Preliminary analysis of phytochemicals and in vitro free radical scavenging activity of Dhanwantaram Kashayam
Abstract
Dhanwantaram Kashayam (DK) is a polyherbal decoction used in Ayurveda for the postnatal care of mothers and for treating gynaecological diseases. It is also used as a growth stimulant in children as well as a regenerative medicine. Present study was to assess the various phytochemicals present in DK and to elucidate the role of DK as an effective antioxidant and free radical scavenger. Phytochemicals such as total phenolic content, total flavonoids and total tannins were assessed using standard biochemical methods. This study also investigated it’s in vitro antioxidant activity by ferric reducing antioxidant power (FRAP) assay and the free radical scavenging activity by assessing the scavenging activities on 2’2-Diphenyl-1-Picryl Hydrazine (DPPH), 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS), phosphomolybdenum, hydrogen peroxide, nitric oxide and the hydroxyl radical. Phytochemical assay reported fairly high levels of tannins 0.22 ± 0.015mg/g, total flavonoids 1.23 ± 0.043 mg/g and total poly phenolic content 10.05 ± 0.94 mg/g. DK was found to have very good antioxidant activity and it scavenged the different free radicals in a dose dependent manner with low IC50 values (DPPH: 2.08±0.051, ABTS:38.46±2.75, phosphomolybdenum 50.4±2.63, H2O2: 57.9±3.15, NO: 57.25±3.7, all expressed in µg/ml. Values were significant with p<0.05). Results of this study clearly revealed that the DK is rich in phytochemicals and is a good source of natural antioxidants and is an efficient scavenger of peroxide radicals. This supports the use of DK in Ayurveda as a regenerative medicine, but further studies are needed to correlate the in vitro observations with its pharmacological effects in vivo.
INTRODUCTION
Plants synthesize different phytochemicals [1, 2] which are good antioxidants help in the fight against damages caused by reactive oxygen species (ROS) in animals [3, 4]. The vitamins A, C, E, and different phenolic compounds such as flavonoids, tannins, and lignins are the major phytochemicals with significant antioxidant capacity [3]. Other phytochemicals such as beta carotene, ascorbic acid and different phenolic compounds found in plants were found to have anti-inflammatory activity [5], and prevent or ameliorate symptoms of degenerative diseases like diabetes, Alzheimer’s [6], Parkinson’s [7], and certain cancers [8, 9].
Oxidative stress is reported to be the major cause of the pathogenesis of chronic diseases like diabetes. Reactive oxygen species (ROS) and free radicals play major roles in the progression and development of diseases like cancer, asthma and diabetes [10]. Phytochemicals are found to protect our body by lessening oxidative stress as well as by removing ROS [11]. Dhanwantaram Kashayam (DK) is a poly herbal formulation in the form of a decoction and the herbs used for its preparation are having regeneration property [12]. DK has excellent antioxidant properties as well [13].
Drugs of plant origin from Ayurveda have already proved to be excellent leads for drug development [14]. DK is used in Ayurveda for many ailments and improving general health without any noticeable side effects, but as in the case of many other ayurvedic drugs, no objective, verifiable data exist to support many such claims [15]. As DK is well known as a rejuvenating drug as well as having a suggested ability of preventing tissue regeneration in Ayurveda, in the present study, we investigated on the phytochemical components with antioxidant capacity of DK. We mainly focused on the possible phytochemical components like total tannins, total flavonoids and total phenolic content. In vitro antioxidant power as well as free radical scavenging activity through the assays like ferric reducing antioxidant power (FRAP) and 2’2-diphenyl-1-picryl hydrazine (DPPH) scavenging activity, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical scavenging assay, phosphomolybdenum assay, hydroxyl radical scavenging activity, hydrogen peroxide-scavenging activity and nitric oxide scavenging activity of DK were also studied to delineate its mode of action.
The observations in this study provides information about the phytochemical composition as well as the antioxidant power of DK. This information throws light into the possible mode of action and provide experimental support for its use in Ayurvedic system of medicine in managing various diseases initiated by oxidative stress. Unravelling pharmacological information of Ayurvedic drugs in contemporary scientific language would help in expanding its acceptance worldwide in the management of various disorders. This will also help in the development of new cost-effective drugs from natural resources.
MATERIALS AND METHODS
Chemicals
All biochemicals used for the study were obtained from Sigma-Aldrich, Bangalore, India and Sisco Research Laboratories (Mumbai, India). Folin–Ciocalteu’s reagent, methanol, ethanol, sulfuric acid, sodium carbonate, sodium nitrite, sodium hydroxide, sodium acetate, acetic acid, aluminium chloride were purchased from Loba Chemie Pvt Ltd, Mumbai, India and iron (III) chloride, 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′- azinobis-(3-ethylben-zothiazoline-6-sulfonicacid) (ABTS), 2,4,6-tris(1-pyridyl)-5-triazine (TPTZ), were purchased from Sigma– Aldrich. Gallic acid was from HiMedia Laboratories, Mumbai, India. All chemicals and reagents used in the study were analytical grade.
DK collection
DK (Batch No. 518524) used in this study was purchased from ‘Kottakkal Arya Vaidya Sala’ (Kottakkal, Kerala, India) in the form of decoction.
Estimation of total tannins
The total tannin content was determined using the method of Broadhurst and Jones, 1978 [16]. Different concentrations of tannic acid (0.5-2.5ml) were taken in test tubes labelled as standard. 0.5ml of DK was taken in test tube marked as test and the volume of all the tubes were made up to 3ml with methanol. 5 ml of HCI- Vanillin regent (equal mixture of 4% HCl in methanol and 2% vanillin in methanol) was added to all the tubes. Tubes were incubated at 300C in water bath for 30 minutes and cooled in room temperature. Absorbance was measured at 495 nm against methanol blank using a UV-Vis spectrophotometer (Shimadzu, UV-1800, Japan). The total tannin content was expressed in terms of tannic acid equivalent (mg TAE/ml).
Total flavonoid content
Total flavonoid content was estimated using the method of Barros et al. 2010 [17]. 500 μl of DK was taken in a test tube and to this added 2ml of deionized water and 150 μl of sodium nitrite solution (5% w/v). After that, the mixture was incubated for 6 min at room temperature. After incubation, 150 μl of aluminium nitrate solution (10% w/v) was added and allowed to stand for another 6 min. Then added 3 ml of NaOH solution (4% w/v) and made up the volume to 6ml with deionized water. The reaction mixture was kept in dark at room temperature for 15 min. The intensity of the pink colour developed indicated the concentration of the flavonoid content in the DK. The intensity of the developed pink colour was measured at 510 nm using a spectrophotometer. Total flavonoid content was calculated with the help of a standard curve with catechin and the flavonoid content was expressed as mg catechin equivalents (CE)/g.
Total phenolic content
Total Phenolic Content was estimated according to the method of Kosar et al. [18]. 50 μl of DK and 2 ml of distilled H2O were in a test tube and added 250 μl of undiluted Folin-Ciocalteu reagent. 750 μl sodium carbonate (Na2CO3, (20% w/v) was added to the mixture to neutralise it. The volume was made up to 5 ml with deionised H2O. The mixture was incubated for two hours in the dark and at room temperature with continuous shaking. The absorbance of the developed colour was measured at 765 nm in a spectrophotometer. The total phenolic contents were determined from the linear equation of a standard curve prepared using Gallic acid as standard. The levels of total phenolic compounds were expressed as mg/g gallic acid equivalent (GAE) of DK.
Ferric reducing antioxidant power (FRAP) of DK
Ferric reducing antioxidant power of DK was assayed according to the method of Vijayalakshmi and Ruckmani [19]. Different volumes of the DK (10-50µl) were taken in different test tubes and the volumes were made up to 50µl with distilled water. 2.5ml of 0.2M sodium phosphate buffer (pH 6.6) and 2.5ml of 1% potassium ferricyanide [K3Fe(CN)6] solution was added to this. Mixed well using a vortex machine and incubated for 20min at 50oC which reduced the ferricyanide into ferrocyanide. 2.5ml of 10% trichloroacetic acid to the mixture to stop the reaction and centrifuged at 3,000 rpm for 10min. 2.5ml of the supernatant was mixed with equal volume of deionized water and 0.5ml of 0.1% ferric chloride. The colour intensity was measured at 700nm using a UV-visible spectrophotometer (Shimadzu, UV-1800, Japan). Ascorbic acid (AA) was used as a reference standard and the calibration curve was prepared by plotting the absorbance against the concentrations of FeSO4 used to reduce specific concentrations of AA. The reducing powers of the samples were calculated using the standard curve of the reference standard and expressed as mg/ml.
Scavenging power of DK on DPPH
The DPPH scavenging activity was measured by the method of Blois [20]. Different volumes of DK were taken in different test tubes and the difference in volume was compensated by adding distilled water. Control was prepared without DK, but volume compensated by distilled water. To this 1ml of 0.1 mM DPPH Solution was added and made up to a final volume of 4ml with 95% ethanol. The mixture was vigorously shaken and incubated for 30 minutes in the dark at room temperature. The colour of the reaction mixture changed from purple to yellow causing a decrease in absorbance at wavelength 517 nm in proportion to the DPPH scavenging power of the DK. The reduction in the color intensity of the solution by free radicals (DPPH) was measured at 517nm against ethanol blank using a spectrophotometer.
Ascorbic acid concentrations varying between 0-12 μg/ml (0, 2, 4, 6, 8, 10 and 12) prepared from a stock solution containing 100µg ascorbic acid in one ml of ethanol were used for making standard curve for antioxidant activity assay.
The capability of DK to reduce DPPH was determined by using the following equation and expressed as % of inhibition:
![]()
Effect of DK in scavenging ABTS radical
Free radical scavenging activity of DK was determined by ABTS cation radical scavenging assay [21]. ABTS·+ cation radical was produced by adding 7.4 mM ABTS (2, 20-azinobis-(3-ethylbenzothiazoneline-6-sulphonic acid) in water with 2.45 mM potassium per sulphate (1:1). The mixture was prepared 12-16 h before use and stored at room temperature in dark. The solution was diluted with 98% of ethanol to obtain an absorbance of 0.7 -1.5 at 734 nm. Different volumes (10-100 μl) of DK were taken in different test tubes containing the above reagent, and mixed thoroughly. The difference in volume of DK added was compensated by adding 98% ethanol. Kept at room temperature for 90 min during which the colour change from slightly yellow to an intensely turquoise (blue/green) colour with an absorbance at 405 nm. The OD of the developed colour was examined at 405 nm using a UV–vis spectrophotometer. OD of the reagent without adding DK, but 100 μl 98% ethanol was taken as the initial absorbance. Likewise, antioxidant capacity of trolox was also determined. The capability to scavenge the ABTS radical cation was calculated using the following equation;
![]()
Where, Ab1: the absorbance of the control (ABTS solution without DK/trolox), and Ab2: the absorbance in the presence of DK or trolox.
Reducing power of DK on phosphomolybdenum
Phosphomolybdenum method of Prieto et al. was employed to measure the antioxidant activity of DK [22]. Different volumes of DK were taken in different test tubes and the volume was made to 100μl using distilled water. 1 ml of reagent solution (0.6 M sulphuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate) was added to these test tubes. The tubes were capped and incubated in a water bath at 95°C for 90 min. After cooling to room temperature, the absorbance of the mixture was measured at 765 nm against a blank. Percent inhibition was calculated by using the formula and IC50 was calculated.
![]()
Effect of DK on hydroxyl radicals
The deoxyribose method of Kunchandy and Rao [23] was used to assess the hydroxyl radicals scavenging activity of DK. The competition between deoxyribose and DK for hydroxyl radicals generated from the Fe3+/ascorbate/ EDTA/ H2O2 system (Fenton reaction) was assessed in this method. The hydroxyl radicals attack deoxyribose and form thiobarbituric acid reactive substance (TBARS) as final product. Various volumes of DK (10- 100 μl) was taken in different test tubes and the volumes were made up to 100 μl with distilled H2O. 900 μl of the reagent containing deoxy ribose (28mM), FeCl3 (0.1mM), EDTA (0.1mM), H2O2 (1mM), ascorbic acid (0.1mM), KH2PO4-KOH buffer (20mM, pH 7.4) was added to each test tube with DK. The mixture was incubated for 1hr at 37°C. After incubation 0.5 ml of this reaction mixture was added to 1 ml of 2.8% TCA. Colour was developed by adding 1 ml of 1% aqueous thiobarbituric acid (TBA) and incubating the mixture at 90°C for 15 min. The mixture was cooled and the absorbance was measured at 532 nm against a blank containing all reagents except DK. Mannitol, a classical OH. scavenger was used as a positive control. Percentage of inhibition was evaluated by comparing with the test and blank solutions.
The ability to scavenge the hydroxyl radical was calculated using the equation;
![]()
Ability of DK to scavenge hydrogen peroxide
The ability of DK to scavenge hydrogen peroxide was determined by the method of Ruch et al. [24]. Different concentrations of DK (10, 20, 40, 60, 80, and 100 μg) were taken in test tubes and the volumes were made up to 0.4 mL with 50 mM phosphate buffer (pH 7.4). 0.6 ml of hydrogen peroxide solution (2 mM hydrogen peroxide in 50 mM phosphate buffer- pH 7.4) was added to each test tube and vortexes. After 10min the absorbance of the remaining hydrogen peroxide was measured at 230 nm against a blank containing all reagents except hydrogen peroxide.
Hydrogen peroxide scavenging ability was calculated by the equation:
![]()
Nitric oxide scavenging activity of DK
NO scavenging activity was measured by the method of Marcocci et al. [25] with minor modifications. The scavenging activity of DK was tested against the nitric oxide generated from sodium nitroprusside. Varying volumes (10-100 µl) of DK were taken in different test tubes and the difference in the volume used was compensated by the addition of phosphate buffer. 1ml of Sodium nitroprusside (10 mM) in PBS (0.25 M, pH 7.4) was added to each test tube and incubated at 25ºC for 5 h. A mixture containing all reagents except DK, but with 100 µl of buffer was used as control. After 5 h 0.5 ml of Griess reagent (equal volumes of 1% sulphanilamide in 20% glacial acetic acid and 0.1% (w/v) naphthyl-ethylenediamine dihydrochloride) was added. The absorbance of the pink coloured mixture was measured at 546 nm. Ascorbic acid was used as standard. % NO scavenging activity was determined by the equation:
![]()
Where, A0 and A1: Absorbance of mixture before and after reaction with Griess reagent.
Calculation of IC50 values
A scatter graph was plotted in excel with concentration on X axis and % activity on Y axis. Equation for slope was generated from the excel software. IC50 value was calculated using the equation, where Y=50, M and C values were obtained from the equation itself.
Statistical analysis
Analysis was carried out in triplicate and data are represented as Mean ± SD (Standard Deviation) of six parallel measurements. Results were analyzed by using the SPSS -21 version software for windows. Statistical significance was calculated by using one – way ANOVA (analysis of variance). Differences with p-values equal to or less than 0.05 were considered as statically significant.
RESULTS
Phytochemicals in DK
Significant levels of tannins, phenolics and flavonoids were present in DK. Total tannin content was found to be 0.22 ± 0.015 mg TAE/g and total flavonoid content of DK was 1.23 ± 0.043mg CE/g. The results also showed that DK has high total phenolic content (10.05 ± 0.94 mg GAE/g).
FRAP activity of DK
Quantitative measurement of the antioxidant potential of DK was measured by using FRAP assay. Blue coloured Fe+2-tripyridyltriazine was formed from colourless Fe+3 by the action of electron donating antioxidant (DK). The intensity of the blue colour developed was measured by assessing the change in absorbance at 593 nm. The FRAP value of 1 ml of DK was 36.29 ± 0.90µg/ml. This FRAP value signifies the reducing power of DK and the ability to act as an efficient antioxidant.
DPPH radical scavenging activity of DK
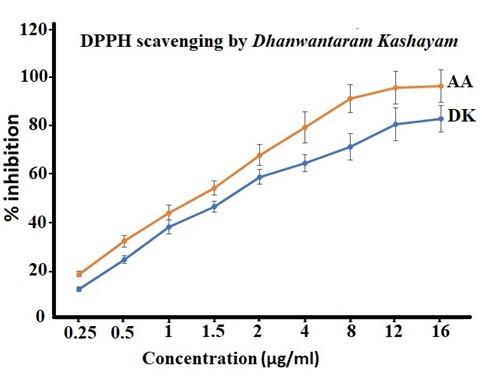
DK demonstrated strong percentage inhibition and DPPH radical scavenging activity (Figure 1), but not as strong as ascorbic acid. IC50 value of DK was 2.08 ± 0.11 µg/ml which was found to be double of ascorbic acid which was 1.03 ± 0.051 µg/ml. The result from the antioxidant assay showed that DK can scavenge the radical to a great extent.
ABTS.+ radical scavenging activity of DK
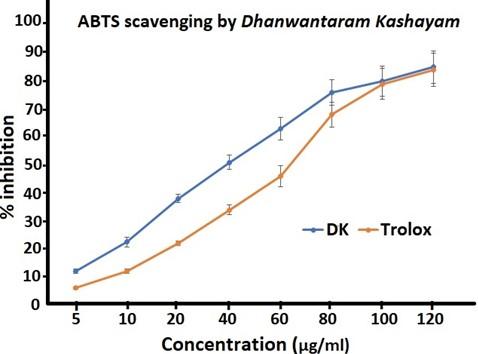
The capacity of DK to scavenge the ABTS.+ radical cation was assessed and the antioxidant capacities were expressed by IC50 value which indicated the concentration of DK needed to scavenge 50% of ABTS.+ radical. As shown in Figure 2, the IC50 value of the ABTS• + radical scavenging activity was 38.46 ± 2.75µg/ml. Comparing these values with standard (Trolox IC50 = 66.66 ± 1.51 µg/ml), it is obvious that DK is more effective in scavenging the ABTS.+ radical cation than trolox.
Reduction of phosphomolybdenum by DK
Antioxidant potency of DK was also assayed by the formation of green phosphomolybdenum complex which revealed strong effects of DK on reducing Molybdenum radical with an IC50 value of 50.4 ± 2.63μg/ml.
Hydroxyl radical scavenging activity of DK
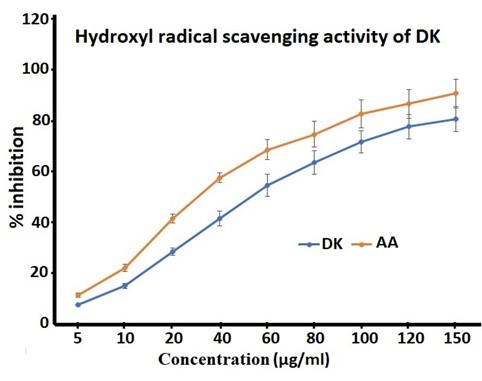
Hydroxyl radical is the most reactive among the oxygen radicals, which are reported to induce severe damage to biomolecules such as DNA, proteins and lipids and also causes lipid peroxidation which is the root cause of many tissue damages, cancer and cell death. Hence removal of this free radical is important in protecting life. Hydroxyl radical scavenging assay also showed DK as having notable effect. The results showed concentration dependent inhibition of DK against hydroxyl radical-induced degradation of deoxyribose in Figure 3.
Hydrogen peroxide scavenging activity of DK
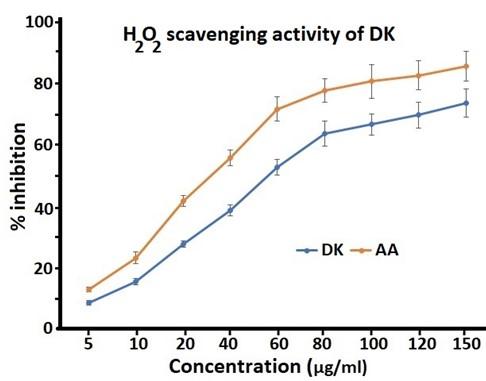
Hydrogen peroxide is nonreactive and unstable, but at high concentrations it is toxic to living cells. Certain cellular metabolic processes change H2O2 into hydroxyl radicals; which comes under the harmful group free radicals. Effective removal of H2O2 is important to maintain cellular health and normal functions. Therefore, the H2O2 scavenging effects of DK was evaluated and he results are shown in Figure 4. DK was found to have strong scavenging capacity with an IC50 of 57.9 ± 3.15 μg/ml, but was higher than standard ascorbic acid which was 35.8 ± 2.76 μg/ml. The H2O2 scavenging ability of DK was dose dependent which increased with increasing dose.
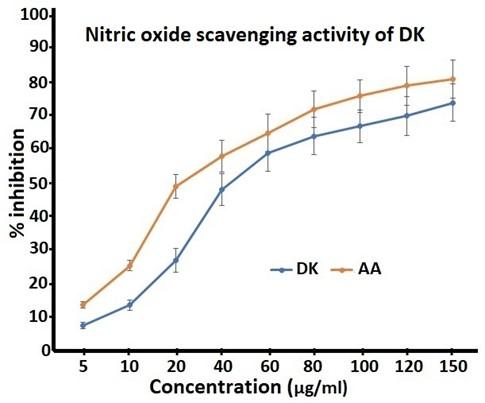
Nitric-oxide radical scavenging activity of DK
Nitric oxide (NO) is a physiologically important chemical produced in the body and regulate various physiological processes. NO scavenging capacity of DK was determined by the decrease in the absorbance at 546 nm which was caused by the antioxidants in DK. Optical density of NO was changed with increasing concentrations of DK. This OD change was in proportion to the amount of nitric oxide scavenged and the same was monitored. Dose-dependent increase in nitric-oxide radical scavenging activity was observed at all the studied concentrations of DK with an EC50 value of 57.25 ± 3.7 μg/ml, exhibited in Figure 5. The result shows that DK is a potent scavenger of NO radicals.
DISCUSSION
Phytochemicals are secondary metabolites produced by plants which protect plants from diseases. This phytochemicals are widely used for the treatment of numerous diseases like cancer and diabetes as they were found to prevent or delay symptoms of diseases due to antioxidant stress [26]. The present study determined the concentration of phytochemicals such as total tannins, total flavonoid and total phenolics in DK. Total tannins content was reasonably high in the DK (0.22 ± 0.015 mg TAE/ml). Besides, tannins act as potential drugs for the treatment of type II diabetes as it have the ability to enhance glucose uptake and inhibit adipogenesis [27]. The free radical scavenging ability as well as the ability to activate antioxidant enzymes of tannins is well reported. Various mechanisms including decrease in intestinal absorption of glucose [28], reduction in food intake [29], induction of β cell regeneration [30], enhancing insulin activity [31] are suggested as the main mechanisms behind the hypoglycemic effect of tannins. DK is used in Ayurveda to treat diabetes and we could suggest the involvement of tannins in this beneficial effect as tannin was reported to have anti-hyperglycemic activity in diabetic rats [32].
Flavonoids are large family of phytochemical compounds that are further divided into several subclasses namely anthocyanidins, flavanols, flavanones, flavonols and isoflavones [33]. Flavonoids are a group of hydroxylated phenolic substances known to act as potent free radical scavengers and have the ability to chelate metals [34]. In biological system it also reduces 𝛼-tocopherol radicals [35], and inhibits oxidases [36]. Bahadoran et al. in 2013 reported reduce risk of diabetes through consumption of flavonoids or flavonoid-rich foods [37]. Our present study revealed the richness of DK in flavonoids that are water-soluble polyphenolic molecules having antioxidant, free radical scavenging, and antimicrobial activities, [38] and this explain the beneficial effects of DK in various disease conditions and as a rejuvenating tonic.
The beneficial role of flavonoids in the positive modulation of carbohydrate and lipid metabolism is established in vitro animal models and human studies. The important role of flavonoids in attenuating hyperglycemia, insulin resistance, dyslipidemia, and adipose tissue metabolism are well demonstrated in these models. Alleviation of oxidative stress and stress-sensitive signaling pathways by flavonoids is well reported [37,39,40]. The ability of phenolic compounds to inhibit the absorption of amylase makes it useful in the treatment of diseases affecting carbohydrate absorption, such as diabetes [41].
Multiple phenolic hydroxyl groups in flavonoids makes them strong antioxidants which can effectively scavenge the reactive oxygen species [42]. The present study revealed the significant concentration of poly phenolic compounds in DK which bestow high antioxidant ability to DK. Our findings provide a good pharmacological logic for the therapeutic efficiency of DK in nerve disorders and as a growth stimulant [43]. High total polyphenol content increases the antioxidant activity as there is a linear correlation between phenolics content and antioxidant activity [44]. Plant derived drugs such as DK having free radical scavenging and antioxidant capacity [45] are used in many diseases like diabetes, cancer, inflammation and cardiovascular disease [46].
Phytochemicals increase antioxidant enzymes like catalase, GPx and GRd, which regulate blood glucose level and increase the insulin production in our body [47]. Plenty of plants are used in Ayurveda to treat diabetes mellitus and DK is the versatile product of 40 herbal ingredients. These plants containing natural antioxidants especially tannins and flavonoids have the ability to maintain pancreatic β-cells performance and decrease the glucose level in the blood [48]. Flavonoids, tannins, phenolic compounds, and alkaloids are the most common herbal active ingredients used for treating diabetes [49].
FRAP measure the ability of a compound to reduce ferric ion (Fe3+) to ferrous (Fe2+) ion and is a unique method for assessing antioxidant power [50]. In biological system, the reducing activity of an antioxidant against the oxidative effects of reactive oxygen species can be measured by FRAP assay. In this assay the Fe2+ produced is determined by measuring the absorbance maximum at 593 nm [50]. Increasing absorbance indicates increased production of Fe2+ which indicates increase in the reductive ability of antioxidants in the DK. AS previously reported; most herbal ingredients of DK have strong antioxidant activities [13]. In the present study the reducing power of DK was estimated to be 36.29 ± 0.90µg/ml and this excellent antioxidant power of DK might be one of the reasons for the benefits offered by DK.
DPPH radical scavenging activity is one of the commonly used methods for determining the antioxidant activity of plant extracts. In the present study, the antioxidant activity of the DK extract was analyzed by using this method and IC50 value of the DK extract was found to be 2.04 ± 0.11µg/ml. The addition of various concentration of DK (1 -15 µg/ml) caused the ethanolic solution of DPPH turns from deep violet color to light yellow color with increasing concentration of DK increasing the colour change. The main principle of this assay was reduction of the DPPH by antioxidants. Phytochemical analysis of DK extract had already shown that DK was rich with phenols, flavonoids and tannins. Presence of these components may be the reason for the reduction DPPH [51], and lower IC50 value of DK. Previous studies [38] had suggested that plant extracts or drugs that contain flavonoids, and polyphenols donate hydrogen atom to free radicals and thereby neutralize it.
The potential of DK to scavenge free radical was also assessed by ABTS radical inhibition assay, and DK was found to have an IC50 value of 38.46 ± 2.75 μg/ml, showing a strong activity. At concentrations higher than 80µg/ml, the ABTS scavenging activity of DK did not increase proportionately with the concentration. Suspecting precipitation of DK at higher concentration, the mixture was centrifuged at 5000rpm which resulted in a thin film of brown substance sedimenting at the bottom of the test tube. Oversaturation might have resulted in the precipitation of part of the DK which made it unavailable for scavenging ABTS. The antioxidant activities measured by ABTS or DPPH assay could be correlated to the concentration and chemical structures of the antioxidants present [52]. DK was found to have a reasonably good concentration of Total tannins, a high molecular weight phenolic compound. They are found to have high ability to quench free radicals [53]. Phytochemical components of DK justify the good ABTS scavenging activity of DK.
Antioxidant activity of materials could possibly be attributed to numerous mechanisms and binding of transition metal ion catalyst is one of these mechanisms. Molybdenum is a transition metal and the phosphomolybdate assay is another routine test used to evaluate the total antioxidant capacity [54]. We observed an IC50 value of 50.4 ± 2.63 μg/ml which is indicative of significant antioxidant power of the drug. The strong antioxidant activity of DK revealed by this assay might be due to the presence of phenolics compounds present [55].
The results of this study also revealed the strong scavenging activity of DK on hydroxyl radical, hydrogen peroxide and nitric oxide which might have resulted by the presence of bioactive flavonoids. H2O2 is not very reactive, but is important to the body due to its ability to penetrate into biological membranes. Its decomposition may produce hydroxyl radicals (OH•) [56] which can initiate lipid peroxidation and cause damage to cell membrane and DNA. The results of this study showed that the DK had potent H2O2 scavenging activity. Scavenging of H2O2 by DK could be attributed to the richness of DK in antioxidant compounds. The antioxidant components of DK could donate electron and may thus neutralize H2O2 to water. The DK was found to scavenge the H2O2 radicals with an excellent inhibition percentage and its ability was comparable to ascorbic acid and has 61% ability in comparison to ascorbic acid.
Hydroxyl radicals, an active oxygen species cause lipid peroxidation and are responsible for different damages in the body. DK was able to scavenge the hydroxyl radicals and the low IC50 value indicates high hydroxyl radical scavenging activity of DK. We have already reported the ability of DK to suppress lipid peroxidation in diabetic rats [57] and the hydroxyl radical scavenging ability of DK revealed in this study provides a possible explanation to the reduction in lipid peroxidation in DK fed rats. NO is very reactive and alter the structure and function of cellular components. In this assay linear time-dependent nitrite production occurs from sodium nitroprusside and got reduced by the antioxidants in DK. Antioxidant principles in the DK compete with oxygen and react with nitric oxide which inhibit the generation of nitrite. DK is rich in flavonoids and phenolic compounds and their nitric oxide scavenging activity are well reported [58].
In the DPPH, ABTS+ and phosphomolybdenum assay methods, hydrogen and electron transfer occur from antioxidants to DPPH−, ABTS+ and Mo (VI) complex, but in these methods the transfers occur at different redox potentials and also depend on the structure of the antioxidant. This difference can be used to identify the presence of different antioxidants to some extent. DPPH− and ABTS+ scavenging assays detect antioxidants such as flavonoids and polyphenols, whereas the phosphomolybdenum method usually detects antioxidants such as ascorbic acid, some phenolics, a-tocopherol, and carotenoids [55]. DK answered all these assays and is an indication of the presence of different antioxidants which establish its strong antioxidant activity.
CONCLUSION
Present study revealed the richness of DK with total tannins, total flavonoids and total phenolic content. DK is also found to have strong antioxidant and free radical scavenging activity. The strong antioxidant and free radical scavenging activity of DK suggested by the presence of these phytochemicals may be the reason for its beneficial impact in biological system as reported in Ayurvedic system of medicine.
ACKNOWLEDGEMENTS
No animals were used in this study. S Renganathan acknowledges the financial assistance in the form of Research fellowship from the University of Calicut.
AUTHOR CONTRIBUTIONS
Conceptualization: S Renganathan and RG Pillai; Methodology: S Renganathan and RG Pillai; Investigation: S Renganathan and RG Pillai; Formal analysis: S Renganathan and RG Pillai; Sample collection: S Renganathan and RG Pillai; Resources: S Renganathan and RG Pillai; Writing, review and editing: S Renganathan and RG Pillai. All authors have read and agreed to the published version of the manuscript.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Gibson EL, Wardel J, Watts CJ. Fruit and Vegetable Consumption, Nutritional Knowledge and Beliefs in Mothers and Children. Appetite. 1998;31(2):205-228.
- [2]Mathai K. Nutrition in the Adult Years. In Krause’s Food, Nutrition, and Diet Therapy (10thedn)., ed. L.K. Mahan and S. Escott-Stump. American Cancer Society: 2000;271:274-275.
- [3]Suffredini IB, Sader HS, Gonçalves AG, Reis AO, Gales AC, Varella, AD, et al. Screening of antibacterial extracts from plants native to the brazilian amazon rain forest and atlantic forest. Braz J Med Biol Res. 2004;37(3):379–384.
- [4]Boots AW, Haenen GR, Bast A. Health effects of quercetin: From antioxidant to nutraceutical. Eur J Pharmacol. 2008;585(2-3):325–337.
- [5]Pillai RG. Curcumin prolongs graft survival in mice models of corneal allografts by suppressing the expression of inflammatory factors. JETIR. 2019;6(6):272-277.
- [6]Silveira AC, Dias JP, Santos VM, Oliveira PF, Alves MG, Rato L, Silva BM. The Action of Polyphenols in Diabetes Mellitus and Alzheimer’s Disease: A Common Agent for Overlapping Pathologies. Curr Neuropharmacol. 2019;17(7):590–613.
- [7]Kujawska M, Jodynis-Liebert J. Polyphenols in Parkinson’s Disease: A Systematic Review of In Vivo Studies. Nutrients. 2018;10(5):642. doi: 10.3390/nu10050642.
- [8]Duthie SJ, Ma A, Ross MA, Collins AR. Antioxidant supplementation decreases oxidative DNA damage in human lymphocytes. Cancer Res. 1996;56(6):1291–1295.
- [9]Pillai RG. Antiproliferative actions of Ziziphus jujube fruit extract is mediated through alterations in Bcl2-Bax ration and through the activation of caspases. Chem Biol Lett. 2020;7(1):41-46.
- [10]Phaniendra A, Jestadi DB. Free radicals: properties, sources, targets, and their implication in various diseases. IJCB. 2015;30(1):11-26.
- [11]Ngoci SN, Mwendia CM, Mwaniki CG. Phytochemical and cytotoxicity testing of Indigofera lupatana Baker F. J Anim Plant Sci. 2011;11:1364-1373.
- [12]Joshi KS, Bhonde R. Insights from Ayurveda for translational stem cell research. J Ayurveda Integr Med. 2014;5(1):4-10.
- [13]Sruthi CV, Sindhu A. A Comparison of the antioxidant property of five Ayurvedic formulations commonly used in the management of vatavyadhis. J Ayurveda Integr Med 2012;3(1):29-32.
- [14]Rayudu V, Raju AB. Effect of Triphala on dextran sulphate sodium-induced colitis in rats. Ayu. 2014;35(3):333-338.
- [15]Panigrahi B, Sharma S, Sitapara B, De S, Nariya M. Safety profile of Ayurvedic poly-herbomineral formulation – Bacnil capsule in albino rats. Ayu. 2019;40(3):185-191.
- [16]Broadhurst RB, Jones WT. Analysis of Condensed Tannins Using Acidified Vanillin. J Sci Food Agric. 1978;29(9):788-794.
- [17]Barros L, Carvalho AM, Morais JS, Ferreira ICFR. Strawberry-tree, blackthorn and rose fruits: Detailed characterisation in nutrients and phytochemicals with antioxidant properties. Food Chem. 2010;120(1):247–254.
- [18]Koşar M, Göger F, Başer KHC. In vitro antioxidant properties and phenolic composition of Salvia virgata Jacq. from Turkey. J Agri Food Chem 2008;56(7):2369–2374.
- [19]Vijayalakshmi M, Ruckmani K. Ferric reducing anti-oxidant power assay in plant extract. Bangladesh J Pharmacol. 2016;11(3):570-572.
- [20]Blois MS. Antioxidant determinations by the use of a stable free radical. Nature. 1958;181(4):1199-1200.
- [21]Re R, Pellegrini N, Proteggente A, Yang M, Rice-Evans C. Antioxidant activity applying an improved ABTS radical cationdecolorization assay. Free Radic Biol Med. 1999;26(9-10):1231-1237.
- [22]Prieto P, Pineda M, Aguilar M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem. 1999;269(2):337-341.
- [23]Kunchandy E, Rao MNA. Oxygen radical scavenging activity of curcumin. Int Journal of pharmaceut. 1990;58(3):237-240.
- [24]Ruch RJ, Cheng SJ, Klaunig JE. Prevention of cytotoxicity and inhibition of intercellular communication by antioxidant catechins isolated from Chinese green tea. Carcinogenesis. 1989;10(6):1003-1008.
- [25]Marcocci L, Maguire JJ, Droy-Leffix MT, Packer L. The nitric oxide scavenging property of Ginkgo biloba extract. Biochemical and Biophysical Research Communications. 1994;201(6):748-755.
- [26]Saxena M, Saxena J, Nema R, Singh D, Gupta A. Phytochemistry of Medicinal Plants. J Pharmacogn Phytochem. 2013;1(6):168-182.
- [27]Kumari M, Jain S. Tannins: An Antinutrient with Positive Effect to Manage Diabetes. Res J Recent Sci. 2012;1(12):1-8.
- [28]Shimizu M, Kobayashi Y, Suzuki M, Satsu H, Miyamoto Y. Regulation of intestinal glucose transport by tea catechins. Biofactors. 2000;13(1-4):61-65.
- [29]Kao YH, Hiipakka RA, Liao S. Modulation of endocrine systems and food intake by green tea epigallocatechin gallate. Endocrinology. 2000;141(3):980-987.
- [30]Kim MJ, Ryu GR, Chung JS, Sim SS, Min DS, Rhie DJ, et al. Protective effects of epicatechin against the toxic effects of streptozotocin on rat pancreatic islets: in vivo and in vitro. Pancreas. 2003;26(3):292-299.
- [31]Anderson RA, Polansky MM. Tea enhances insulin activity. J Agric Food Chem 2002;50(24):7182-7186.
- [32]Pinent M, Blay M, Blade MC, Salvado MJ, Arola L, Ardevol A. Grape seed derived procyanidins have an antihyperglycemic effect in streptozotocin-induced diabetic rats and insulinomimetic activity in insulinsensitive cell lines. Endocrinology. 2004;145(11): 4985-4990.
- [33]Testa R, Bonfigli AR, Genovese S, Nigris VD, Ceriello A. The Possible Role of Flavonoids in the Prevention of Diabetic Complications. Nutrients. 2016; 8(5):310.
- [34]Arts IC, Hollman PC. Polyphenols and disease risk in epidemiologic studies. Am J Clin Nutr 2005;81(1):317S–325S.
- [35]Hirano R, Sasamoto W, Matsumoto A, Itakura H, Igarashi O, Kondo K. Antioxidant ability of various flavonoids against DPPH radicals and LDL oxidation. J Nutr Sci Vitaminol 2001;47(5):357–362.
- [36]Lima CC, Lemos RP, Conserva LM. Dilleniaceae family: an overview of its ethnomedicinal uses, biological and phytochemical profile. J Pharmacogn Phytochem. 2014;3(2):181–04.
- [37]Bahadoran Z, Mirmiran P, Azizi F. Dietary polyphenols as potential nutraceuticals in management of diabetes: A review. J Diabetes Metab Disord. 2013;12(1):43.
- [38]Uddin MS, Hossain MS, Mamun AA, Tewari D, Asduzzaman M, Islam MS, et al. Phytochemical analysis and antioxidant profile of methanolic extract of seed, pulp and peel of Baccaurea ramiflora Lour. Asian Pacific Journal of Tropical Medicine. 2018;11(7):443-450.
- [39]Johnston K, Sharp P, Clifford M, Morgan L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett. 2005;579(7):1653–1657.
- [40]Jung UJ, Lee MK, Jeong KS, Choi MS. The hypoglycemic effects of hesperidin and naringin are partly mediated by hepaticglucose-regulating enzymes in C57BL/KsJ-db/db mice. J Nutr. 2004;134(10):2499–2503.
- [41]Sales PM, Souza PM, Simeoni LA, Magalhães PO, Silveira D. α-Amylase Inhibitors: A Review of Raw Material and Isolated Compounds from Plant Source. J Pharm Pharm Sci. 2012;15(1):141–183.
- [42]Cao G, Sofic E, Prior RL. Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radic Biol Med. 1997;22(5):749-760.
- [43]Renganathan S, Srivastava AS, Pillai RG. Dhanwantharam kashayam, an AyurvedicPolyherbal Formulation, Reduces Oxidative Radicals and Reverts Lipids Profile towards Normal in Diabetic Rats. Biochem Biophys Rep. 2020; 22. doi:10.1016/j.bbrep.2020.100755.
- [44]Khan RA, Khan MR, Sahreen S, Ahmed M. Assessment of flavonoids contents and in vitro antioxidant activity of Launaea procumbens, Chem Cent J. 2012;6:43. doi: 10.1186/1752-153X-6-43.
- [45]Al-Snafi AE. Medicinal plants with antioxidant and free radical scavenging effects (part 2): plant based review. IOSR Journal Of Pharmacy. 2016;6(7):62-82.
- [46]Khan A, Suleman M, Baqi A, Samiullah, Ayub M. Phytochemicals and their role in curing fatal diseases: A Review. Pure Appl Biol. 2019;8(1):343-354.
- [47]Rizvi SI, Mishra N. Traditional Indian medicines used for the management of diabetes mellitus. J Diabetes Res. 2013.
- [48]Kooti W, Farokhipour M, Asadzadeh Z, Larky DA, Samani MA. The role of medicinal plants in the treatment of diabetes: a systematic review. Electronic Physician. 2016;8(1):1832-1842.
- [49]Rashid AM, Hossain MS, Hassan NB, Dash KKM, Sapon A, Sen MK. A review on medicinal plants with antidiabetic activity. J Pharmacogn Phytochem. 2014;3(4):149-159.
- [50]Benzie IFF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 1996;239(1):70-76.
- [51]Boly R, Lamkami T, Lompo M, Dubois J, Guissou IP. DPPH Free Radical Scavenging Activity of Two Extracts from Agelanthus dodoneifolius (Loranthaceae) Leaves. IJTPR. 2016;8(1):29-34.
- [52]Oszmianski J, Wolniak M, Wojdylo A, Wawer I. Comparative study of polyphenolic content and antiradical activity of cloudy and clear apple juices. J Sci Food Agric. 2007;87(4):573–579.
- [53]Hagerman AE, Riedl KM, Jones GA, Sovik KN, Ritchard NT, Hartzfeld PW, Riechel TL. High Molecular Weight Plant Polyphenolics (Tannins) as Biological Antioxidants. J Agric Food Chem. 1998;46(5):1887-1892.
- [54]Prieto P, Pineda M, Aguilar M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Analytical Biochemistry. 1999;269(2):337–341.
- [55]Pourreza N. Phenolic Compounds as Potential Antioxidant. Jundishapur J Nat Pharm Prod. 2013;8(4):149–150.
- [56]Sasikumar V, Kalaisezhiyen P. Evaluation of Free Radical Scavenging Activity of Various Leaf Extracts from Kedrostis foetidissima (Jacq.) Cogn. Biochemistry & Analytical Biochemistry, 2014, 3:2 DOI: 10.4172/2161-1009.1000150.
- [57]nganathan S, Pillai RG. Antioxidant activities of Dhanwantaram Kashayam- an ayurvedic polyherbal formulation alleviates diabetic complications in rats, J Diabetes and Metabolic Disorders, 2020, DOI 10.1007/s40200-020-00655-5
- [58]Parul R, Kundu SK and Saha P, Nitric Oxide Scavenging Activity Of Methanol Extracts Of Three Bangladeshi Medicinal Plants, The Pharma Innovation Journal, 2013; 1 (12): 83-90.