Evaluation of selective mitis salivarius agar for the isolation of Streptococcus mutans and its resistance pattern in Bangladesh
Abstract
Since Streptococcus mutans appears to be the most common cause of dental caries, appropriate laboratory media is necessary for the proper detection and management of this bacterium. The aim of this study was to evaluate the mitis salivarius agar (MSA) compared to conventional blood agar media (BAM) for detection of bacterium. This study was conducted in the Department of Microbiology of the Rajshahi Medical College, Rajshahi, Bangladesh from April 2017 to December 2017. The sample, dental swab was taken from 200 children, aged between 6-18 years who underwent dental caries and residing in Rajshahi district. All specimens were cultured to identify and compare the morphologic characteristics of its colonies both in MSA and BAM. In this study, antimicrobial susceptibility testing was also performed. This prospective observational study was conducted through regular and continuous monitoring of the results. Out of 200 specimens, the growth rate was 82%. Higher growth was observed in MSA (39.5%) than BAM (24.1%). Of them, we found 53.1% multi-drug resistant mutans. The most resistance was to Penicillin G (100%) followed by Azithromycin (95.3%). The study findings would help to increase the detection of mutans and its pattern for proper treatment towards the improvement of children’s dental health in Bangladesh.
INTRODUCTION
Dental caries is one of the most significant and common infectious diseases in the human oral cavity with bacterial metabolic processes that cause damage in hard tissue of the tooth structure [1]. It is considered as a major public health problem globally due to its high prevalence and significant social impact. Dental caries has plagued human since the dawn of civilization and still constitutes a major public health concern at global scale [2,3]. This is mostly due to colonization of Streptococcus mutans as a causative agent for dental caries [4]. The bacteria S. mutans has the capacity to metabolize the fermentable carbohydrate into organic acid which causes fall in pH to increase the risk of enamel solubility [5]. The untreated dental caries can lead to pain, tooth loss, infection, inflammation, and death in severe cases. The presence of bacterial flora may be seen in different area of teeth for ample dentin enamel junction beneath white spot lesion, gaps between cavity walls and restoration, areas of penetrated caries, fissures, and other adjacent areas [6]. The cell wall biosynthesis inhibitor is the effective antibiotic against the bacteria or mutans. The Streptococcus mutans is gram positive coccus which is usually susceptible to cell wall biosynthesis thus inhibiting antibiotics [7]. Unfortunately, further difficulties in treating dental caries with conventional antibiotics is observed over time. The recent study on cariogenic S. mutans showed a gradual increase in resistant pattern of bacteria to Penicillin, Erythromycin, Ciprofloxacin, and other antibiotics [8]. Though antimicrobial resistance is not new in the world, the frequencies, patterns, and distribution of resistant bacteria vary with geographic locations [9]. The breadth of resistance in single organism is unprecedented and mounting particularly in developing countries like Bangladesh [10]. The mitis salivarius agar (MSA) is the most potential media to detect S. mutans [11]. Though the conventional blood agar media (BAM) is commonly being used in most of the laboratories in Bangladesh to identify mutans, MSA would be the potential alternative over BAM. Hence, the researcher felt the necessity to conduct the study to compare the performance of MSA with BAM for isolation of mutans.
Furthermore, to the best of our knowledge, there is no such specific research in Rajshahi district which created more importance to conduct the research. Therefore, the aim of this study was to evaluate MSA compared to BAM for detection of bacteria and its resistance pattern to reduce the health burden among school children in Bangladesh.
MATERIAL AND METHODS
Sampling method
A prospective observational cohort study was conducted in the Department of Microbiology of Rajshahi Medical College, Rajshahi, Bangladesh from April 2017 to December 2017. The outpatient department (OPD) of Rajshahi Medical College Hospital was the sample collection source. The multi-stage random sampling technique was used in this study. In first stage, 600 children with dental caries were randomly selected. In second stage, caries with high severity, and children outside the district were excluded. In third stage, children with unwillingness to participation were excluded. Finally, children aged from 6 years to 18 years were selected to take dental swab for isolation of organisms. Age, address, and gender records were taken during swab collection. The purpose of the study was shared with the parents of the selected children, and verbal consent was also taken from the children as well as the parents.
The S. mutans were identified by culture on selective (MSA) and non-selective media (BAM) and performed biochemical test. All laboratory measures including temperature, infection control measures, prevention of cross-contamination, use of biosafety cabinet and other procedures were strictly followed. The maintenance of environment and media, monitoring of growth using standard interval was recorded regularly and reported timely. Study approval was taken from respective authority of Rajshahi Medical College and ethical approval was also taken from ethical committee of Rajshahi Medical College, Rajshahi. Study approval was taken from respective authority of Rajshahi Medical College and ethical approval was also taken from ethical review committee of Rajshahi Medical College, Rajshahi (RMC/ERC/2016-2017/53).
Whereas, 600 participants randomly selected for swab collection>240 existed following exclusion criteria>200 participants who were willing to participate.
Collection of dental swab specimen
The specimen as samples were taken from the patients who exhibited the sign and symptoms of plaque and dental caries. Participating children were instructed for not to brush teeth, not to eat or drink anything for at least two hours until swab collection. Two sterilized swab sticks were used to collect swab from caries site, one (a) for staining & microscopy and another (b) for culture and sensitivity testing.
Culture and identification
The collected specimens were inoculated in both selective mitis salivarius agar and blood agar media. The inoculated plates were incubated aerobically at 370C for 24 hours to see the growth. The predominant and morphologically different colonies from mitis salivarius agar were sub-cultured, using standard streak plate technique on nutrient agar media for pure culture. Further identification of isolated bacterial strains was facilitated by hemolysis on sheep blood agar plate, distinctive cell shape on light microscopy and biochemical tests including catalase test, coagulase test, sugar fermentation tests. Isolation and identification of bacteria was done following standard procedure [12].
Antimicrobial susceptibility test
Antimicrobial susceptibility test was performed by modified Kirby-Bauer disc diffusion method using Mueller-Hinton agar media and commercially available antimicrobial discs. The discs were selected as per clinical and laboratory standards institute (CLSI) guidelines, 2017. The susceptibility tests were on Amoxicillin (30 mcg), Azithromycin (30 mcg), Erythromycin (15mcg), Ciprofloxacin (05 mcg), Livofloxacin (05 mcg), Pencillin-G (10 units), and Vancomycin (30 mcg) were performed in the laboratory. Having growth, additional time of 24 hours was needed to see the resistance.
Preservation of the antibiotic discs
Antibiotic discs are available in local market. After purchasing and checking, antimicrobial discs were kept at 20-80C temperature. Prior to use, the container was allowed to warm up slowly at room temperature to minimize condensation of moisture following the microbiological standard [12].
Quality control
Before disc diffusion with the clinical isolates, a representative of each batch of the discs was standardized by testing against reference strains of Staphylococcus aureus ATCC No.25923. Following the appropriate quality control, the colonies of S. mutans showed the expected raised, convex, opaque, pale blue “granular frosted glass” appearance in selective mitis salivarius agar. On 5% sheep blood agar, colonies were small, and grey, white appearance was also observed with alpha hemolysis.
Biochemical characteristics
S. mutans showed positive reaction to Voges-proskaur (VP) test, glucose, sorbitol, and mannitol with the production of acid with optochin resistance. To analyze the biochemical characteristics, catalase test, VP, sugar fermentation test and optochin sensitivity test were done. The negative catalase test indicates hydrogen per-oxide was stable. Subsequently, for sugar fermentation test, sodium hydroxide was added in 0.5% aqueous acid fuchsin to form Andrade’s indicator to turn the color into yellow. Then, peptone and Andrade’s indicators was dissolved in 1 liter water where 20 g of sugar was also added in sugar fermentation test. From the total solution, 3 ml of solution was distributed in each standard test tube that kept in an autoclave at 1210 centigrade for 15 minutes to avoid growth of other organisms. Then S. mutans was inoculated in these sugar sets. Again, incubation was performed at 370C for 24 hours to see the changes of color. Pink color indicates a positive result. Consequently, the other tests were done following appropriate procedure.
Statistical analysis
All relevant information and laboratory findings were recorded in a pre-designed Excel data sheet and performed a gross analysis using statistical package for social science (SPSS) version 23. The Pearson Chi-square test was done to know the association between gender and microbial growth. The value of P <0.05 was considered as statistically significant. Study results were presented in the form of tables, and charts.
RESULTS
Respondent’s characteristics and growth pattern
Out of 200 respondents, 106 was boy and 94 was girl with dental caries. Of them, more growths were observed among girls (98.9%) than boys (65.1%) which was highly significant (P<0.001). Surprisingly, in spite of presence of caries, no growth was observed among 36% cases with most predominant among boys (Table 1).
Table 1. Distribution of bacterial growth.
Growth rate according to types of bacteria
Among the total positive isolate of bacterial colony (162), the Streptococcus mutans (S. mutans) was predominant (32.0%) in dental caries followed Staphylococcus aureus (23.5%) Streptococcus mitis (16.0%) and Streptococcus salivarius (9.5%) respectively in MSA (Table 2).
Table 2. The rate of bacterial growth detected in MSA.
Media performance on growth
However, the detection rate of S. mutans was lower (24.0%) in conventional blood agar media (BAM) than MSA (Figure 1). The subsequent bacterial growth was also lower in BAM. On the other hand, the Gram positive mutans and other bacteria have several species which was not separately counted in this study.
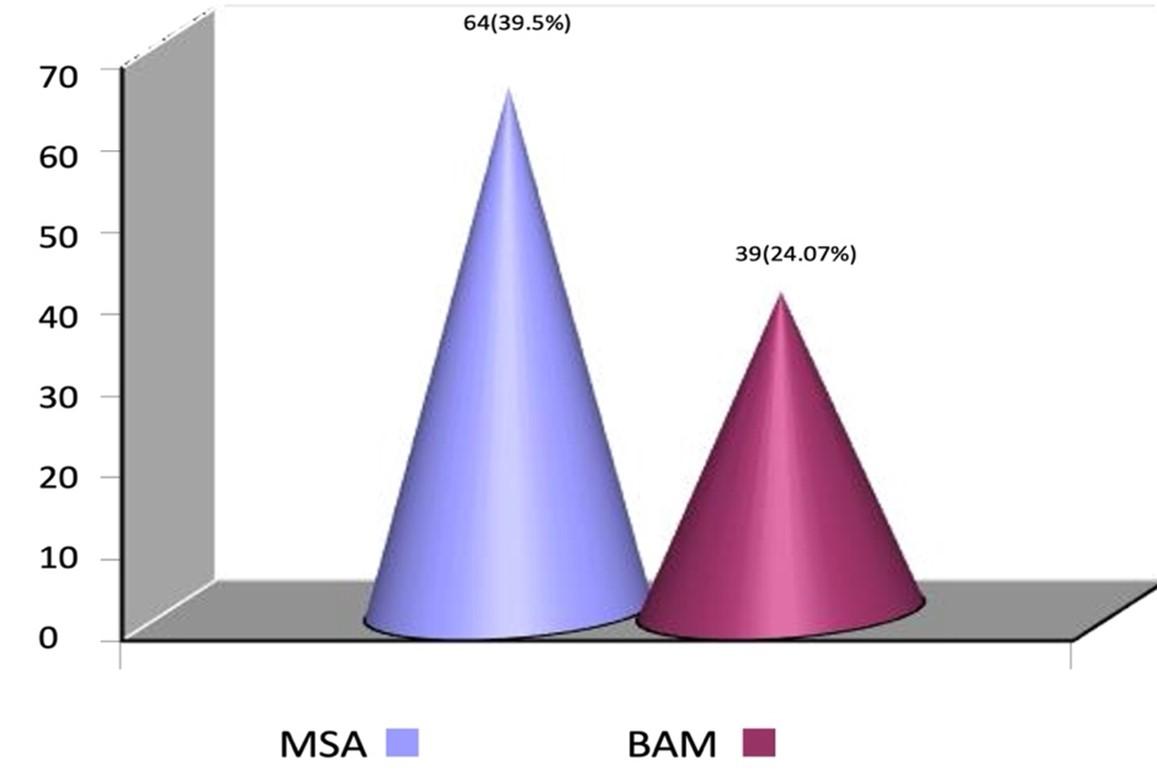
Drug resistance pattern of S. mutans
During drug susceptibility testing, S. mutans showed 100% resistant to penicillin G. followed by Azithromycin (95.31%), Ciprofloxacin (87.5%), Amoxicillin (78.25%). Alternatively, the S. mutans showed highest sensitivity to Vancomycin (100%) followed by Livofloxacin (95.31%); and subsequently, Erythromycin (76.56%) (Table 3).
Table 3. Antibiotic resistance and susceptibility pattern of S. mutans.
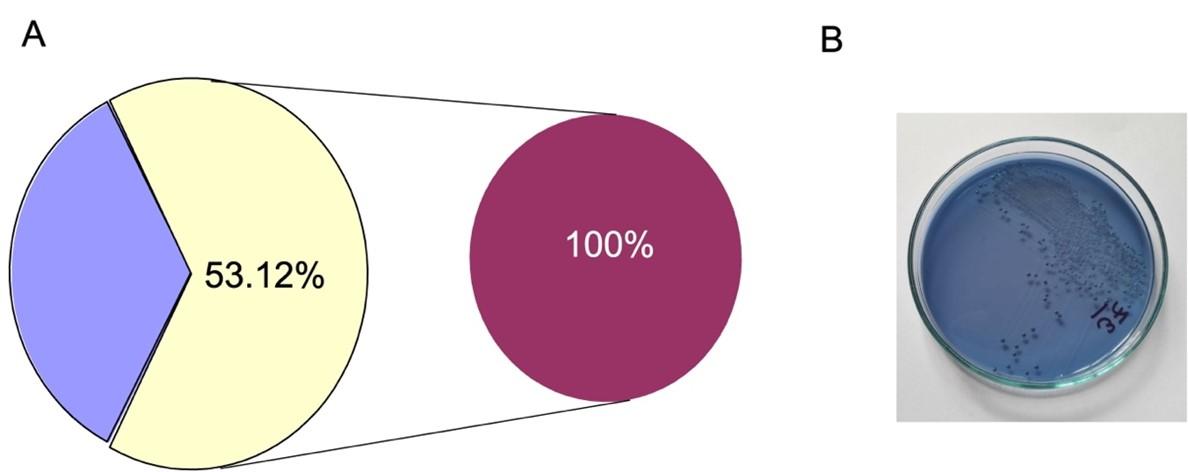
Multi-drug resistance rate of S. mutans
However, along with single drug resistance, the multi-drug resistance was also performed. In our study, out of 60 isolates, 34 (53.12%) were multi-drug resistance which is mostly alarming (Figure 2).
DISCUSSION
Health is a common theme in most culture and is a fundamental human right without distinction of race, religion, and political belief, economic and social condition. Dental caries, because of its ubiquitous nature, remains one of the most prevalent afflictions of mankind [13]. In our study, out of 200 dental swabs, 162 (81%) yielded culture positive. Among them S. mutans was the predominant 64 (39.50%) bacterial isolates. S. mutans is a potent cariogenic bacterium. Caries occurs when a susceptible tooth surface is colonized with cariogenic bacteria. S. mutans was collected around the tooth and gums and dietary source of sucrose or refined sugar was converted to lactic acid from fermentation of carbohydrates. If it left in contact with the tooth, this acid dissolves the hydroxyapetite crystal structure of tooth which causes caries [14]. The finding concerning the high frequency of S. mutans greatly coincided with studies in Nepal, Iraq and in Egypt [9,15,16]. For comparative growth study, we used selective mitis salivarius agar (MSA) and conventional blood agar (BA) media. Our result revealed that 64 (39.5%) isolates out of total 162 isolates, were recovered from MSA . This finding consistent with other studies and their observations were 38%, and 40% respectively [7,16]. The principal identification of S. mutans was usually made from characteristic morphology of its colonies on 5% sucrose containing MSA agar media. Isolation of S. mutans in BA media were 39 (24.07%), out of total 162 isolates. This finding was almost similar with some studies conducted in Bangladesh and in Saudi Arab [17,18]. Their observations were 28.80%, and 22.98% in BA media. These studies showed that the mitis salivarius agar media recovered a higher number of S. mutans than BA. As several studies have shown similar results indicates that mitis salivarius agar media is an excellent, highly effective, and most selective media for the isolation of S. mutans.
The antimicrobial agents are using widely for treatment and prevention of complication of dental caries. But now, use of antibiotic has become very much crucial due to spread of antibiotic resistance [19]. Regarding antibiogram, we found more than 53% multidrug resistant mutans. This study was nearly similar with the study in India where multidrug resistance was 23 (57.7%) [20]. These findings should draw attention of global policy makers towards appropriate initiative as soon as possible. First line drugs for S. mutans as recommended by Tierney and Colleagues include Penicillin [18]. In this study, mutans had not shown high sensitivity to Amoxicillin which represents the Penicillin depicts not to use Amoxicillin in dental caries. In the present study isolates of S. mutans showed 78.12% resistant to Amoxicillin which was comparatively lower resistant than other antibiotics. The other studies in Nepal and Bangladesh also found similar results [8,17]. However, antibiotics, particularly Amoxicillin is being prescribed frequently by non-graduate doctors such medicine shopkeepers, village doctors. Even many Bangladeshi people are taking Amoxicillin without consultation with graduate physicians thus creating resistance.
In our study, S. mutans showed complete resistance to Penicillin G. This study was consistent with several studies in different countries [5,14,18,20]. But the reverse finding was also observed in the study conducted by El-Sherbiny in Egypt population where S. mutans were most sensitive to Penicillin G [16]. We found, the St. mutans was 87.50% resistant to Ciprofloxacin which was nearly similar to other study where S. mutans were 80% resistant to Ciprofloxacin. [16]. However, alternative results were also observed in other studies where mutans were most sensitive to Ciprofloxacin. This may be due to geographical distribution [9,12,14]. The S. mutans were highly sensitive to Vancomycin (100%) which is consistent with other study conducted in Egypt [16]. Alternatively, the resistant result was also observed in other study [7]. Subsequently, the S. mutans were 95.31% sensitive to Levofloxacin which was similar to the study conducted in Bangladesh. We also observed the resistance pattern against the antibiotic Azithromycin and the S. mutans showed 95.31% resistance towards Azithromycin which was similar to other studies conducted in India [7,20]. They revealed the emergence of complete or 100% resistant to Azithromycin with all the isolates of S. mutans. Erythromycin is used as a substitute of Penicillin, especially in a person having Penicillin allergy. Our study showed 76.56% sensitive to Erythromycin. Several studies were conducted in different settings and found almost similar results [14,16,21,22]. In this study, a substantial resistance was observed to a number of commonly used antibiotics. This may be due to inappropriate use of antibiotics which is rampant in Bangladesh. Hence, it is important to periodically monitor the antibiotic resistance pattern in different regions. Selective number of antibiotics were chosen for this study to do antimicrobial sensitivity tests to encourage minimum use of antibiotics in dentistry and to get maximum efficacy of antibiotic. However, we carefully observed, reviewed, and recorded each of the findings without being biased. Our measurement and performance of selective media would help detecting dental caries towards prevention. Along with global awareness on anti-microbial resistance (AMR), our findings would also help realize the scenario among young children. In our study, we did not take socio-economic and socio-demographic information to identify the food habit as source of dental caries. The parent’s education and children life-style data which was important. Though we carefully observed the growth and resistance pattern, it was not declared as an experimental study due to improper knowledge on prior approval from ICMJE.
CONCLUSION
In this current study, we observed that mitis salivarius agar is more efficient, less laborious, and most selective media for isolation of Streptococcus mutans as well as there is an emergence of multidrug resistance. It is determined that the prevalence and severity of dental caries are greater in the current study, with more decaying teeth than filled teeth. The findings of this baseline survey revealed that dental caries is a serious public health concern, and there is a dearth of preventative and restorative dental care facilities with appropriate laboratory media, as well as public awareness in this region. The result of this process makes it important to implement primary prevention and greater restorative treatment to reduce caries prevalence and to preserve caries-free childhood. Research on the oral diseases, such as dental caries, has opened new opportunities for establishing a balance between diet and oral health. The research is important, as the available databases have only a few clinical studies with strong scientific evidence proving the effectiveness of this media.
ACKNOWLEDGEMENTS
The authors gratefully acknowledge the technical support provided by department of Microbiology of Rajshahi Medical College and Dental Unit of Rajshahi, Medical College, Rajshahi. We would like to thank Md. Mominul Islam, Department of Physiology, Bangladesh Agricultural University, Mymensingh-2202, Bangladesh for his critical review of this article. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
AUTHORS CONTRIBUTION
JF; Conceived and designed the experiments; performed the experiments; contributed reagents, materials, analysis tools and recorded data: MAS; conceived and designed the experiments, analyzed the data, guided to draft the manuscripts, and improved accordingly. MRC, MSA, AT, MMO, MNM; reviewed and corrected the manuscripts and provided technical inputs. MRC, AT; analyzed and interpreted the data.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Yoo S-Y, Park S-J, Jeong D-K, Kim K-W, Lim S-H, Lee S-H, et al. Isolation and characterization of the mutans streptococci from the dental plaques in Koreans. J Microbiol 2007;45:246–55.
- [2]Aljanakh M. Prevalence and severity of dental caries among public school students aged 16-l8 in Hai’l, Kingdom of Saudi Arabia. Int J Health Sci (Qassim) 2017;11:50.
- [3]Okada T, Takada K, Fujita K, Ikemi T, Osgood RC, Childers NK, et al. Differentiation ofbanding patterns between Streptococcus mutans and Streptococcus sobrinus isolates in rep-PCR using ERIC primer. J Oral Microbiol 2011;3:7190.
- [4]Joshi N, Sujan SG, Joshi K, Parekh H, Dave B. Prevalence, severity and related factors of dental caries in school going children of Vadodara city–An epidemiological study. J Int Oral Heal JIOH 2013;5:35.
- [5]Salman HA, Senthikumar R. Identification and antibiogram profile of Streptococcus mutans and Streptococcus sobrinus from dental caries subjects. J App Pharm Sci 2015;5:54–7.
- [6]Simonović DD, Kocić B, Nedeljković NS, Gašić J, Dačić S, Jovanović N. Microbiological status of different areas of tooth. FactaUniversitatis Ser Med Biol 2002;9:236–9.
- [7]Dhamodhar P, Murthy S, Channarayappa SSS, Indiresha HN. Prevalence, characterization and heterogeneity studies on Streptococcus mutans isolated from Bangalore urban population. Int J Pharm Bio Sci 2014;5:122–8.
- [8]Prakash D, Ramesh K, Gopinath N, SS SK, Varuvelil GJ. Antibacterial efficacy of Syzygium aromaticum extracts on multi-drug resistant Streptococcus mutans isolated from dental plaque samples. J Biochem Technol 2014;3:155–7.
- [9]Yadav K, Prakash S, Yadav NP, Sah RS. Multi-Drug Resistance of Bacterial Isolates among Dental Caries Patients. Janaki Med Coll J Med Sci 2015;3:37–44.
- [10]Levy SB, Marshall B. Antibacterial resistance worldwide: causes, challenges and responses. Nat Med 2004;10:S122–9.
- [11]Hossain MS, Hossain MS, Alam S, Nibir YM, Tusty TA, Bulbul SM, et al. Genotypic and phenotypic characterization of Streptococcus mutans isolated from dental caries. BioRxiv 2020.
- [12]Collee JG, Fraser AG, Marmion BP. Simmons (Eds). A Mackie and McCartney Practical Medical Microbiology1996.
- [13]Bhardwaj VK, Sharma KR, Luthra RP, Jhingta P, Sharma D, Justa A. Impact of school-based oral health education program on oral health of 12 and 15 years old school children. J Educ Health Promot 2013;2.
- [14]Arikalan S, Mohankumar A. Antibiogram of Streptococcus mutans isolated from dental caries patients. Int J Med Heal Res 2016;2:79–83.
- [15]Al-Mudallal NHA, Al-Jumaily EFA, Muhimen NAA, Al-Shaibany AA-W. Isolation and identification of mutan’s streptococci bacteria from human dental plaque samples. AlNahrain J Sci 2008;11:98–105.
- [16]El Sherbiny GM. Control of growth Streptococcus mutans isolated from saliva and dental caries. Int J Curr Microbiol App Sci 2014;3:1–10.
- [17]Borty SC, Hafiz KM Bin, Ali MM, Begum K, Ahammed T, Monir MS, et al. Isolation, identification and antibiogram profile of bacteria isolated from dental caries patients of Mymensingh district of Bangladesh. Asian J Med Biol Res 2015;1:244–53.
- [18]Marip A, Kumar A, Al Salamah AA. Prevalence of dental caries bacterial pathogens and evaluation of inhibitory concentration effect on different tooth pastes against Streptococcus spp. African J Microbiol Res 2011;5:1778–83.
- [19]Jain P, Pundir RK. Antibiotic sensitivity pattern of Streptococcus mutans against commercially available drugs. J Pharm Res 2009;2:1250–2.
- [20]Chowdaiah M, Kumar S, Dhamodhar P. An overview on the prevalence of drug resistant Streptococcus mutans in dental caries patient. Int J Res Eng Technol 2016;17:15–8.
- [21]William B, Rwenyonyi CM, Swedberg G, Kironde F. Cotrimoxazole prophylaxisspecifically selects for cotrimoxazole resistance in Streptococcus mutans and Streptococcus sobrinus with varied polymorphisms in the target genes folA and folP. Int JMicrobiol 2012;2012.
- [22]Gharajalar SN, Hassanzade M. Antibacterial properties of Carum copticum essential oil against Streptococcus mutans and Streptococcus sobrinus isolated from canine dental plaque. Vet Med (Praha) 2017;62:654–60.