Towards the antimicrobial, therapeutic and invasive properties of Mikania micrantha Knuth: a brief overview
Abstract
Plant-derivatives impose a huge momentum in the field of medical science lately due to their wide-spectrum therapeutic attributes. Owing to the emerging drug resistance and hazardous side-effect of synthetic drugs, phytochemicals are now coming into play as a source of new and effective therapeutics. Mikania micrantha is a medicinal plant commonly found in tropical Asian countries including Bangladesh. The pharmacological significances of this plant were reported earlier which include a diverse range of antimicrobial and therapeutic potencies. However, the rapid-growing nature and covering surrounding flora reckoned M. micrantha as one of the world’s most invasive weeds. Therefore, it is essential to understand if the therapeutic essence of M. micrantha outweighs its invasiveness. In this brief review, we tried to explore the biological activities of M. micrantha. The future perspectives regarding the management of its invasiveness were also highlighted in this limited scope.
INTRODUCTION
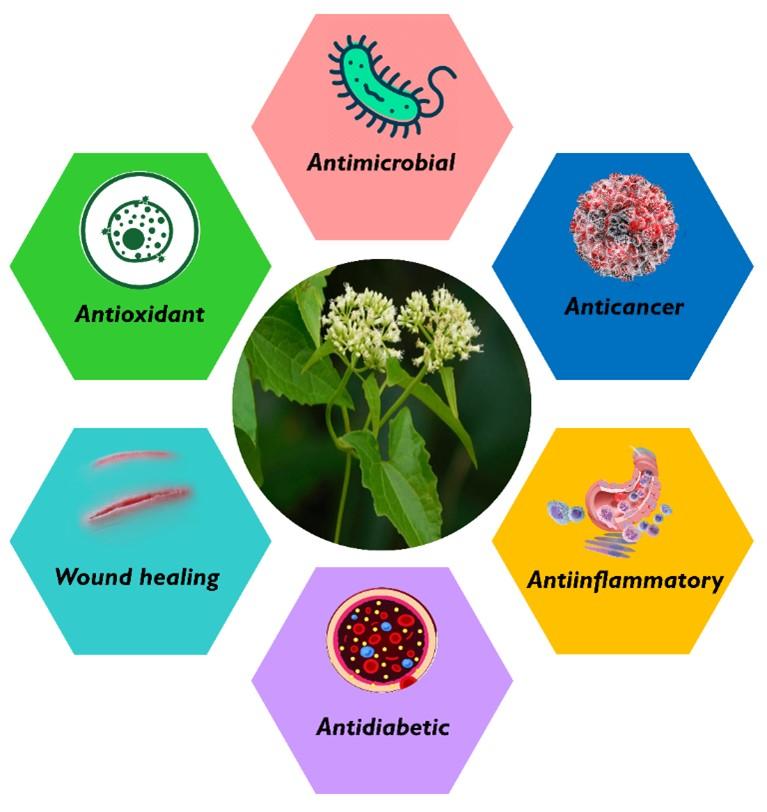
Mikania micrantha Knuth is a fast-growing tropical herb, also known as mile-a-minute or bitter vine, which belongs to the Asteraceae family [1]. It is native to the tropical zones of Central and South America but is now widely distributed in Southeast Asia, Pacific Islands, South China, etc. [1,2]. Traditionally, it has been used as folk medicine in many areas around the world. For example, a poultice made from the leaves of M. micrantha is used to treat venomous biting of insects [3,4] and the leaf juice is used to reduce skin rashes and itches [5]. In Jamaica, its most popular uses are for wound dressings and promote the healing of sores as folk medicine [6]. Furthermore, it is used to mitigate stomach ache, jaundice, fever, rheumatism, cold, and respiratory diseases [4]. Modern pharmacological studies provide scientific evidence that bitter vine possesses outstanding therapeutic potencies, i.e., antimicrobial, anti-inflammatory, cytotoxic, anticancer, antidiabetic, antioxidant, and wound healing activities [4,7]. Therefore, M. micrantha has gained the attention of natural product chemists because of its numerous biological potencies. Due to its fast-growing nature and invading surrounding flora, however, M. micrantha has been recorded as one of the 100 worst invasive alien species in the world [8], and the second most serious weed in South Pacific regions [9].
Natural products have played an important role in the discovery of drugs and therapeutics. Phytochemicals are small molecules with diverse chemical profiles and more “drug-like” than synthetic compounds, hence, they are considered as good candidates for the development of drug leads [10]. In recent decades, the genus Mikania under the Asteraceae family has been extensively studied due to their diverse chemical compositions [11,12]. For instance, M. micrantha has been reported to contain several classes of bioactive chemical substances, i.e., terpenoids (sesquiterpene lactones and diterpenes), polyphenols and flavonoids [12]. Likewise, the presence of various terpene-derivatives, especially mikanolide and miscandenin, is responsible for the antibacterial and analgesic activities of the plant [13,14]. The recent emergence of multidrug-resistance in pathogenic bacteria poses a new threat to our current therapeutic advances, thereby, pressing urges to find new antimicrobial agents [15].
An adequate amount of research has been done on the biological properties of M. micrantha while a few review works were conducted either on its medicinal or invasive activities. To the best of our knowledge, however, no article encompasses both sides of this plant with prospective control management. In this review, therefore, we aimed to explore and summarize both therapeutic and invasive attributes of M. micrantha plant. Figure 1 shows the major biological attributes reported previously. The biological activities and their mechanism of actions were reviewed based on the existing pieces of literature.
ANTIMICROBIAL ACTIVITIES
Antibacterial properties
Antibiotics have been using since 1940s to treat bacterial infections [16]. Due to the selective pressure involved, the bacteria are prone to develop resistance against antibiotics which is now considered as a major global health concern [15]. Most of the common bacterial pathogens have now become resistant to almost all classes of antibiotics in empirical use [17]. In addition, the discovery of new antibiotics is getting rare. For instance, only one antibiotic named daptomycin was discovered in the last 50 years [18]. Therefore, there is a pressing need to find an alternative therapeutics. Plant-derived phytochemicals could be useful in such scenarios because of their antimicrobial potencies, non-toxic nature, and bioavailability [5]. Plants belong to Mikania genus have been reported to possess antibacterial activity against a large number of bacteria [12].
In a study, methanolic extract (at a concentration of 200 mg/ml) of the M. micrantha was able to potentially inhibit the growth of six bacterial strains where antibacterial activity against Bacillus cereus was equivalent to the antibiotic ciprofloxacin [19]. Also, leaf and flower extracts showed moderate inhibitory activity against the growth of B. cereus, Escherichia coli, Shigella sonnii and Streptococcus pyogenes [13,14,20]. Furthermore, various extracts of M. micrantha containing tannins, flavonoids and polyphenols exhibited potential antibacterial activities against multidrug-resistant pathogenic bacteria i.e., Pseudomonas aeruginosa, Salmonella typhii, Staphylococcus aureus, Staphylococcus epidermidis, Bacillus subtilis, E. coli, and Streptococcus pneumoniae [13,21,22]. This is plausible because polyphenols have toxicity towards microbial enzyme while structural features of flavonoids may help to gain entry into the bacterial cell which eventually leads to multiple component inactivation [23-24]. Likewise, sesquiterpene lactones (SLs) from different plants provides antimicrobial activity against a wide range of microorganisms including bacteria, virus, and fungi [25]. For instance, M. micrantha derived sesquiterpene lactones showed significant inhibitory activity against S. aureus, B. subtilis, Micrococcus luteus, B. cereus, Ralstonia solanacearum, Xanthomonas oryzae pv. oryzae, Xanthomonas campestris pv. vesicatoria, and X. campestris pv. citri [5]. Therefore, M. micrantha represents a potential source of novel antibacterial phytochemicals that needs further investigation in vivo. A summary of the antibacterial activity by different parts of M. micrantha is provided in Table 1.
Antifungal attributes
Approximately a billion people around the world have skin, nail, and hair infections caused by pathogenic fungi. Although most of them are curable, the mortality rate of fungal diseases is similar to tuberculosis [26]. In addition, plant pathogenic fungi living in or on plant tissues have an enormous impact on agriculture that imposes a major challenge to overcome [27]. In response to fungal attack, the plant synthesizes various metabolites as a part of their defense mechanism [28]. Table 1 includes a list of antifungal activities by different parts of M. micrantha. The leaf extracts of M. micrantha have an inhibitory effect against the germination of the spore by several fungal species such as Exserohilum turcicum, Colletotrichum lagenarium, Pseudoperonospora cubensis, and Botrytis cinerea [5]. A study revealed that the presence of glycosides and quinones is responsible for the antifungal activity against the Fusarium moniliforme, Fusarium eridiforme, Fusarium proliferatum, and Sclerotium rolfsii [29]. Quinone targets the cell-wall polypeptides, membrane-bound enzymes leading to the inactivation of protein synthesis and cellular function [30]. Furthermore, ethyl acetate extracts of M. micrantha leaves, flowers and stems were able to completely inhibit the growth of several phytopathogenic fungi, i.e., Epidermophyton floccosum var. nigricans, Microsporum gypseum, Microsporum canis, and Trichophyton rubrum while petroleum ether extract provided significant inhibition [2].
Antiparasitic activities
More than a quarter of the world population is infected with soil-borne helminths like hookworm and Ascaris species – several million people are affected by intestinal protozoal diseases, i.e., amebiasis and giardiasis [31,32]. In addition, parasites often cause the death of wildlife as a consequence of the altered behavior of their hosts [33]. For centuries, natural derivatives have been used for the treatment of parasitic diseases. Both in vivo and in vitro experiments suggest that the plant-derived lactones, alkaloids, and tannins have antiparasitic activity [34]. For example, sesquiterpene lactones of M. micrantha have been reported to exhibit significant antiprotozoal activity against Trypanosoma cruzi and Leishmania braziliensis [35]. Laura et al., 2017 also reported that α and β-unsaturated lactone groups in the sesquiterpene lactone are the major determinants of antiprotozoal activity. Likewise, alkaloids, flavonoids, saponins, phenolic compounds present in the methanolic extract of M. micrantha have a mild antihelmintic activity that causes paralysis and death of adult earthworm Pheretima posthuma in a dose-dependent manner [36]. Some notable antiparasitic activities of M. micrantha are provided in Table 1.
Table 1. Previously reported biological attributes of the different part of Mikania micrantha plant.
THERAPEUTIC ATTRIBUTES
Anti-inflammatory attributes
There are many side effects associated with the administration of non-steroidal anti-inflammatory drugs (NSAIDs) including the risk of gastrointestinal (GI) and cardiovascular complications [37]. Medicinal plants that’s possess anti-inflammatory potency with little or no side effects have been using by Africans for centuries [38]. For example, ethanolic extracts of M. micrantha leaves were found to have anti-inflammatory activity in a dose-dependent manner. Deori et al., 2017 reported that at a certain dose it can reach up to the effect provided by aspirin at the dose level of 100 mg/kg. Moreover, the study speculated a significant weight reduction in rat adjuvant arthritis at a dose of 200 mg/kg and 400 mg/kg [39]. In another study, ethanolic extracts of M. micrantha showed higher anti-inflammatory activity than M. scandens but lower than that of aspirin which was estimated based on the ability to inhibit hypotonic solution and heat-induced hemolysis of human erythrocytes in vitro [14]. Furthermore, micrantholides from M. micrantha has exhibited better anti-inflammatory effect in tetradecanoylphorbol acetate (TPA) induced mouse ear edema (Table 1) [3]. Moreover, hexane and ethyl acetate extracts of M. micrantha stems and leaves also provided a significant reduction of inflammation in vivo (Table 1) [21]. Therefore, further investigations are necessary for the identification of relevant bioactive chemicals alternative to NSAIDs.
Cytotoxic and anticancer potencies
Cancer is the second major global disease accounting for ⁓13% of global death each year [40]. Today’s cancer treatments are mostly chemotherapeutics and have plenty of side effects. Therefore, natural derivatives with less toxicity could be a better replacement for those synthetic drugs. Many plants in the Asteraceae family are found to have cytotoxic effects against cancer cells [41]. Likewise, various extracts and phytochemicals from M. micrantha are reported to have cytotoxic and anticancer potentials (Table 1). For instance, M. micrantha derived flavonoids have shown a dose-dependent anticancer effect against Ehrlich ascites carcinoma cells in Swiss albino mice [42]. Also, an aqueous extract obtained from M. micrantha showed in vitro anticancer activity against human cancer cell lines, K562 and HeLa [43]. Dou et al., 2013 also reported in vivo growth inhibition of murine Sarcoma 180 cells (S180). In their study, the growth inhibition of S180 occurred via antiproliferation, apoptosis, and cell cycle arrest. Lack of cellular apoptosis causes malignancy, hence, restoring the cellular regulation on programmed cell death could reduce cancer growth. In addition, plenty of studies suggested that using apoptosis in cancer treatments is feasible [44]. Among other phytochemicals, sesquiterpene lactones (SLs) are known to exhibit major antiproliferative effects [25]. Nine SLs isolated from the aerial parts of M. micrantha provided cytotoxic activities on three cancer cell lines due to their specific structural features [3]. These structural features may cause specific damage to the oncogenic marker included in the tumor cells. These studies concluded that M. micrantha may contain phytochemicals with anticancer potency. However, further investigation of their mechanism of action is warranted to extrapolate their potentiality in clinical practice.
Antidiabetic characteristics
According to the International Diabetes Federation (IDF), diabetes affects about 285 million people across the world [45]. Most commonly used oral and injectable antidiabetic agents have different side effects like nausea, vomiting, diarrhea, cardiovascular complications, etc. [46]. Therefore, natural derivatives with fewer side effects and efficacy can be used as an alternative antidiabetic agent. Many plants under the Asteraceae family have been reported to provide antidiabetic activity [47]. For example, extracts of M. micrantha is reported to have a hypoglycemic effect (Table 1) [48]. The blood glucose level of alloxan-induced diabetic rats was significantly reduced (p<0.05) when treated with ethanolic extract of M. micrantha, probably due to the stimulation of insulin-producing β-cells of the pancreas [49]. Furthermore, Nurdiana et al., 2013 reported a 72% reduction in blood glucose level which is higher than metformin, a first-line type-2 diabetes medicine [48]. They suggested insulin-mimetic activity could be associated with this process.
Antioxidant properties
Aerobic metabolic processes within the cell produce reactive oxygen species (ROS) which leads to cell damage and even cell death [50]. Antioxidants reduce the oxidative stress in cells by quenching those free radicals [51]. A large number of plant-derived compounds have been reported to possess free radical scavenging activity [52]. The methanolic extract of M. micrantha leaves showed significant antioxidant activity in ferric-reducing antioxidant potential (FRAP) assay (Table 1). In FRAP assay, the antioxidant phytochemical reduces iron from ferric (Fe3+) to ferrous (Fe2+) state [53]. In addition, phenolic compounds from M. micrantha exhibit more potent 2,2-diphenyl-1-picrylhydrazyl (2,2-DPPH) radical scavenging activity than L-ascorbic acid (Table 1) [54]. These phenolics act as chain-breaking antioxidants by shifting its hydrogen (H) atom as proton from hydroxyl (OH) group to the chain carrying ROO• radicals [55]. Furthermore, methanolic extract of M. micrantha showed prominent antioxidant activity on 1,1-diphenyl-2-picrylhydrazyl (1,1-DPPH) which may be due to the presence of the phenolic compounds in the extract [36].
Wound healing aspects
Medicinal plants are reported to promote wound healing by boosting blood clotting and fighting infection with lower side effects [56]. Traditionally, herbal medicine used in India and China for wound dressing and healing of sores by our ancestors because of its fascinating healing power. The ethanolic extract of M. micrantha showed a significant acceleration in wound healing on fibroblast cells even in a lower dose (Table 1) [57]. Histological studies suggest that the ethanolic extract of M. micrantha improves the healing process on diabetic wounds in rats by increasing granulation tissue and collagen deposition [48]. For wound healing, collagen protein acts as a scaffold in connective tissue and deposition of collagen results in increased tensile strength of the wound site [58]. Moreover, an ointment made from M. micrantha showed a moderate rate of wound healing in male Wistar albino rats probably by enhancing collagen concentration [59].
INVASIVE CHARACTERISTICS
Biological invasion has become one of the most critical environmental problems in the 21st century which leads to world-wide extensive economic losses [60]. M. micrantha has been listed among the top 100 worst invasive alien species [8]. Rhizosphere soil of M. micrantha causes a significant decrease (⁓21%) of shoot length of Panicum antidotale. Further, leaf leachate of genus Mikania contributes to the decreased rice seed germination in non-sterile soil [61]. In a study, M. micrantha is negatively correlated with Ageratum conyzoides, Bidens pilosa, Borreria latifolia, Digitaria sanguinalis, and Galinsoga parviflora [62]. Moreover, the leaf extract of M. micrantha inhibited the germination of Raphanus sativus and Oryza sativa due to the presence of allelochemicals in the extract [63].
In Kolkata, the state capital of West Bengal, a total of 86 sites were surveyed to identify the presence of this exotic alien plant. The survey revealed that 13 sites were highly infested with M. micrantha while moderate infestation was found at 39 sites [64]. The rapid spreading of M. micrantha is responsible for yield loss by several cash crops, i.e., sugarcane, lemon, orange, and banana [65]. Due to its involvement with tea, rubber, and other crop infestation, Mikania has been declared a class-I weed in Queensland (Australia) under the Rural Land Protection Act. Moreover, it has been ranked amongst the top three worst weed plant by the Secretariat of the Pacific Community [66].
BIOLOGICAL CONTROL
Despite the potential applicability, the invasiveness of M. micrantha should be controlled as a part of crop management as well as to protect the biological diversity. There are two major weed control strategies – chemical and mechanical approaches [67]. For example, chemical herbicides including atrazine, bensulfuron-methyl, and prometryne have significant bioactivity and selectivity to the germination and seedling of M. micrantha plant [65]. Furthermore, herbicides such as 2,4-dichlorophenoxyacetic acid, sulfometuron-methyl, and glyphosate are widely used in farming systems and forest systems to control M. micrantha infestation [68]. On the other hand, the mechanical method is also used to control various weeds including M. micrantha. For instance, a study suggests a manual cutting strategy before flowering in Nepal which results in a 91% mortality of M. micrantha [69]. However, mechanical methods cause soil disturbance which may consequently lead to erosion. Likewise, chemical-based herbicides can compromise the soil fertility and environment that pose serious threats to humans and wildlife. Moreover, certain weeds are prone to develop resistance against these chemicals [68].
Therefore, biological control could be more beneficial over the conventional methods due to several reasons – permanent control of weeds, host-specific control, cost-effectiveness and low health risk [70,71]. The term ‘biological control’ refers to the control of weeds by introducing natural antagonistic pathogens. Interestingly, several arthropods are known to interfere with the growth of M. micrantha plant. For example, Acalitus sp. causes shortened internodes with reduced flowering and Liothrips mikaniae causes small to moderate lesions on M. micrantha leaves [72]. Furthermore, co-cultivation of sweet potato (Ipomoea batatas) and M. micrantha results in the reduction of shoot length and increased rate of M. micrantha inhibition [62]. The rust fungus Puccinia spegazzinii reduced M. micrantha population by ⁓50% through selective growth suppression without affecting the other plants [73]. These antagonistic pathogens can be used to control the overgrowth of M. micrantha. In Papua New Guinea, for instance, P. spegazzinii was released in 15 provinces as a specific control strategy against M. micrantha infestation to compensate for the economic loss due to reduced yields and high weeding cost [74]. However, more understanding of M. micrantha genetics is needed for fine-tuning the population-dependent effective management approaches [68]. Thus, biological control could play a large role in mitigation and adaptation strategies used to maintain biological diversity as well as human well-being by protecting food and fiber resources.
CONCLUSION
In a nutshell, Mikania micrantha is the reservoir of numerous compounds with pharmacological value in spite of the invasiveness. Based on the previous studies, these bioactive chemicals has been found to hold promising therapeutic value regarding their use in various pathological conditions including pathogen inflicted diseases, malignancies, diabetes, tissue inflammation, and severe wounds. However, further investigations are warranted to shed light on the molecular mechanisms behind their biological actions that are essential in functional drug development. Importantly, considering the rapid and ubiquitous growth of bitter vine, it could also be used in the development of safe and cost-effective medical treatments for developing countries like Bangladesh. For this purpose, the extensive loss due to its invasive nature should be minimized through eco-friendly biological control and novel genetic engineering approaches.
AUTHOR CONTRIBUTIONS
ZN conceived the idea; ZN, MMS, and ZH wrote the draft manuscript; ZN critically revised and finalized the manuscript; ZN supervised the whole work. All authors approved the final version of the manuscript.
CONFLICTS OF INTEREST
The authors declare no conflict of interest.
References
- [1]Day MD, Clements DR, Gile C, et al. Biology and Impacts of Pacific Islands Invasive Species. 13. Mikania micrantha Kunth (Asteraceae). Pacific Sci 2016; 70(3): 257-85. doi:10.2984/70.3.1
- [2]Jyothilakshmi M, Jyothis M, Latha MS. Antidermatophytic Activity of Mikania micrantha Kunth: An Invasive Weed. Pharmacognosy Res 2015; 7(Suppl 1):S20-5. doi:10.4103/0974-8490.157994
- [3]Ríos VE, León A, Chávez MI, Torres Y, Ramírez-Apan MT, Toscano RA et al. Sesquiterpene lactones from Mikania micrantha and Mikania cordifolia and their cytotoxic and anti-inflammatory evaluation. Fitoterapia 2014; 94:155-63. doi:10.1016/j.fitote.2014.02.006
- [4]Ishak AH, Shafie NH, Norhaizan ME, Bahari H. Nutritional, Phytochemical and Pharmacological Properties of Mikania micrantha Pertanika J Sch Res Rev 2016; 2(3):123-32.
- [5]Li Y, Li J, Li Y, Wang X, Cao A. Antimicrobial Constituents of the Leaves of Mikania micrantha B. K. PLoS One 2013; 8(10):e76725. doi:10.1371/journal.pone.0076725
- [6]Bakir M, Facey PC, Hassan I, Mulder WH, Porter RB. Mikanolide from Jamaican Mikania micrantha. Acta Cryst 2004; C60(11): 798-800. doi:10.1107/S0108270104017809
- [7]Juliani HR. Physical and Chemical properties, Composition, and Biological Activity of Essential Oils of Philippine Medicinal Plants Recommended Citation. J Med Act Plants 2017; 5(2):28-35. doi:10.7275/R5P84933
- [8]Clout M, De Poorter M. International Initiatives Against Invasive Alien Species. Weed Technol 2005; 19(3): 523-7. doi:10.1614/WT-04-126.1
- [9]Khadka A. Assessment of the perceived effects and management challenges of Mikania micrantha invasion in Chitwan National Park buffer zone community forest, Nepal. Heliyon 2017; 3(4):e00289. doi:10.1016/j.heliyon.2017.e00289
- [10]Laurella LC, Frank FM, Sarquiz A, Alonso MR, Giberti G, Cavallaro L et al. In vitro evaluation of antiprotozoal and antiviral activities of extracts from Argentinean Mikania species. Sci World J 2012; 2012:121253. doi: 10.1100/2012/121253
- [11]Reidel RVB, Nardoni S, Mancianti F, Anedda C, El Gendy AEG, Omer EA et al. Chemical composition and antifungal activity of essential oils from four Asteraceae plants grown in Egypt. Zeitschrift für Naturforsch C 2018; 73(7-8):313-8. doi:10.1515/znc-2017-0219
- [12]Rufatto LC, Gower A, Schwambach J, Moura S. Genus Mikania: chemical composition and phytotherapeutical activity. Rev Bras Farmacogn 2012; 22(6):1384-403. doi:10.1590/S0102-695X2012005000099
- [13]Matawali A, Ping L, Siew H, Gansau J. Antibacterial and Phytochemical Investigations of Mikania micranthaB.K. (Asteraceae) From Sabah, Malaysia. Trans Sci Technol 2016; 3(2):244-50.
- [14]Khatun R, Nasrin L, Roy S, Tantry MA, Abdur Rahman MA. Comparative antimicrobial evaluation of available Mikania species in Bangladesh. Int J Plant Res 2017; 7(2):36-8.
- [15]Nain Z, Islam MA, Minnatul Karim M. Antibiotic Resistance Profiling and Molecular Phylogeny of Biofilm Forming Bacteria From Clinical and Non-clinical Environment in Southern Part of Bangladesh. Int J Enteric Pathog 2019; 7(2):37-43. doi:10.15171/ijep.2019.10
- [16]Ventola CL. The antibiotic resistance crisis: part 1: causes and threats. P&T 2015; 40(4):277-83. PMID: 25859123.
- [17]Cole ST. Who will develop new antibacterial agents? Phil Trans R Soc B 2014; 369(1645):20130430. doi:10.1098/rstb.2013.0430
- [18]Lewis K. Antibiotics: Recover the lost art of drug discovery. Nature 2012; 485:439-40. doi:10.1038/485439a
- [19]Borkataky M, Bhushan Kakoty B, Saikia L. Antimicrobial activity and phytochemical screening of some common weeds of asteraceae family. Int J Pharm Sci Rev Res 2013; 23(1):116-20.
- [20]Jyothilakshmi M, Jyothis M, Sankunni Latha M. Mikania micrantha – a Natural Remedy to Skin Infections. Int J Curr Microbiol Appl Sci 2016; 5(2):742-5. doi:10.20546/ijcmas.2016.502.083
- [21]Pérez-Amador MC, Muñoz Ocotero V, Ibarra Balcazar R, García Jiménez F. Phytochemical and pharmacological studies on Mikania micranthaB.K. (Asteraceae). Phyton 2010; 79:77-80.
- [22]Harahap NI, Nainggolan M, Harahap U. Formulation And Evaluation Of Herbal Antibacterial Gel Containing Ethanolic Extract Of Mikania micrantha Kunth Leaves. Asian J Pharm Clin Res 2018; 11(3):429. doi:10.22159/ajpcr.2018.v11i3.22211
- [23]Cowan MM. Plant Products as Antimicrobial agents. Clin Microbiol Rev 1999; 12(4):564-82. doi: 10.1128/CMR.12.4.564
- [24]Cushnie TPT, Lamb AJ. Antimicrobial activity of flavonoids. Int J Antimicrob Agents 2005; 26(5):343-56. doi:10.1016/j.ijantimicag.2005.09.002
- [25]Matejić J, Šarac Z, Ranđelović V. Pharmacological Activity of Sesquiterpene Lactones. Biotechnol Biotechnol Equip 2010; 24(sup1):95-100. doi:10.1080/13102818.2010.10817819
- [26]Bongomin F, Gago S, Oladele R, Denning D. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J Fungi 2017; 3(4):E57. doi:10.3390/jof3040057
- [27]Doehlemann G, Ökmen B, Zhu W, Sharon A. Plant Pathogenic Fungi. Microbiol. Spectr 2016; 5(1):1-23. doi:10.1128/microbiolspec.FUNK-0023-2016
- [28]Chitarra GS. Germination inhibitors of fungal spores: identification and mode of action. PhD Thesis 2003. Web: https://edepot.wur.nl/121471
- [29]Baral B, Bhattarai N, Vaidya GS. Pharmacological and Antagonistic Potentials of Mikania micrantha. Nepal J Sci Technol 2012; 12:75-84. doi:10.3126/njst.v12i0.6483
- [30]Omojate GC, Enwa FO, Jewo AO, Eze CO. Mechanisms of Antimicrobial Actions of Phytochemicals against Enteric Pathogens – A Review. J Pharm Chem Biol Sci 2014; 2(2):77-85.
- [31]Omojate GC, Enwa FO, Jewo AO, Eze CO. Mechanisms of Antimicrobial Actions of Phytochemicals against Enteric Pathogens – A Review. J Pharm Chem Biol Sci 2014; 2(2):77-85.
- [32]Chacon-Cruz E. Intestinal Protozoal Diseases. eMedicine J 2003; 999282. Web: https://emedicine.medscape.com/article/999282
- [33]Borgsteede FHM. The effect of parasites on wildlife. Vet Q 1996; 18(sup3):138-40. doi:10.1080/01652176.1996.9694717
- [34]Athanasiadou S, Githiori J, Kyriazakis I. Medicinal plants for helminth parasite control: facts and fiction. Animal 2007; 1(9):1392-400. doi:10.1017/S1751731107000730
- [35]Laurella LC, Cerny N, Bivona AE, et al. Assessment of sesquiterpene lactones isolated from Mikania plants species for their potential efficacy against Trypanosoma cruzi and Leishmania sp. PLoS Negl Trop Dis 2017; 11(9):e0005929. doi:10.1371/journal.pntd.0005929
- [36]Dev UK, Hossain MT, Islam MZ. Phytochemical Investigation, Antioxidant Activity and Anthelmintic Activity of Mikania micrantha World J Pharm Res 2015; 4(5):121-30.
- [37]Sostres C, Gargallo CJ, Arroyo MT, Lanas A. Adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs, aspirin and coxibs) on upper gastrointestinal tract. Best Pract Res Clin Gastroenterol 2010; 24(2):121-32. doi:10.1016/j.bpg.2009.11.005
- [38]Oguntibeju O. Medicinal plants with anti-inflammatory activities from selected countries and regions of Africa. J Inflamm Res 2018; 11:307-17. doi:10.2147/JIR.S167789
- [39]Deori C, Dutta G, Das S, Phukan D, Gogoi G. To Evaluate The Anti-Inflammatory Activity Of Ethanolic Extract Of Leaves Of Mikania micrantha On Experimental Animal Models. J Evol Med Dent Sci 2017; 6(50):3818-21. doi:10.14260/Jemds/2017/825
- [40]Aliomrani M, Jafarian A, Zolfaghari B. Phytochemical Screening and Cytotoxic Evaluation of Euphorbia turcomanica on Hela and HT-29 Tumor Cell Lines. Adv Biomed Res 2017; 6(1):68. doi:10.4103/2277-9175.192734
- [41]Uzun K, İkiz P, Daşkın R, Gürbüz P, Yalçın FN. Cytotoxic Potentials of Some Asteraceae Plants from Turkey on HeLa Cell Line. Proceedings 2017; 1(10):1068. doi:10.3390/proceedings1101068
- [42]Debaprotim D, Suvakanta D, Jashabir C. Evaluation of Anticancer Activity of Mikania micrantha Kunth (Asteraceae) Against Ehrlich Ascites Carcinoma in Swiss Albino Mice. Int J Pharm Res Allied Sci 2014; 3(2):9-18.
- [43]Dou X, Zhang Y, Sun N, Wu Y, Li L. The anti-tumor activity of Mikania micrantha aqueous extract in vitro and in vivo. Cytotechnology 2014; 66(1):107-17. doi:10.1007/s10616-013-9543-9
- [44]Wong RSY. Apoptosis in cancer: From pathogenesis to treatment. J Exp Clin Cancer Res 2011; 30(1): 87. doi:10.1186/1756-9966-30-87
- [45]Hu FB. Globalization of Diabetes: The role of diet, lifestyle, and genes. Diabetes Care 2011; 34(6):1249-57. doi:10.2337/dc11-0442
- [46]Marín-Peñalver JJ, Martín-Timón I, Sevillano-Collantes C, Cañizo-Gómez FJ del. Update on the treatment of type 2 diabetes mellitus. World J Diabetes 2016; 7(17):354-95. doi:10.4239/wjd.v7.i17.354
- [47]Sidhu MC, Sharma T. A Database of Antidiabetic Plant Species of Family Asteraceae, Euphorbiaceae, Fabaceae, Lamiaceae and Moraceae. Int J Herb Med 2013; 1(2):187-99.
- [48]Samsulrizal N, Suhaimi NA, Sopian NFA, Hatta SKM, Jamil NM. Hypoglycaemic, Antioxidant and Wound Healing Activities of Mikania micrantha Leaves Extract in Normal and Alloxan-Induced Diabetic Rats. Sci Lett 2013; 7(2):6-10.
- [49]Nurhayati WHW, Arlizan NT, Nurdiana S. Effect of Mikania micrantha leaf extract on the level of blood glucose and hepatic glycogen in the normal and alloxan-induced diabetic rats. Nat Prod 2013; 9(10):398-402.
- [50]Sharma P, Jha AB, Dubey RS, Pessarakli M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J Bot 2012; 2012:1-26. doi:10.1155/2012/217037
- [51]Krishnaiah D, Sarbatly R, Nithyanandam R. A review of the antioxidant potential of medicinal plant species. Food Bioprod Process 2011; 89(3):217-33. doi:10.1016/j.fbp.2010.04.008
- [52]Gupta V, Sharma S. Plants as natural antioxidants. Nat Prod Radiance 2006; 5(4):326-34.
- [53]Lallianchhunga MC, Ali MA, Lalchhandama C, Lalmuanthanga C, Devi LI. Antioxidant Activity of Methanolic Extract of Mikania micrantha World J Pharm Res 2016; 5(4):879-86.
- [54]Dong LM, Jia XC, Luo QW, Zhang Q, Luo B, Liu WB et al. Phenolics from Mikania micrantha and Their Antioxidant Activity. Molecules 2017; 22(7): E1140. doi:10.3390/molecules22071140
- [55]Foti MC. Antioxidant properties of phenols. J Pharm Pharmacol 2007; 59(12):1673-85. doi:10.1211/jpp.59.12.0010
- [56]Raina R, Prawez S, Verma PK, Pankaj NK. Medicinal Plants and their Role in Wound Healing. Vet Scan 2008; 3(1):1-7.
- [57]Sarimah MN, Mizaton HH. The Wound Healing Activity Of Mikania micrantha Ehanolic Leaf Extract. J Fundam Appl Sci 2018; 10(6):425-37.
- [58]Rangaraj A, Harding K, Leaper D. Role of collagen in wound management. Wounds UK 2011; 7(2):54-63.
- [59]Sahu DS, Upadhyaya SN, Bora M, Puia L, Chowdhary S, Rao MM. Acute Dermal Toxicity And Wound Healing Activity Of Mikania micrantha Ointment In Rats. Indian J Appl Res 2019; 9(1):4-6. doi:10.36106/ijar
- [60]Qiu L, Zhang D, Huang H, Xiong Q, Zhang G. BP Neural Network Based Prediction of Potential Mikania micrantha Distribution in Guangzhou City. For Res 2018; 7(1):216. doi:10.4172/2168-9776.1000216
- [61]Kaur R, Malhotra S, Inderjit. Effects of invasion of Mikania micrantha on germination of rice seedlings, plant richness, chemical properties and respiration of soil. Biol Fertil Soils 2012; 48(4):481-8. doi:10.1007/s00374-011-0645-2
- [62]Shen S, Xu G, Clemen DR, Jin G, Liu S, Zhang F et al. Effects of Invasive Plant Mikania micrantha on Plant Community and Diversity in Farming Systems. Asian J Plant Sci 2015; 14(1):27-33. doi:10.3923/ajps.2015.27.33
- [63]Sahu A, Devkota A. Allelopathic Effects of Aqueous Extract of Leaves of Mikania micranthaB.K. on Seed Germination and Seedling Growht of Oryza sativa L. and Raphanus sativus L. Sci World 2013; 11(11):91-3. doi:10.3126/sw.v11i11.8559
- [64]Banerjee AK, Dewanji A. Mikania micranthaB.K.–a potential and economical threat to global biodiversity with special emphasis on Indian context. Conf Pap 2012. doi:10.13140/2.1.4486.9763
- [65]Shen S, Xu G, Zhang F, Jin G, Liu SML, Chen AYZ. Harmful effects and chemical control study of Mikania micranthaB.K in Yunnan, Southwest China. African J Agric Res 2013; 8(44):5554-61. doi:10.5897/AJAR2013.7688
- [66]Devi M. Invasive Species Mikania micrantha an Environmental Threat, Its Control With Reference To Eri Silk Worm. Int J Adv Sci Eng Technol 2017; 5(3):7-10.
- [67]Melander B, Rasmussen IA, Bàrberi P. Integrating Physical and Cultural Methods of Weed Control: Examples from European Research. Weed Sci 2005; 53(3):369-81. doi: 10.1614/WS-04-136R
- [68]Clements DR, Day MD, Oeggerli V, Shen SC, Weston LA, Xu GF et al. Site‐specific management is crucial to managing Mikania micrantha. Weed Res 2019; 59(3):155-69. doi:10.1111/wre.12359
- [69]Rai RK, Sandilya M, Subedi R. Controlling Mikania micrantha HBK: How effective manual cutting is? J Ecol Environ 2012; 35(3):235-42. doi:10.5141/JEFB.2012.029
- [70]Culliney TW. Benefits of Classical Biological Control for Managing Invasive Plants. CRC Crit Rev Plant Sci 2005; 24(2):131-50. doi:10.1080/07352680590961649
- [71]Islam MA, Nain Z, Alam MK, Banu NA, Islam MR. In vitro study of biocontrol potential of rhizospheric Pseudomonas aeruginosa against Fusarium oxysporum sp. cucumerinum. Egypt J Biol Pest Control 2018; 28(1):90. doi:10.1186/s41938-018-0097-1
- [72]Cock MJW. Potential biological control agents for Mikania micrantha HBK from the Neotropical region. Trop Pest Manag 1982; 28(3):242-54. doi:10.1080/09670878209370717
- [73]Day MD, Kawi AP, Fidelis J, Tunabuna A, Orapa W, Swamy B et al. Biology, Field Release and Monitoring of the Rust Fungus Puccinia spegazzinii ( Pucciniales : Pucciniaceae ), a Biological Control Agent of Mikania micrantha (Asteraceae) in Papua New Guinea and Fiji. XIII Int Symp Biol Control Weeds 2011; S5:211-7.
- [74]Matawali A, Chin LP, Eng HS, Boon LH, Gansau JA. In-vitro evaluation of anti-kinase, anti-phosphatase and cytotoxic activities of Mikania micranthaB.K. (asteraceae) from Malaysia. J Chem Pharm Sci 2016; 9 (2): 696-701.