The possible regulations through cross-generation transmission on childhood obesity
EDITORIAL
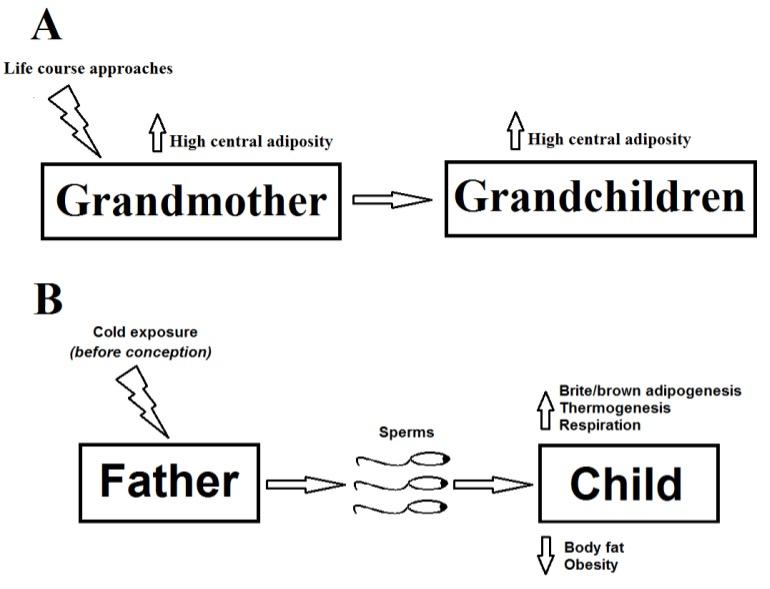
Obesity and overweight are considered as one of the top health problem worldwide, obesity may induce both physical and mental health consequences [1, 2]. Obesity and overweight is regulated by both genetics and environmental factors which can control the balance between the lipid accumulation and energy expenditure, thus several bio-functional markers [3] of adipose tissues involve in the regulation of obesity, and obesity is also regulated by developmental age and nutrition [4]. Two interesting findings in the flied of lipid metabolism and obesity have been recently released on Pediatric Obesity by R. Somerville et al [5] and on Nature Medicine by Wenfei Sun et al [6]. Both studies focused on a very important topic in childhood obesity that is “How do a life course approach and living environment of previous generations affect lipid metabolism and obesity in the child?”. The work done by R. Somerville et al [5] showed that the central adiposity in grandmother, not grandfather, was positively consistent with that in children at the age of 5 and 9. However, cold exposure (CE) in the father, not mother, before conception may benefit for systemic metabolism as well as prevent overweight and obesity in the children as proved in the study done by Wenfei Sun et al [6]. In both humans and animals, CE increases the browning of white adipocytes and the thermogenesis of both brite (the brown adipocytes induced in white fat depots) and classical brown adipocytes, these effects lead to reduction in lipid accumulation and body fat, but increase in glucose and insulin sensitivity as well as systemic metabolism [7-12]. As results, CE was proved to reduce overweight and diet-induced obesity [10, 13].
The first report was conducted in a prospective cross-generational cohort of 1094 children (5 and 9 years old), 1082 mothers, and 745 grandparents of these children [5]. As waist circumference (WC) in one of indicator for determining obesity and overweight [14, 15], authors have performed mediation analysis on WC of study cohort and found a significant positive relationship of grandmother WC and grandchildren WC, but they did not see that correlation in WC between grandfather and grandchildren. This result indicates that cross-generation transmission maybe one of factors regulating childhood obesity (Fig. 1A).
The second report shows that environmental effects on father lead to changes in the systemic metabolism of offspring [6]. This has opened potential ways to control lipid metabolism and prevent obesity in human by optimizing ambient temperature at parents’ living places. It has been known that cold exposure activates the classical brown adipose tissue and induces functionally brite adipocytes in white fat depots in both human and animal models. The function of these thermogenic (brown and brite) adipocytes can improve the systemic metabolism and reduce fat mass [16-18, 12, 19], thus the cold exposure is considered as a potential therapy for controlling obesity and overweight [16, 18, 12]. In the current report [6], Wenfei Sun et al not only proved the anti-obesity effect of cold exposure but also went to a further step, because they showed that the browning effect of CE could transfer through from generations. Analyzing a cohort of 8,440 subjects, researchers found higher activities of thermogenic adipocytes in both brown (BAT) and white (WAT) fat tissues in children from the parents who were exposed to cold before impregnation or during pregnancy. Further investigations by authors showed that the CE effects on brown/brite adipocytes were only regulated through the paternal lineage. These findings in humans were supported by the studies in mice, they found that higher thermogenesis and respiration were partially induced by an increase in BAT activation of pups from the fathers exposed to the cold, and the paternal CE (P-CE) could improve systemic metabolism and protected mouse offspring from diet induced obesity. Mechanically, investigators proved that the increase in BAT function of offspring maybe due to the changes in brown adipogenesis and neurogenesis induced by P-CE, and the elevated formation of thermogenic cells in offspring stimulated by P-CE was a cell autonomous manner. Thus, the important findings by Wenfei Sun et al [6] suggest that CE in the father before conception may benefit for systemic metabolism as well as prevent overweight and obesity in the child (Fig. 1B).
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare.
References
- [1]Chu DT, Minh Nguyet NT, Dinh TC, Thai Lien NV, Nguyen KH, Nhu Ngoc VT et al. An update on physical health and economic consequences of overweight and obesity. Diabetes Metab Syndr. 2018; 12(6): 1095-1100.
- [2]Chu DT, Minh Nguyet NT, Vu Thi N, Nguyen Vu TL, Duc Duy V, Nguyen L et al. An update on obesity: Mental consequences and psychological interventions. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2019; 13:155-160.
- [3]Chu DT, Malinowska E, Gawronska-Kozak B, Kozak LP. Expression of Adipocyte Biomarkers in a Primary Cell Culture Models Reflects Preweaning Adipobiology. Journal of Biological Chemistry. 2014; 289:18478-18488.
- [4]Chu DT, Malinowska E, Jura M, Kozak LP. C57BL/6J mice as a polygenic developmental model of diet-induced obesity. Physiological Reports. 2017; 5:20.
- [5]Somerville R, Khalil H, Segurado R, Mehegan J, Viljoen K, Heinen M, et al. Childhood central adiposity at ages 5 and 9 shows consistent relationship with that of the maternal grandmother but not other grandparents. Pediatric Obesity. 2018; 13(12):778-785.
- [6]Sun W, Dong H, Becker AS, Dapito DH, Modica S, Grand G, et al. Cold-induced epigenetic programming of the sperm enhances brown adipose tissue activity in the offspring. Nat Med. 2018; 24(9):1372-1383.
- [7]Saito M, Okamatsu-Ogura Y, Matsushita M, Watanabe K, Yoneshiro T, Nio-Kobayashi J, et al. High Incidence of Metabolically Active Brown Adipose Tissue in Healthy Adult Humans: Effects of Cold Exposure and Adiposity. Diabetes. 2009; 58:1526-1531.
- [8]van Marken Lichtenbelt WD, Vanhommerig JW, Smulders NM, Drossaerts JM, Kemerink GJ, Bouvy ND, et al. Cold-Activated Brown Adipose Tissue in Healthy Men. New England Journal of Medicine. 2009; 360:1500-1508.
- [9]Ouellet V, Labbé SM, Blondin DP, Phoenix S, Guérin B, Haman F, et al. Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J Clin Invest. 2012; 122(2):545-52.
- [10]Boon MR, van den Berg SA, Wang Y, van den Bossche J, Karkampouna S, Bauwens M, et al. BMP7 Activates Brown Adipose Tissue and Reduces Diet-Induced Obesity Only at Subthermoneutrality. PLoS One. 2013; 8(9):e74083.
- [11]Nagano G, Ohno H, Oki K, Kobuke K, Shiwa T, Yoneda M, et al. Activation of Classical Brown Adipocytes in the Adult Human Perirenal Depot Is Highly Correlated with PRDM16–EHMT1 Complex Expression. PLoS ONE. 2015; 10:e0122584.
- [12]Chu DT and Tao Y. Human thermogenic adipocytes: a reflection on types of adipocyte, developmental origin, and potential application. Journal of Physiology and Biochemistry. 2017; 73:1-4.
- [13]Ravussin Y, Xiao C, Gavrilova O, Reitman ML. Effect of Intermittent Cold Exposure on Brown Fat Activation, Obesity, and Energy Homeostasis in Mice. PLoS ONE. 2014; 9:e85876.
- [14]Janssen I, Heymsfield SB, Allison DB, Kotler DP, Ross R. Body mass index and waist circumference independently contribute to the prediction of nonabdominal, abdominal subcutaneous, and visceral fat. The American Journal of Clinical Nutrition. 2002; 75:683-688.
- [15]Hanh NTH, Tuyet LT, Dao DTA, Tao Y, Chu DT. Childhood Obesity Is a High-risk Factor for Hypertriglyceridemia: A Case-control Study in Vietnam. Osong Public Health and Research Perspectives. 2017; 8:138-146.
- [16]Chu DT, Tao Y, Son LH, Le DH. Cell source, differentiation, functional stimulation, and potential application of human thermogenic adipocytes in vitro. Journal of Physiology and Biochemistry. 2016; 73:315-321.
- [17]Chu DT and Gawronska-Kozak B. Brown and brite adipocytes: Same function, but different origin and response. Biochimie . 2017; 138:102-105.
- [18]Chu DT and Tao Y. A homologous stem cell therapy for obesity and its related metabolic disorders. Medical Hypotheses. 2017; 103:26-28.
- [19]Chu DT, Tao Y, Taskén K. OPA1 in Lipid Metabolism: Function of OPA1 in Lipolysis and Thermogenesis of Adipocytes Hormone and Metabolic Research. 2017; 49:276-285.