Assessment of the anti-leukemic and antioxidant potential of the methanol extract of a wild, edible, and novel mushroom, Astraeus hygrometricus, and unraveling its metabolomic profile
Abstract
Mushrooms are enriched with a plethora of bioactive molecules that play a vital role in the prevention of human diseases. The balance between ROS generation and cancer growth is one of the main prerequisites for efficient cancer treatment. In this study, to testify the aforesaid theory, five wild edible mushrooms were initially screened based on their anti-proliferative efficiency, and the best mushroom extract was selected for the assessment of their antioxidant potentialities in vitro in various artificially generated free radicals such as DPPH, ABTS+, and by FRAP experiment. The reason behind the antiproliferative potentiality and antioxidant capability of the most potent extract was also correlated by profiling its metabolites through GC-MS analysis. The study reveals, that the methanolic extract of Astraeus hygrometricus is the most potent anti-leukemic extract (IC50 22.7 ±0.23 µg/mL) followed by Serpula sp. (75.7 ±0.44 µg/mL), Phallus sp. (60.53±0.36 µg/mL), Tricholoma sp. (53.76±0.46 µg/mL), Lentinus sp. (58 ±0.13 µg/mL) against the Jurkat cell line. The assessment of the antioxidant profile Astraeus hygrometricus reveals its moderate antioxidant efficacy against several artificially generated free radicals, such as DPPH (76.9±0.16 µg/mL), ABTS+ (142±0.66 µg/mL) and moderate iron chelating efficacy (32.37±2.31µM). The GC-MS analysis of both methanol and ethyl acetate extracts of Astraeus hygrometricus have found 53 and 52 compounds respectively, with wide diverse ranges of chemically classified biomolecules such as alkane, alcohol, fatty acid, organic acid, polycyclic and heterocyclic compounds, amino acid, vitamin, and hormone, etc. with a wide array of biological activity such as anticancer and antioxidant potentiality. In conclusion, it can be said that these wild edible mushroom Astraeus hygrometricus are a repository of novel biomolecules that can be used in the treatment of Leukemia in the future.
INTRODUCTION
The basic prerequisite for all living organisms is energy which is produced by the oxidation process, and the generation of reactive oxygen species (ROS) as a byproduct is an inevitable outcome of it. However, the indigenous antioxidant mechanism to counter oxidative stress is present. Still, the continuous production of these uncontrolled free radicals in vivo may lead to various health disorders and even lead to the development of various non-communicable diseases like cancer [1–3]. Today's cancer treatment is mostly confined to the use of different combinations of chemotherapeutic drugs, but most of the commercially available chemotherapeutic drugs show various degrees of side effects, most commonly the overproduction of ROS, and thus leads to oxidative stress. To counteract the negative consequences of the phenomenon, various synthetic antioxidant molecules such as butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), tert-butylated hydroxyquinine (TBHQ), etc. have been used as an adjuvant in chemotherapy, but these synthetic antioxidant drugs in most the cases show various side effects [4,5]. So, to allay the negative consequences of the existing drugs, there is an urgent need for the development of safe anticancer and antioxidant exogenous drugs preferably from natural sources.
In recent years, mushroom-derived foods, as it contains a wide range of secondary metabolites have attracted attention as a natural reservoir of effective commercial antioxidant and anticancer molecules [6-8]. Mushroom contains a wide variety of bioactive compounds such as diverse phenol and flavonoid molecules, polysaccharides, glycosides, tocopherols, carotenoids, alkaloids, volatile oils, organic acids, and has the potential to serve as an alternative reservoir for naturally occurring antioxidants, anti-inflammatory, cardiovascular, anti-microbial, immunomodulation, anticancer, and anti-diabetic drug [3,9–11]. Low molecular weight substances isolated from wood degrading fungus Cerrena unicolor have shown dual efficiency, as an anticancer molecule towards two breast cancer cell lines MDA-MB-231, MCF7 and one prostatic carcinoma cells PC3 with IC50 value less than 7 mg/mL and also as antioxidant molecule against artificially generated free radicle 1,1-diphenyl-2-picryl-hydrazyl, with promising half-maximal inhibitory concentration ranging in between 20.39 μg/mL to 64.14 μg/mL [12]. Another impotent β-Glucan, lentinan isolated from edible mushroom Lentinus edodes, is also used as both an anticancer and antioxidant agent [13].
Profiling of total primary and secondary metabolome has been considered as one of the reliable methods to investigate primarily the possible group of molecules responsible for the desired bioactivity. In order to look at the metabolomic profile, GC-MS method has been widely used to detect several abundant molecules such as steroids, essential oil, triterpenoid, diterpenoid, phytosterol group of compounds etc. that are responsible for bioactivity of any extract are individually detected reliably, even they are present in heterogenous extract [14]. The metabolomic profiling, through the GC-MS analysis of methanolic extract of a wild mushroom Phallus sp. has revealed presence of different derivatives of anticancer sterol ergo sterol thus directly pointing out its anti-cancer efficacy of Phallus sp. [4].
Therefore, the present study was carried out to compare the anticancer potentiality between methanol and ethyl acetate extracts of five wild edible mushrooms Astraeus hygrometricus, Phallus sp., Lentinus sp., Tricholoma sp., and Serpula sp. and the most effective extract of best mushroom was selected for antioxidant assessment to investigate the relationship between anti-cancer activity and ROS management in vitro. Consequently, the metabolomics profile of potent extracts was also conducted to point out the probable bioactive components responsible for this anticancer and antioxidant efficacies.
MATERIALS AND METHODS
Sample collection and identification
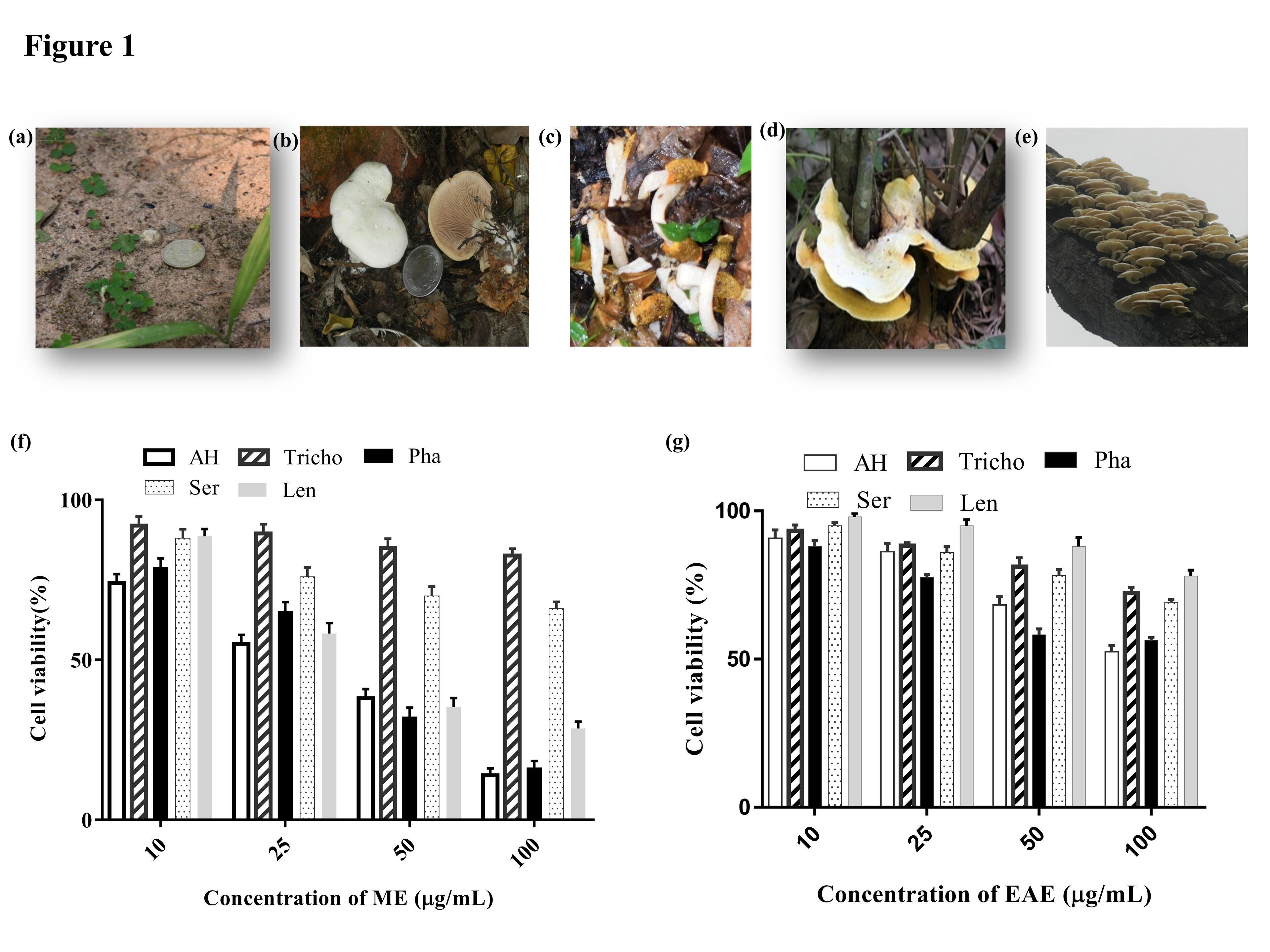
Mushroom fruit bodies were collected from various locations of West Bengal, the eastern state of India in between the months of May to August between 2015 –2019. The mature fruit body of Phallus sp. was collected from Lolegaon (27.0206°N, 88.5650°E) Kalimpong District positioned under Himalayan foothills, Tricholoma sp. and Serpula sp. from Mathurapur (22.1203° N, 88.3943° E) hinterland of Bay of Bengal South 24-Parganas District, Astraeus hygrometricus from Jamboni (22.4502° N, 86.8998° E), Lentinus sp. From Sigram (22.4479° N, 86.8967° E) belonged to the laterite region of Jhargram District (Figure 1a-e). All possible morpho-organoleptic characteristics such as color, size, and the shape were tested immediately after collection.
Identification of the mushroom
Regarding the identification of mushrooms, both classical methods (morphological and anatomical methods) and molecular identification methods were applied. Astraeus hygrometricus, Serpula sp., was identified by Prof. Krishnendu Acharya, and Lentinus sp. was identified by Prof. Santanu Paul with the consultation of various standard published keys, and the specimen sample was preserved in Calcutta University Herbarium with voucher number CUH/AM/681 (Astraeus hygrometricus) and CUH/FN/JH/SP/04 (Lentinus sp.). Identification of the other two samples was validated by molecular identification, by amplifying its conserved internal transcribed spacer regions (ITS1, 2, 5.8s rDNA) along with conventional identification methods.
Molecular identification of the mushroom-
DNA extraction: The genomic DNA of the mushroom was extracted from air-dried samples in accordance with the manufacturer protocol of fungal gDNA Mini Kit (Xcelris Genomics, Ahmedabad, India).
Polymerase Chain Reaction (PCR) -Amplification of its specific Internal transcribed spacer regions (ITS1, 2, 5.8s rDNA), was done by selecting a pair of primer sequence ITS1 (forward primer 5’ TCC GTA GGT GAA CCT GCGG 3’) and ITS4 (reverse primer 5’ TCC TCC GCT TAT TGA TAT GC3’) [15]. The PCR reaction mixture was prepared as per the method described by Ray et.al. 2020[4]. The amplification reaction was designed as initial denaturation for 4 min at 94°C followed by 35 cycles consisting of 1 minute at 94°C (denaturation) then 1 minute in 56°C for annealing, 1 min at 72°C (extension), and finally one cycle for 10 min at 72°C for final elongation [4].
Sequencing- Automated DNA sequencing method, performed on an ABI3730XL-15104-028DNA Analyzer (Applied Bio systems, USA) was used for the sequencing of the ITS regions. The same sets of primers identical with amplicons for the ITS rDNA region were used for the sequencing reaction. The newly generated sequences were deposited in GenBank (www.ncbi.nlm.nih.gov).
Preparation of mushroom extracts
The collected mushroom samples were dried and ground to powder. Three types of solvents hexane, ethyl acetate, and methanol were selected based on the polarity for the preparation of mushroom extracts [8,16]. In brief, initially, 50 g of mushroom powder was taken and then percolated in hexane for three days to remove the fatty substances and the hexane extract was prepared through filtration followed by evaporation in a rotary evaporator. Repetition of the same procedure was performed to prepare the Ethyl Acetate (EAE) and Methanol Extract (ME) respectively. All the extracts were stored at 4°C for further experiments. The methanol extract was selected for further evaluation of the antioxidant and antiproliferative properties of selected mushrooms.
Chemicals
All chemicals that were used for the experiments were analytical grade and freshly prepared. DMSO, Gallic acid (Merck), Sodium Carbonate (Merck), FolinCiocalteau (Merck), Quercetin (SRL), Sodium nitrite (Himedia), Aluminium chloride (Merck), Sodium hydroxide (SRL), 2, 2-diphenyl-1-picrylhydrazyl (DPPH) (SRL), Methanol (Merck), Acetic acid (Merck), Thiobarbituric acid, n-Butanol (Himedia), ABTS, FeSO4,7H2O (MERK), acetate buffer, Sodium phosphate buffer, Phosphate buffer.
Cell line and cell culture
In order to assay the efficacy of the anti-proliferative potentiality of the crude extracts, one leukemic cell line, Jurkat (T Acute Lymphoblastic Leukemia) was selected, and the cell line was maintained in the RPMI medium supplemented with 10% FBS, 100U/mL penicillin, and 100 U/mL streptomycin incubated at 37oC in a humidified atmosphere containing 5% CO2.
MTT assay
The antiproliferative effect of both the crude extracts ME and EAE against Jurkat cells was determined by the MTT dye uptake method in accordance with Gopal et.al. 2014 [17]. Briefly, 1X104 cells were seeded in a 96-well plate with various concentrations of mushroom extracts 10, 25, 50 100 µg/mL for 24 hours at 37oC. Thereafter, 25μL MTT solution (5 mg/mL in PBS) was added to each well and the optical density (OD) was optically measured at 590. To measure 100% lysis of the cell, after the incubation of 2.5 hours at 37oC, 0.1 mL extraction buffer (20% SDS) was added and incubated overnight at 37oC, and finally, the OD was recorded at 590 mn. The percentage of cell viability was calculated according to the following equation: % cell viability = (O.D. sample – O.D. 100%lysis) / (O.D. 0%lysis – O.D.100%lysis) × 100 [17].
Measurement of the antioxidant potentiality of the ME
Determination of DPPH radical scavenging activity
DPPH scavenging activity of methanolic extract of mushrooms was evaluated according to the method of Hajra et al. 2018 and Pal et al. 2019 [18,8] with slight modification. Briefly, 800μL of DPPH solution (0.1mM/mL methanol) was added to 200 μL of ME extract ranging from 100 μg/mL-1000 μg/mL and incubated for one hour in the dark, the absorption was finally spectrophotometrically recorded at 517 nm. The experiment was performed in duplicates for each concentration. The percent of scavenging of DPPH radical was calculated according to the following equation: % DPPH reduction = [{(Ac–As)/Ac} ×100] (Where; As is the absorbance of sample, Ac is the absorbance of control) [8].
Determination of ABTS•+radical scavenging activity
The ABTS•+assay was performed in accordance of the method of Ray et al. 2020 [4] with minute modifications. Briefly, the working solution of ABTS•+ was prepared by allowing the reaction of the two stock solutions (7.4 mM ABTS and 2.6 mM potassium persulfate solution) in equal quantities for 12 hours at room temperature in the dark. After that, the final reaction solution of the ABTS radical was prepared by diluting the aforesaid solution in such a way, that obtain a spectrometric measurement of the absorbance of 0.7 (± 0.2) at 734 nm. ME of each mushroom with the concentration range 100μg/mL to 1000μg/mL was allowed to react with 2.85 mL of the ABTS solution for 2 hours in a dark condition, finally the absorbance was taken at 734 nm using the spectrophotometer. The percent of scavenging of the ABTS•+radical was calculated according to the following equation: % ABTS•+radical reduction = [{(Ac–As)/Ac} ×100] (Where; As is the absorbance of sample, Ac is the absorbance of control) [4].
Determination of percentage inhibition of lipid peroxidation
Thiobarbituric acid-reactive species (TBARS) assay was considered to measure the percentage inhibition of lipid peroxidation (LPO) by ME in accordance with the protocol of Tokuret al. 2006 [19] with some modifications. Briefly, 20% v/v egg homogenate and 0.1 mL ME (concentration range 100μg/mL-1000μg/mL) along with 0.07 M FeSO4 was incubated for 30 minutes. After that, acetic acid (pH 3.5), TBA in 1% SDS, was added to the reaction mixture and was vortexed and heated at 95°C for an hour. Finally, Butanol was added to each tube after the cooling down of the reaction mixture, and the tubes were centrifuged at 3000 rpm for 10 min. The upper organic layer was taken for spectroscopic measurement at 532 nm. Percentage inhibition of LPO by the crude methanolic extract was calculated according to the following equation: [(Absorbance Control-Absorbance Sample)/ Absorbance Control] ×100.
Determination of FRAP activity
FRAP activity of the ME was evaluated by following the protocol of Benzie et.al.1996 [20] with minor modification. In brief, the working solution of FRAP was prepared by mixing 300 mM acetate buffer, TPTZ, and FeCl3, 6H2O solution (10:1:1), and then warmed at 370C before use. Mushroom extracts (1.5 mL) were allowed to react with 2.85 ml of the FRAP solution for 30 minutes in dark conditions and finally, the resulted colored product [ferrous tripyridyltriazine complex] was measured at 593 nm by spectrophotometer. The biological property of any mushroom extract depends on the phytochemical compounds that present in the extracts. The phenol and flavonoid are considered the major phytochemical components present in biological mixtures. So, the total phenol and flavonoid content were considered and also quantitatively evaluated.
Determination of total phenol content
The total phenol content of the methanol extract of the five mushrooms was evaluated according to the method of Mridha et al. 2017 and Ray et al. 2020 by using the Folin-Ciocalteu reagent [4,21]. Briefly, an equal volume of the ME and 0.2 N Folin-Ciocalteu reagent was incubated for 3 minutes, followed by the addition of 10% Sodium carbonate and the mixture was vortexed and incubated in dark at room temperature for 40 minutes and finally, it was spectrophotometrically measured at 760 nm. Gallic acid was considered as standard phenol and a concentration range of Gallic acid (100µg/mL-1000 µg/mL) was prepared as a standard curve. The experimental replica was prepared in a duplicate manner. The result was expressed as mg of gallic acid equivalents (GAE) per gram of methanolic extract.
Determination of total flavonoid content
The total flavonoid content of the methanolic extracts of mushrooms was evaluated according to the method of Kamtekar et al., 2014 [22]. Briefly, 1 mL of the crude extract, 4 mL of distilled water, and 5% sodium nitrite solution were incubated for 5 minutes, followed by the addition of 10% aluminum chloride and 1 M NaOH, and the whole mixture was vortexed and finally spectrophotometrically measured at 510 nm. Different concentrations of Quercetin (10µg/mL-100 µg/mL) were considered to prepare a standard curve of standard flavonoid. The experimental replica was prepared in a duplicate manner. The result was expressed as mg of Quercetin equivalents (QE) per gram of methanolic extract.
Identification by Gas chromatography-mass spectrometry (GC-MS) analysis
The GC-MS profile of both EAE and ME of Astraeus hygrometricus was performed by gas chromatographic detectors 7890A (Agilent-Technologies) coupled with a mass spectrometer (MSD 7000). A capillary non-polar column HP-5MS (5% Phenyl Methyl Silox: Agilent 19091S-433) with the dimension of 30 m X 250 μm, along with film thickness: 0.25μm was considered to perform gas chromatographic analysis and gas chromatographic analysis and helium mobile phase with a constant flow rate of 2.25 mL min-1. 1 μL of the sample with a split ratio of 2:1 at 320 °C was considered for the injection and the oven was programmed as initially at 60 °C, then raised to 320°C for 12 min at 6 °C min-1 rates. The mass spectrometric analysis was programmed in a full scan module, with an electron impact of 70 eV in a range of 50-550 (m/z). The phytochemicals were identified by analyzing and comparing their related fragmentation profile with spectra present in the library NIST version 2.2 [4,14].
Statistical analysis
All the experimental data, that was obtained from three independent experiments represented as mean ± standard deviation and created by GraphPad Prism, version 7.0 (GraphPad Software Inc, San Diego, CA, USA).
RESULTS
Identification of the mushroom
DNA barcoding is a new emerging and reproducible molecular tool frequently used for the identification and taxonomic revision of biological specimens. Phallus sp. was confirmed by our previous publication and reference to the Genbank accession number was MT00752 [4]. Tricholoma sp. was verified through molecular identification method through DNA barcoding, the newly generated sequence of 661 bp long, was submitted to Genbank with accession number MT103100. The closest hit of MT103100 is Tricholoma sp. BAB-4947 (GeneBank accession number KR155037) with sequence identity 661/662 (99%); gaps 1/662 (0%).
Extraction yield
The extraction yield from both ME and EAE of five mushrooms (Lentinus sp., Astraeus hygrometricus, Serpula sp., Tricholoma sp., Phallus sp.) was depicted in Table 1. As the percentage of the yield was much higher in ME than EAE, it can be interpreted that there was more polar compound in mushrooms than non-polar compounds. There was no significant difference (P<0.05) in the extraction yield between the extracts of the two mushrooms
Table 1. Percentage yield of the methanolic and ethyl acetate extract of the collected mushrooms.
Antiproliferative assay
MTT result indicated that the two different extracts of the five different genera of mushrooms showed dose-dependent differential anti-proliferative efficacy against Jurkat cells (Figure 1 f, g) and the result was also found to be statistically significant (p<0.05). The efficacy of the methanolic extract (ME) of the five different mushrooms were evaluated and it has been found that Astraeus hygrometricus showed the highest anti-proliferative activity with IC50 value of 22.7 ±0.23 µg/mL followed by Serpula sp. 75.7 ±0.44 µg/mL, Phallus sp (Pha) 60.53±0.36 µg/mL, Tricholoma sp. (Tricho) 53.76±0.46 µg/mL, Lentinus sp. 58 ±0.13 µg/mL against the Jurkat cell line. On the other hand, we also checked the anti-proliferative activity of the ethyl acetate (EAE) of the same mushrooms (Figure 1g), in that case, all ethyl acetate extracts showed much less efficacy than ME extracts. So, the result indicated that among five different mushrooms, in all cases the ME extracts performed well than EAE extracts, in particular, the ME extract of Astraeus hygrometricus showed robust anti-proliferative activity on leukemic cells (Jurkat) in both cases with promising IC50 values than its counterpart EAE (IC50 68.9 ±0.33 µg/mL).
This result led us to carry the metabolic profiling of both methanolic and ethylacetate extracts, which was performed through GC-MS. In order to decipher the link between ROS generation and Cancer we checked the antioxidant potentiality of the methanolic extracts of all the mushrooms.
line (f,g).
DPPH radical scavenging assay
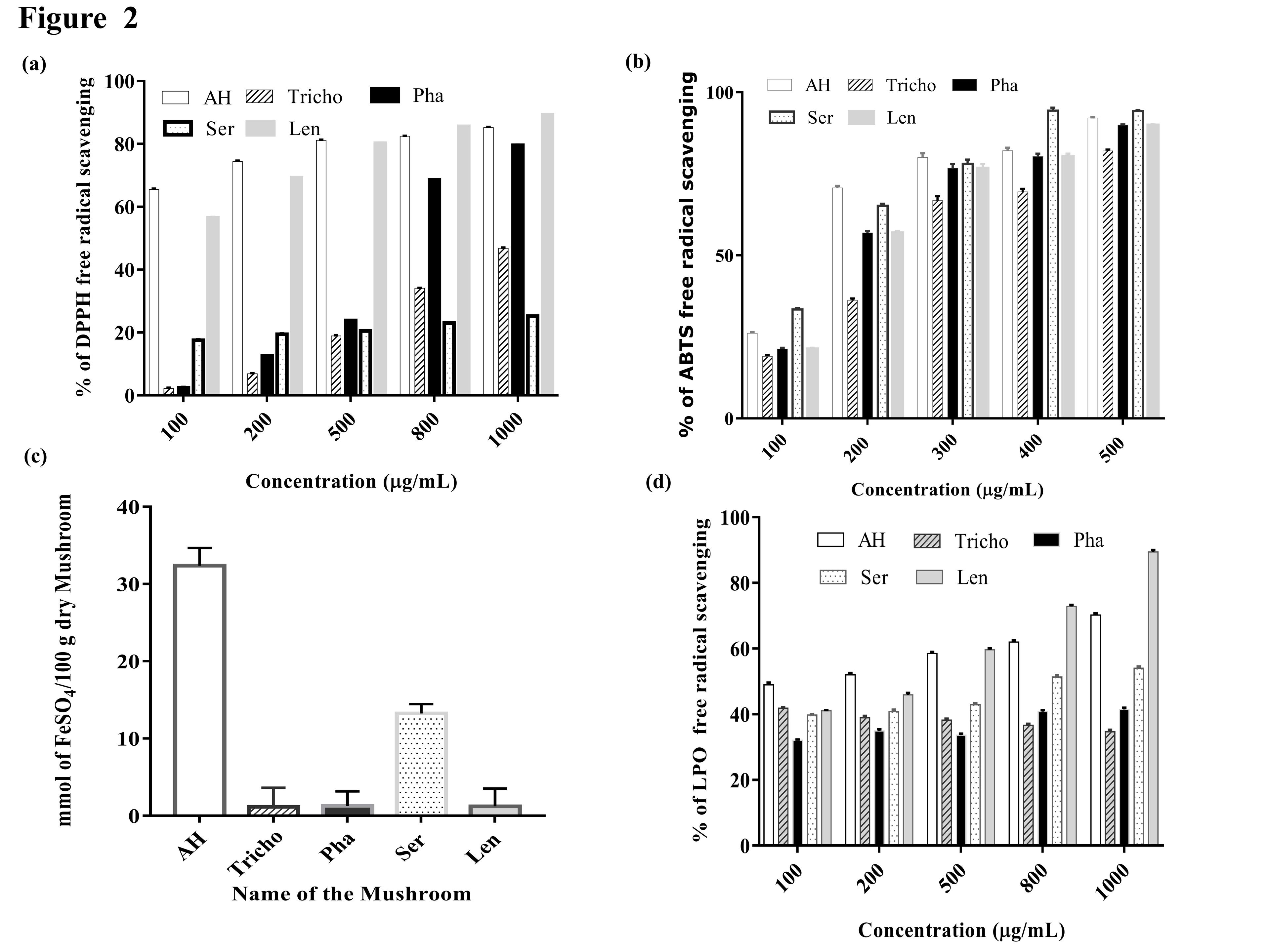
The result of the DPPH radical scavenging assay was represented (Figure 2a) in respect of its free radical scavenging potentiality which was carried out with five different concentrations (100, 200, 500, 800, and 1000 μg/mL) of methanolic extract (ME) of five mushrooms Lentinus sp. (Len), Astraeus hygrometricus (AH), Serpula sp. (Ser), Tricholomasp. (Tricho), Phallus sp. (Pha). Results indicated that the free radical scavenging potentiality of ME of five mushrooms occurred in a concentration-dependent manner. In respect of the IC50 value of the standard antioxidant molecule Ascorbic acid 9.9 ± 0.62 µg/mL, Astraeus hygrometricus (AH) and Phallus sp. (Pha) have shown promising results with IC50 value 76.9±0.16 µg/mL and 632.91±0.32 µg/mL respectively. The IC50 value of Lentinus sp. (Len) was less than 100 µg/mL and Tricholoma sp. (Trico) and Serpula sp. (Ser) was more than 1000 µg/mL.
ABTS•+ free radical scavenging assay
ABTS•+ free radical scavenging assay was performed with a concentration range (100-500 µg/mL) of the ME of five mushrooms (Figure 2b). The percentage of scavenging of ABTS•+ free radical proportionally increased with the increase of the concentration of mushroom extract in a linear manner. The maximum scavenging percentage of ME at the concentration of 500 µg/mL was ranging between 80% to 92%. The IC50 value of the five mushrooms Astraeus hygrometricus (AH), Phallus sp. (Pha), Lentinus sp. (Len), Tricholoma sp. (Tricho), and Serpula sp (Ser). AH, Tricho, Pha, Ser, Len against the ABTS•+ free radical was 142±0.66 µg/mL, 227.27 ±0.72 µg/mL, 178.5 ± 0.32 µg/mL, 153.8 ± 0.48 µg/mL and 178.5 ± 0.36 µg/mL respectively. This result indicated that antioxidant molecules present in ME extract of mushrooms were more effective on DPPH free radical than ABTS•+ free radical.
Lipid peroxidation assay
The Lipid Peroxidation inhibition (LPO) potentiality of any biological extract is a depiction of its antioxidant property. The LPO of ME of five mushrooms with a concentration range of (100-1000 μg/mL) is represented in (Figure 2d). The IC50 value of five mushrooms as follows Astraeus hygrometricus (AH) 192.2±0.25 µg/mL, Tricholoma sp. (Tricho) 1440±0.32 µg/mL, Phallus sp. (Pha) 1219 ± 0.65 µg/mL, Serpula sp. (Ser) 925 ±0 .65 µg/mL and Lentinus sp. (Len) 561.79 ± 0.48µg/mL with respect to the IC50 value of the standard antioxidant Ascorbic acid 29.63 ±0.22 µg/mL.
FRAP
The result of the FRAP assay the antioxidant activities of the mushrooms is represented in (Figure 2c) and expressed as the concentrations of antioxidants having a ferric reducing ability equivalent to that of 1 mM of FeSO4. The extracts showed a considerable antioxidant effect, Astraeus hygrometricus (AH) 32.37±2.31µM, Tricholoma sp. (Tricho) 1.15±2.50 µM, Phallus sp. (Pha) 1.27±1.9 µM, Serpula sp. (Ser) 13.23±1.23µM, Lentinus sp. (Len) 1.22 µM ±2.3 and equivalent of FeSO4/100 g dry mushroom. All the mushroom samples possessed antioxidant activity, while Astraeus hygrometricus (AH) and Serpula sp (Ser), showed significantly the highest results better than standard ascorbic acid, 2.017±2.9 µM of FeSO4/100 g equivalent.
Total phenol content
The total phenolic content of ME of five mushrooms was determined by the Folin-Ciocalteu method and is expressed as mg of gallic acid equivalents per gram of extract represented in (Figure 3e). The total phenol content of the mushroom extracts was calculated from the equation y=0.0002x-0.0125 (x = gallic acid concentration, y =absorbance of ME, obtained from the standard curve of different concentration of gallic acid, R2=0.9974) and the result indicated that the Astraeus hygrometricus, Tricholoma sp., Phallus sp., Serpula sp. and Lentinus sp., (AH, Tricho, Pha, Ser, and Len) contained 3.427 ± 0.33 mg, 1.237 ± 0.65 mg,1.367±0.9 mg,1.512 ±04 mg and 2.4±0.62 mg of GAE per g of mushroom dry weight.
Total flavonoid content
The total flavonoid content of ME of five mushrooms was determined by Aluminium chloride assay and the result is expressed as milligram of quercetin equivalents flavonoid present per gram of mushroom extract. Total flavonoid content of the mushroom extracts (Figure 3f) was calculated from the equation y=0.001x+0.002, R2=0.953 (x = quercetin concentration, y =absorbance of each mushroom methanolic extract)) and the result indicated that the Astraeus hygrometricus, Tricholoma sp., Phallus sp., Serpula sp., and Lentinus sp. (AH, Tricho, Pha, Ser, and Len) contained 74±0.4 mg, 49 ±0.33 mg, 31±0.90 mg, 14±0.65mg, and 53 ±0.25 mg of GAE per g of mushroom dry weight.
Flavonoid and phenolic compounds are considered as most impactful naturally occurring bioactive compounds which depict the antioxidant profile of any biological extracts [4]. The ME has shown higher flavonoid content than phenol content, so it may be predicted that the flavonoids are a major factor behind the significant antioxidant activity of the ME extracts.
Metabolomic profiling of methanolic extract of Astraeus hygrometricus
Among the five mushrooms, the Astraeus hygrometricus (AH) had shown antiproliferative activity against T cell leukemia cell line, Jurkat, and also exhibited moderate antioxidant activity, that provoked us to execute the GC-MS analysis for a comprehensive assessment of the metabolomic profile of both the ME and EAE extracts. On the other hand, in order to address the less anti-proliferative potentiality of ethyl acetate extract of AH we have compared the metabolomic profiling of both extracts. In this study, compounds obtained from the EAE and ME of AH were identified by comparing the mass spectra with pre-existing molecular library NIST version 2.2. The number of compounds present in each sample at different retention times and percentages of the area is depicted in (Tables 2 and 3). GC-MS analysis of the ethyl acetate (EAE) extract exhibited a total of 52 compounds (Table 2 and Figure 3 a,c) at different retention times. The total compounds in each extract are divided into major and minor compounds. Major compounds are those having a peak area percentage of more than two whereas minor compounds are less than two. These major and minor compounds of the two extracts are compared on the basis of peak area percentage (Figure 4). The major components of both ethyl acetate and methanolic extract are enlisted on the basis of the presence or absence of bioactivity of the compound in (Table 4). Total compounds were categorized into broad chemical groups, in the case of EAE, the compounds were categorized into twelve broad groups viz. alkane (7), alcohol(4), fatty acid(10), aromatic group of compounds (5), secondary metabolites (4), organic acid (11), polycyclic compound (4), amino acid (1), a heterocyclic group-containing compound (1), vitamin (2), polymer (1) and hormone (2). The percentage of the area of respective compounds in accordance with resulted GC peak is represented as a pie chart in (Figure 3b). On other hand, the methanolic extracts (ME) of AH showed a total number of 53 compounds (Figure 3d) along with retention time and area percentage are presented in (Table 3). The compounds are categorized in the same manner of EAE into eleven categories of broad chemical groups viz. monocyclic compound (4), alkane (8), alcohol (4), fatty acid (7), aromatic (6), secondary metabolites (4), organic acid (8), polycyclic (7), heterocyclic (2), vitamin (1) and sugar (2). The percentage of the area of respective compounds in accordance with resulted GC peak is represented as a pie chart in (Figure 3d).
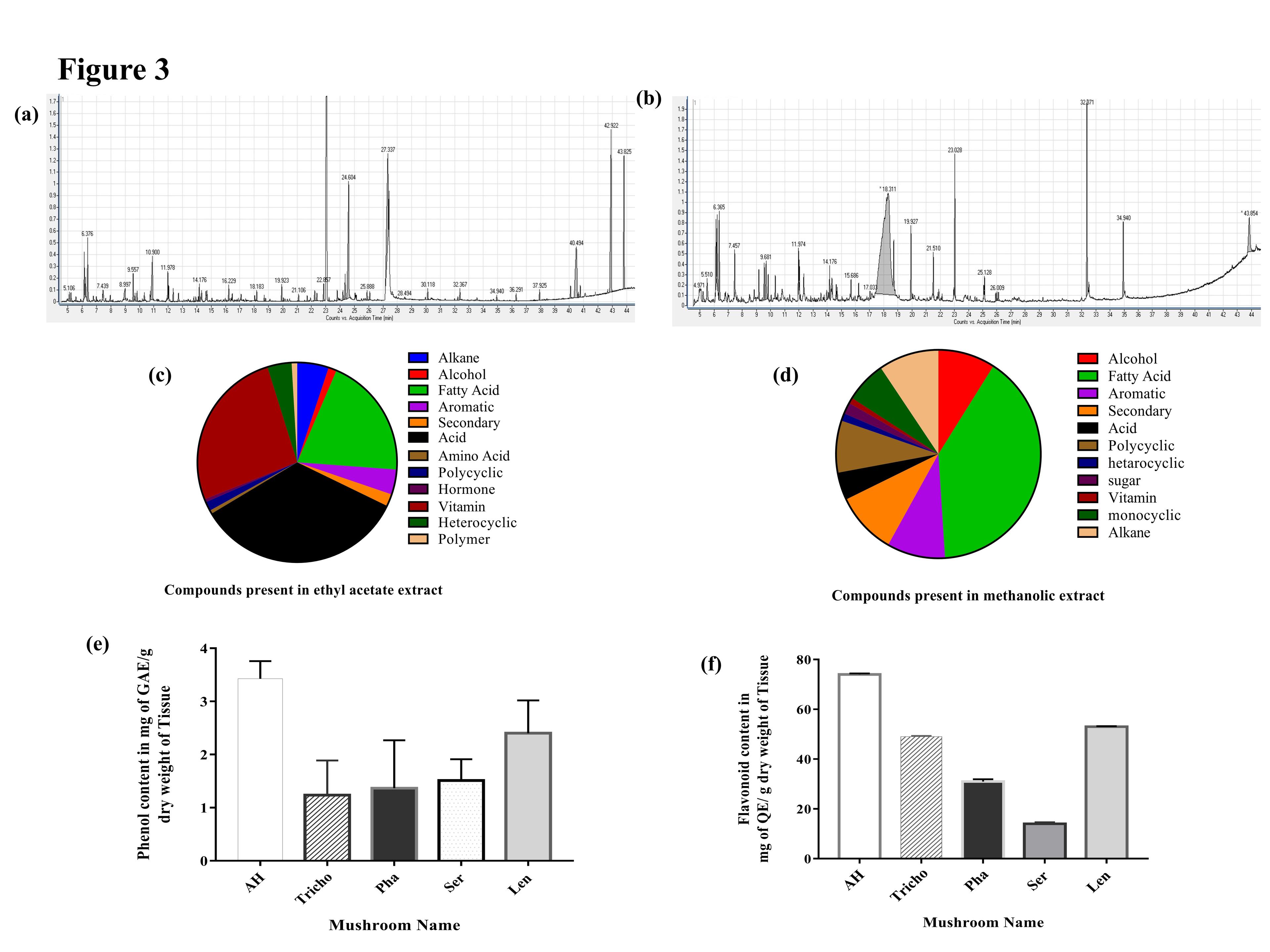
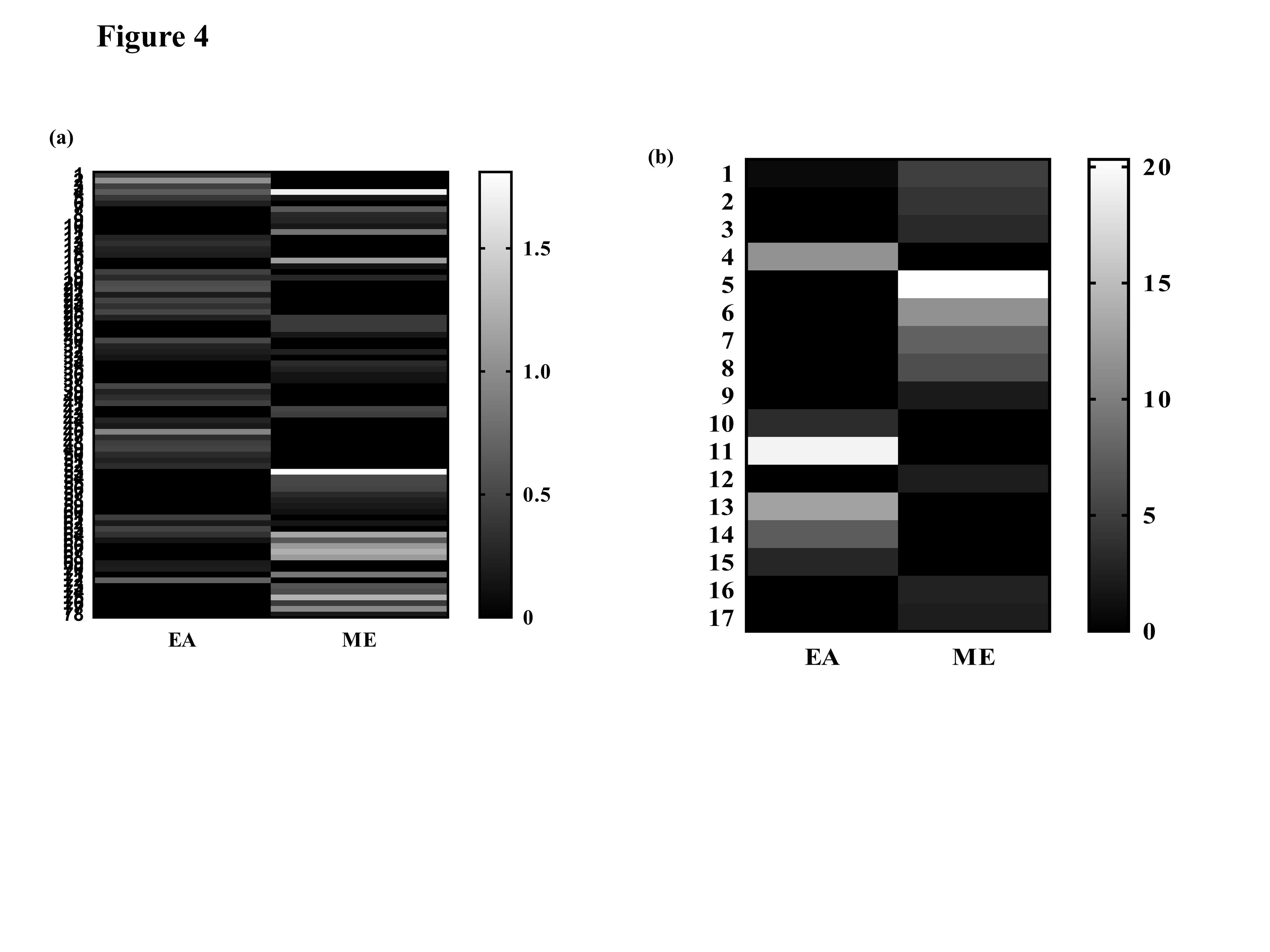
Table 2. List of compounds identified in ethyl acetate extract (EAE) of Astraeus hygrometricus in GCMS analysis.
Table 3. List of compounds identified in methanolic (ME) of Astraeus hygrometricus in GCMS analysis.
Table 4. Bioactivity of major compounds (area >2%) of Ethyl acetate and Methanolic extract of Astraeus hygrometricus.
DISCUSSION
Mushrooms are rich in secondary metabolites [23] and proved to be a source of high-potential anti-oxidant properties and antiproliferative activity [24]. In this study, the five mushrooms are collected, dried, and extracted by solvent percolation method. We have observed that the yielding percentage of methanolic extract of every five mushrooms are appreciable over their respective ethyl acetate extracts. The methanolic extract of Astraeus hygrometricus was selected on the basis of the screening studies between methanolic and ethyl acetate extracts, of five different wild and edible mushrooms viz. Lentinus sp. (Len), Astraeus hygrometricus (AH), Serpula sp. (Ser), Tricholoma sp. (Tricho), Phallus sp. (Pha) collected from different geographical regions of West Bengal. In all cases of initial screening, we have found that ME of Astraeus hygrometricus showed very promising antiproliferative activity by MTT assay that shows indirect cell-mediated cytotoxicity based on the hydrolysis of MTT by mitochondrial dehydrogenases of living cells with IC50 value22.7 ±0.23 µg/mL [25]. According to many reports Cancer and ROS management are directly related phenomena [26], so we have also correlated the antioxidant capacity of all the extracts with their antiproliferative efficacy, in this study, in all cases the methanolic extracts of the Astraeus hygrometricus have expressed much better antioxidant potentiality that fascinated us to array its metabolic profile. This initial data upholds the possibility of the upregulation of intermediate cancer signaling molecules such as PI3K/Akt, Wnt, Notch, mTOR, and JaK/Stat pathways, which can be an interesting experimental sphere in the future [27].
The volatile compounds in Astraeus sp. previously reported indicated that the volatiles found in this Gasteromycetidae were almost exclusively formed from fatty acid [28]. Astraeus hygrometricus from Southwest India reported to content tannin, phenols and flavonoids [29]. In the present study, the investigation of ethyl acetate and methanolic extracts from fruit body of Astraeus hygrometricus revealed the presence of various potent compounds, including Alkane, Alcohol, Fatty acid, aromatic, Secondary metabolites, Acid, Amino acid, Polycyclic, Hormone, vitamin, Polymer, heterocyclic. These bioactive phytoconstituents apart from fatty acid and secondary metabolites are reported as pioneers and could be responsible for the therapeutic ability of methanolic extracts of Astraeus hygrometricus. The analysis was carried out by gas chromatography-mass spectrometry (GC–MS), one of the most widely used techniques for separation of phytoconstituents. The GC–MS investigation of methanolic Astraeus hygrometricus extracts revealed the presence of 53 compounds, which could contribute to the medicinal properties of this mushroom species. As the methanolic extract contains many polar compounds, it can be interpreted that most of the bioactive molecules of the mushroom are polar. By comparing the profile of compounds found in both the methanolic and ethyl acetate extract of Astraeus hygrometricus, it can be inferred that some compounds such as limonene, tetradecane, and hexadecane are common in both extracts but present in different percentages. As it was previously reported C8 compounds, including 1-octen-3-ol, (E)-2-octen-1-ol, 1-octen-3-one, 3-octanone, and 1-octanol, were the main volatile compounds in fresh Astraeus hygrometricus. These C8 compounds are characteristic aroma-active compounds in mushrooms, and they contribute greatly to the flavor properties of Astraeus sp. whereas it was reported previously that Sulfur-containing compounds (dimethyl sulfone) in the pine mushroom [28] here in this study we had also obtained 2 Acetamide -2-deoxy-d-manno lactone, Hexanoic acid 2-ethyl, Acetophenone, Benzeneaceto nitrile 4 Hydroxy, 4-Hydroxy phenylacetamide as C8 compounds 3[2-Methyl propenyl], Octane-5-ethyl-2methyl, 2 Acetamide -2-deoxy-d-manno lactone, 1-Dodecanethiol, 1,8,15,22-Tricosatetrayne ,these are alkanes found only in methanolic extract. Eicosane, 7-hexyl-,1,6- Octadeine 2,7 dimethyl (terpenoid), 2(1H)Naphthalenone octahydro 4a Phenyl-trans, 1- Benzoyl-2-(pyrrolidinomethyl) piperine are secondary metabolites. Benzene-1 methyl-4-(1-methylethenyl), 4-Butoxyphenylacetonitrile, Morpholine 4[2-(4-tert-butyl benzene sulfoxyl cyclo hexyl], Benzene-n –butyl are monocyclic compounds. Above these compounds are found only in the methanolic extract of Astraeus hygrometricus. Literature survey reveals that most of the listed compounds have cytotoxic, anticancer, anti-inflammatory, anti-diabetic and anti-microbial activity except Dinoergosta-5-33-den-3ol, 1H-Indene, 1-ethylidene-, 4a,8a-Methaniminomethano naphthalene 9,11dione,10-phenyl, and Benzene 1methyl 3(1-methyl ethyl) [30]. Among the compounds of methanolic extract Benzene-1 methyl-4-(1-methyl phenyl), (1S,3S,4S,5R)-1-Isopropyl-4-methylbicyclo [3.1.0] hexan-3-ol, 14-Butyl Phenyl Acetonitrile H-Indene, 1-ethylidene, Hexadecanoic acid, 2-hydroxy-1-(hydroxymethyl) ethyl ester, Octadecanoic acid, 2,3-dihydroxy propyl ester, and Eicosane, 7-hexyl are not reported as a bioactive compound. Rest compounds are mainly anti-microbial inactivity. The predominant molecules that exclusively present on methanolic extracts are Cymene, 2 Pyrrolidinone 1- methyl or 1, 2-Benzenedicarboxylic acid, bis (2-methyl propyl) ester may have a role in inducing robust anti-proliferative activity against Jurkat cell line. Though there are two reports of anti-cancer activity by Astraeus hygrometricus on hepatocellular cell lines [31,32] and one tumor regression [33], we are the first to report its anti-leukemic potentiality. Thus, the methanolic extract of Astraeus hygrometricus can be selected as an alternative anti-leukemic drug and also as an antioxidant adjuvant.
CONCLUSION
Screening studies between methanolic and ethyl acetate extracts, of five different wild and edible mushrooms viz. Lentinus sp. (Len), Astraeus hygrometricus(AH), Serpula sp. (Ser), Tricholoma sp. (Tricho), Phallus sp. (Pha) collected from different geographical regions of West Bengal, showed the most robust anti-proliferative activity on Jurkat cell line by the methanolic extract of the Astraeus hygrometricus. As the methanolic extract showed much better anti-proliferative efficacy than its counterpart, the ethyl acetate extract, the antioxidant potentiality of the methanolic extracts of all five mushrooms was only evaluated. Among the five different mushrooms, the methanol extract of Astraeus hygrometricus exhibited appreciable anti-antioxidant efficacy. The anti-proliferative assessment study indicated that the IC50 value of the methanolic extract of Astraeus hygrometricus was 22.7 ± 0.23 µg/mL more efficient than ethyl acetate extract which is 68.9 ± 0.33 µg/mL.
The comprehensive comparison of the metabolomic profile of both extracts of Astraeus hygrometricus was executed through the GC-MS method. GC-MS chromatogram inferred that there were 52 and 53 compounds from ethyl acetate and methanolic extract respectively. These compounds were categorized according to the chemical family, the compounds detected from the ethyl acetate extract were divided into twelve groups whereas in methanolic extract the compounds were categorized into eleven chemical groups. The compounds of each extract were compared and inferred that the Naphthalene, 2-ethyl-, Naphthalene, 1,7-dimethyl-,Naphthalene, 1,4-dimethyl-,Biphenyl, Hexanoic acid 2-ethyl, Octadecanoic acid,2,3-dihydroxy propyl ester, Limonene, Tetradecane, and Hexadecane were common in both the extract but had different percentage area and retention time. On analyzing the bioactivity of the major compounds of each extract it was found that mainly the compounds were reported as antimicrobial, anti-cancer even anti-inflammatory.
In a nutshell, it can be concluded that the methanolic extract of Astraeus hygrometricusis composed of novel biomolecules that have the efficacy of inducing robust anti-leukemic activity. Further, the extract exhibits appreciable antioxidant activity, which may be a promising potent anti-cancer agent in the future.
ACKNOWLEDGMENT
The authors are indebted to the UGC-UPE and UGC-CAS program at the Department of Botany, the University of Calcutta for financial support. The project was funded by Department of Biotechnology, Government of West Bengal (2014-2017).
AUTHOR CONTRIBUTIONS
Prof. Santanu Paul designed the entire experiment. Dr. Krishnendu Acharya identified mushrooms. Ribhu Ray collected mushrooms and performed molecular identification. Amrita Pal collected and dried the mushrooms, prepared extracts, and performed all the laboratory tests. Prof. Santanu Paul and Amrita Pal analyzed data and wrote the manuscript. All authors approved the final manuscript.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci USA 1993; 90:7915–22.
- [2]Tripoli E, Giammanco M, Tabacchi G, Di Majo D, Giammanco S, La Guardia M. The phenolic compounds of olive oil: structure, biological activity and beneficial effects on human health. Nutr Res Rev 2005; 18:98–112.
- [3]Barros L, Ferreira MJ, Queiros B, Ferreira I, Baptista P. Total phenols, ascorbic acid, β-carotene and lycopene in Portuguese wild edible mushrooms and their antioxidant activities. Food Chem 2007; 103:413–9.
- [4]Ray R, Pal A, Paul S. Assessment of the Impact of Wild Stinkhorn Mushroom Extracts on Different Cancer Cell Proliferation and Study of Primary Metabolites. Pharmacogn J 2020; 12.
- [5]Jayalakshmi CP, Sharma JD. Effect of butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT) on rat erythrocytes. Environ Res 1986; 41:235–8.
- [6]Decker E. Phenolics: Prooxidants or Antioxidants? Nutr Rev 1997; 55:396–8.
- [7]Ferreira ICFR, Barros L, Abreu RM V. Antioxidants in wild mushrooms. Curr Med Chem 2009; 16:1543–60..
- [8]Pal A, Chouni A, Das A, Ray R, Paul S. Evaluation of Anti-proliferative Potential and Antioxidant Activity of a Wild Edible Mushroom Macrocybe crassa (Sacc.) Pegler and Lodge. Pharmacogn J 2019; 11:1504–10.
- [9]Sarikurkcu C, Tepe B, Yamac M. Evaluation of the antioxidant activity of four edible mushrooms from the Central Anatolia, Eskisehir – Turkey: Lactarius deterrimus, Suillus collitinus, Boletus edulis, Xerocomus chrysenteron. Bioresour Technol 2008; 99:6651–5.
- [10]Synytsya A, Míčková K, Synytsya A, Jablonský I, Spěváček J, Erban V, et al. Glucans from fruit bodies of cultivated mushrooms Pleurotus ostreatus and Pleurotus eryngii: Structure and potential prebiotic activity. Carbohydr Polym 2009; 76:548–56.
- [11]Patel S, Goyal A. Recent developments in mushrooms as anti-cancer therapeutics: a review. 3 Biotech 2012; 2:1–15.
- [12]Matuszewska A, Jaszek M, Stefaniuk D, Ciszewski T, Matuszewski Ł. Anticancer, antioxidant, and antibacterial activities of low molecular weight bioactive subfractions isolated from cultures of wood degrading fungus Cerrena unicolor. PLoS One 2018; 13:e0197044.
- [13]Zhang M, Zhang Y, Zhang L, Tian Q. Mushroom polysaccharide lentinan for treating different types of cancers: A review of 12 years clinical studies in China. Prog Mol Biol Transl Sci 2019; 163:297–328.
- [14]Chouni A, Pal A, Gopal PK, Paul S. GC-MS Analysis and Screening of Anti-Proliferative Potential of Methanolic Extract of Garcinia cowa on Different Cancer Cell Lines. Pharmacogn J 2020; 13:347–61.
- [15]White TJ, Bruns T, Lee S, Taylor J. Amplification and direct sequencing of fungal ribosomal rna genes for phylogenetics. Pcr Protoc 1990:315–22.
- [16]Chakraborty T, Basak AP, Mridha A, Gopal PK, Paul S. Anti-cancer and anti-oxidant potential of Indian carpet weed Glinus oppositifolius (L.) Aug. DC J Pharmacogn Phytochem 2017; 6:464–8.
- [17]Gopal P, Paul M, Paul S. Curcumin Induces Caspase Mediated Apoptosis in JURKAT Cells by Disrupting the Redox Balance. Asian Pac J Cancer Prev 2014; 15:93–100.
- [18]Hajra D, Paul S. Study of glucose uptake enhancing potential of fenugreek (Trigonella foenum graecum) leaves extract on 3T3 L1 cells line and evaluation of its antioxidant potential. Pharmacognosy Res 2018; 10:347–53.
- [19]Tokur B, Korkmaz K, Ayas D. Comparison of Two Thiobarbituric Acid (TBA) Method for Monitoring Lipid Oxidation in Fish. U J Fish Aquat Sci Cilt Sayı/Issue 2006; 23:3–4.
- [20]Benzie, F IF, Strain JJ. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of ‘. Ferric Reducing Abil Plasma (FRAP) as a Meas ‘“Antioxid Power”’ FRAP Assay 1996; 76:70–6.
- [21]Mridha A, Nandi C, Pal R, Paul S. Studies on few fresh water green algal species reveals Spirogyra triplicata as the repository of high phenolic and flavonoid content exhibiting enhanced anti-oxidant property. J Pharmacogn Phytochem 2017; 6:1291–7.
- [22]Kamtekar S, Keer V, Patil V. Estimation of phenolic content, flavonoid content, antioxidant and alpha amylase inhibitory activity of marketed polyherbal formulation. J Appl Pharm Sci 2014; 4:61–5.
- [23]Wandati T, Kenji G, Onguso J. Phytochemicals in Edible Wild Mushrooms From Selected Areas in Kenya. J Food Res 2013;2.
- [24]Sohretoglu D, Huang S. Ganoderma lucidum Polysaccharides as An Anti-cancer Agent. Anticancer Agents Med Chem 2018; 18:667–74.
- [25]Sun Y, Yin T, Chen X-H, Zhang G, Curtis RB, Lu Z-H, et al. In vitro antitumor activity and structure characterization of ethanol extracts from wild and cultivated Chaga medicinal mushroom, Inonotus obliquus (Pers.:Fr.) Pilát (Aphyllophoromycetideae). Int J Med Mushrooms 2011; 13:121–30.
- [26]Srinivas US, Tan BWQ, Vellayappan BA, Jeyasekharan AD. ROS and the DNA damage response in cancer. Redox Biol 2019; 25:101084.
- [27]Gopal P, Paul M, Paul S. Role of different aberrant cell signalling pathways prevalent in acute lymphoblastic leukemia. Biologia (Bratisl) 2014; 69:1097–107.
- [28]Kakumyan P, Matsui K. Characterization of volatile compounds in Astraeus spp. Biosci Biotechnol Biochem 2009;73:2742–5. https://doi.org/10.1271/bbb.90282.
- [29]Pavithra M, Sridhar KR, Greeshma AA, Tomita-Yokotani K. Bioactive potential of the wild mushroom Astraeus hygrometricus in South-west India. Mycology 2016; 7:191–202.
- [30]Chowdhury P, Paul S. The Potential Role of Mushrooms in The Prevention and Treatment of Diabetes: A Review. J Biol Act Prod from Nat 2020; 10:429–54.
- [31]Dasgupta A, Dey D, Ghosh D, Lai TK, Bhuvanesh N, Dolui S, et al. Astrakurkurone, a sesquiterpenoid from wild edible mushroom, targets liver cancer cells by modulating Bcl-2 family proteins. IUBMB Life 2019; 71:992–1002.
- [32]Nandi S, Chandra S, Sikder R, Bhattacharya S, Ahir M, Biswal D, et al. Characterization and Inception of a Triterpenoid Astrakurkurol, as a Cytotoxic Molecule on Human Hepatocellular Carcinoma Cells, Hep3B. J Agric Food Chem 2019; 67:7660–73.
- [33]Mallick SK, Maiti S, Bhutia SK, Maiti TK. Antitumor properties of a heteroglucan isolated from Astraeus hygrometricus on Dalton’s lymphoma bearing mouse. Food Chem Toxicol 2010; 48:2115–21.
- [34]Al-Marzoqi AH, Hadi MY, Hameed IH. Determination of metabolites products by Cassia angustifolia and evaluate antimicobial activity. J Pharmacogn Phyther 2016; 8:25–48.
- [35]Custódio DL, Burgo RP, Moriel B, Barbosa A de M, Rezende MI, Daniel JF de S, et al. Antimicrobial activity of essential oils from Pimenta pseudocaryophyllus and Tynanthus micranthus. Brazilian Arch Biol Technol 2010; 53:1363–9.
- [36]Talita P de SF, Gil R dos S, Ilsamar MS, Sergio DA, Tarso da CA, Chrystian de AS, et al. Secondary metabolites from endophytic fungus from Lippia sidoides Cham. J Med Plants Res 2017; 11:296–306.
- [37]Marchese A, Arciola CR, Barbieri R, Silva AS, Nabavi SF, Tsetegho Sokeng AJ, et al. Update on Monoterpenes as Antimicrobial Agents: A Particular Focus on p-Cymene. Mater (Basel, Switzerland) 2017; 10.
- [38]Lis-Balchin M, Hart S. A preliminary study of the effect of essential oils on skeletal and smooth muscle in vitro. J Ethnopharmacol 1997; 58:183–7.
- [39]Miller JA, Lang JE, Ley M, Nagle R, Hsu C-H, Thompson PA, et al. Human breast tissue disposition and bioactivity of limonene in women with early-stage breast cancer. Cancer Prev Res (Phila) 2013; 6:577–84.
- [40]Phaechamud T, Mahadlek J, Charoenteeraboon J, Choopun S. Characterization and Antimicrobial Activity of N-Methyl-2-pyrrolidone-loaded Ethylene Oxide-Propylene Oxide Block Copolymer Thermosensitive Gel. Indian J Pharm Sci 2012; 74:498–504.
- [41]Hosseinzadeh Z, Ramazani A, Hosseinzadeh K, Razzaghi-Asl N. An Overview on Chemistry and Biological Importance of Pyrrolidinone. Curr Org Synth 2017; 14.
- [42]Paritala V, Chiruvella KK, Thammineni C, Ghanta RG, Mohammed A. Phytochemicals and antimicrobial potentials of mahogany family. Rev Bras Farmacogn 2015; 25:61–83.
- [43]Banakar P, Jayaraj M. Pharmacognosy, Phytochemistry and GC-MS Analysis of Ethanolic Stem Extract of Waltheria indica L. – A Potent Medicinal Plant. J Biol Act Prod from Nat 2017; 7:369–78.
- [44]Krishnan K, Mani A, Jasmine S. Cytotoxic Activity of Bioactive Compound 1, 2- Benzene Dicarboxylic Acid, Mono 2- Ethylhexyl Ester Extracted from a Marine Derived Streptomyces sp. VITSJK8. Int J Mol Cell Med 2014; 3:246–54.
- [45]Forte G, Fortuna CG, Salerno L, Modica MN, Siracusa MA, Cardile V, et al. Antitumor properties of substituted (αE)-α-(1H-indol-3-ylmethylene)benzeneacetic acids or amides. Bioorg Med Chem 2013; 21:5233–45.
- [46]Astudillo AM, Meana C, Guijas C, Pereira L, Lebrero P, Balboa MA, et al. Occurrence and biological activity of palmitoleic acid isomers in phagocytic cells. J Lipid Res 2018; 59:237–49.
- [47]Elagbar ZA, Naik RR, Shakya AK, Bardaweel SK. Fatty Acids Analysis, Antioxidant and Biological Activity of Fixed Oil of Annona muricata L. Seeds. J Chem 2016; 2016:6948098.
- [48]Johannes E, Litaay M. The Bioactivity of Hexadecanoic Acid Compound Isolated from Hydroid Aglaophenia Cupressin A Lamoureoux As Antibacterial Agent Against Salmonella typhi. Int J Biol Med Res 2016; 6:5469–72.
- [49]Anagement M, Capparaceae CE, Ojinnaka CM, Nwachukwu K The Chemical Constituents and Bioactivity of the seed (Fruit) extracts of Buchholzia JASEM 2015.
- [50]Guerrero RV, Vargas RA, Petricevich VL. Chemical Compounds and Biological Activity of an Extract From Bougainvillea X Buttiana (Var. Rose) Holttum and Standl. Int J Pharm Pharm Sci 2017; 9:42.
- [51]Kalaras MD, Beelman RB, Elias RJ. Effects of postharvest pulsed UV light treatment of white button mushrooms (Agaricus bisporus) on vitamin D2 content and quality attributes. J Agric Food Chem 2012; 60:220–5.
- [52]Oliveira L de M, Teixeira FME, Sato MN. Impact of Retinoic Acid on Immune Cells and Inflammatory Diseases. Mediators Inflamm 2018; 2018:3067126.