Dhooge S, Swarup R, et al. Auxin Transport Promotes Arabidopsis Lateral Root Initiation. Plant Cell 2001;13:843. doi:10.2307/3871344.
- [21]De Smet I, Tetsumura T, De Rybel B, Frey NF d., Laplaze L, Casimiro I, et al. Auxin-dependent regulation of lateral root positioning in the basal meristem of Arabidopsis. Development 2007;134:681–90. doi:10.1242/dev.02753.
- [22]Bhalerao RP, Eklöf J, Ljung K, Marchant A, Bennett M, Sandberg G. Shoot-derived auxin is essential for early lateral root emergence in Arabidopsis seedlings. Plant J 2002;29:325–32. doi:10.1046/j.0960-7412.2001.01217.x.
- [23]Ljung K. Sites and Regulation of Auxin Biosynthesis in Arabidopsis Roots. PLANT CELL ONLINE 2005. doi:10.1105/tpc.104.029272.
- [24]Swarup K, Benková E, Swarup R, Casimiro I, Péret B, Yang Y, et al. The auxin influx carrier LAX3 promotes lateral root emergence. Nat Cell Biol 2008. doi:10.1038/ncb1754.
- [24]Torrey JG. The induction of lateral roots by indoleacetic acid and root decapitation. Am J Bot 1950. doi:10.2307/2437843.
- [25]Blakely LM, Durham M, Evans TA, Blakely RM. Experimental Studies on Lateral Root Formation in Radish Seedling Roots. I. General Methods, Developmental Stages, and Spontaneous Formation of Laterals. Bot Gaz 1982. doi:10.1086/337308.
- [26]Muday GK, Haworth P. Tomato root growth, gravitropism, and lateral development: correlation with auxin transport. Plant Physiol Biochem 1994;32(2): 193-203
- [27]Huala E, Oeller PW, Liscum E, Han IS, Larsen E, Briggs WR. Arabidopsis NPH1: A protein kinase with a putative redox-sensing domain. Science 1997; 80-88. doi:10.1126/science.278.5346.2120.
- [28]Esmon CA, Tinsley AG, Ljung K, Sandberg G, Hearne LB, Liscum E. A gradient of auxin and auxin-dependent transcription precedes tropic growth responses. Proc Natl Acad Sci U S A 2006. doi:10.1073/pnas.0507127103.
- [29]Gruen H. Auxin and fungi. Plant Physiol 1959;10:405–440.
- [30]Gruen H. Auxin and fungi. Plant Physiol 1959;10:405–440.
- [31]Tsavkelova EA, Klimova SY, Cherdyntseva TA, Netrusov AI. Microbial producers of plant growth stimulators and their practical use: A review. Appl Biochem Microbiol 2006;42:117–26. doi:10.1134/S0003683806020013.
- [32]Pagnussat GC, Lanteri ML, Lamattina L. Nitric oxide and cyclic GMP are messengers in the indole acetic acid-induced adventitious rooting process. Plant Physiol 2003. doi:10.1104/pp.103.022228.
- [33]Pagnussat GC, Lanteri ML, Lombardo MC, Lamattina L. Nitric oxide mediates the indole acetic acid induction activation of a mitogen-activated protein kinase cascade involved in adventitious root development. Plant Physiol 2004. doi:10.1104/pp.103.038554.
- [34]Joo JH, Bae YS, Lee JS. Role of auxin-induced reactive oxygen species in root gravitropism. Plant Physiol 2001. doi:10.1104/pp.126.3.1055.
- [35]Joo JH, Yoo HJ, Hwang I, Lee JS, Nam KH, Bae YS. Auxin-induced reactive oxygen species production requires the activation of phosphatidylinositol 3-kinase. FEBS Lett 2005. doi:10.1016/j.febslet.2005.01.018.
- [36]Hu X, Neill SJ, Tang Z, Cai W. Nitric oxide mediates gravitropic bending in soybean roots. Plant Physiol 2005. doi:10.1104/pp.104.054494.
- [37]Pagnussat GC. Nitric Oxide Is Required for Root Organogenesis. PLANT Physiol 2002. doi:10.1104/pp.004036.
- [38]Correa-Aragunde N, Graziano M, Lamattina L. Nitric oxide plays a central role in determining lateral root development in tomato. Planta 2004;218:900–5. doi:10.1007/s00425-003-1172-7.