Evaluation of the thrombolytic activity of the methanolic extract of Melia azedarach fruits and leaves in vitro
Abstract
Melia azedarach is a plant of the Meliaceae family, used worldwide in various medical fields. Because of the widespread incidence of thrombosis worldwide, especially during the coronavirus epidemic, this study was conducted to evaluate the in vitro thrombolytic effect of methanolic extracts of Melia azedarach leaves and fruits. Series of dilutions starting from 2 mg/ml to 20mg/ml were prepared from the methanolic extracts. The results showed that thrombolysis rates were between (18.7%-29.3%) for fruit extract in distilled water, (18.3%-30.1%) in phosphate buffer, (19.3%-35%) for leaves extract in distilled water and (20%-32.3%) in phosphate buffer for dilutions from 2 mg/ml to 20mg/ml. The streptokinase (positive control) had a thrombolytic effect of 47.54 % in distilled water and 44.36 % in phosphate buffer, compared to negative controls of 5.94 % and 6.34 %, respectively. Phytochemical screening found flavonoids and coumarins in leaves extract and only flavonoids in fruit extract, suggesting that the increase in thrombolytic effect may be attributed to these flavonoids and coumarins. The total phenolic content was 15.78 (mg GAE\1g) in leaves extract and 3.64 (mg GAE\1g) in fruit extract, while the total flavonoid content was 0.813 (mg QE\1g) in leaves extract and 0.17 (mg QE\1g) in fruit extract. In conclusion, these results showed that Melia azedarach has a thrombolytic effect.
INTRODUCTION
Coagulation is a complex physiological process performed to stop bleeding, where the blood clot is formed of fibrin, platelets, red and white blood cells [1]. This happens when a blood vessel is damaged, and the platelets are activated, change their shape, then stick to the injury site and each other to create a plug that covers the damaged part and prevents blood from leaking [2]. During this process, the dissolved fibrinogen transforms into undissolved fibrin and then intertwines with the platelets in the plug to create a network that traps more platelets and cells [2]. But when dysfunction occurs in this process, it will lead to three possibilities: increasing the risk of bleeding, blood clotting, or increasing both. When a blood clot is formed, in this case, it is called thrombosis [3].
Infectious diseases and respiratory infections were the leading causes of death worldwide until 1990 when cardiovascular diseases overtook them. After the coronavirus infection, respiratory disorders resurfaced as the leading cause of death, and the risk of thrombosis in respiratory patients has risen from 5% to 16% [4]. The widely utilized thrombolytic agents have shown numerous drawbacks in recent years. These include alteplase, anistreplase, streptokinase (SK), urokinase (UK), and plasminogen activator (tPA), whereas the most commonly used agents are UK and SK [5]. However, these drugs are not very safe due to the immunogenicity factors, high risk of bleeding, allergic reaction, and lack of specificity. Because of the deficiencies of existing thrombolysis agents, extensive research is conducted to find more effective drugs [6].
Plants are the primary sources of natural compounds. According to some reports, approximately 30% of pharmaceuticals worldwide are derivatives from plants, whereas they are considered safer and less toxic in terms of side effects. So that more research is needed to find a new source of natural compounds that have thrombolytic effects [5].
Melia azedarach is commonly known as the chinaberry tree, pride of India, bead tree, and Cape lilac. It is a kind of deciduous tree from the mahogany family, Meliaceae. It is a small to medium-sized tree or shrub, ranging in height from 5 to 15 meters. It is native to Asia and then moved to America, Africa, and Arab countries [7,8].
According to previous studies, Melia azedarach contains many chemical compounds, including terpenes, steroids, alkaloids, flavonoids, anthraquinones, lignans, and coumarins [9]. This plant has several effects: liver protective, anti-fertility, antioxidant, antipyretic, anti-viral, anti-bacterial, anti-nephrolithiasis, anti-cancer, anti-ulcer, anti-protozoal, and anti-parasitic effects, and helps heal wounds [7].
This study was conducted to evaluate the in vitro thrombolytic effect of methanolic extract of Melia azedarach fruits and leaves due to the increasing death rates as a result of thrombosis, particularly during the coronavirus pandemic, and the need for a new source of natural compounds that affect thrombosis effectively and without side effects.
MATERIALS AND METHODS
Plant collection and drying
Yellow ripe fruits and green leaves were collected in September from Damascus university garden, then were identified by Prof. Dr. Imad Kady, Department of Pharmacognosy and Medicinal Plant, Faculty of Pharmacy, Damascus University. The collected parts were dried in the shade for 3-4 days and then crushed.
Preparing the extracts
A 100g of each dried fruit and leaves were combined with 250 ml methanol 99% and soaked for a week at room temperature. After a week, the extracts were filtered and dried at room temperature until their weight stabilized.
Phytochemical screening
A preliminary phytochemical screening was performed on methanolic extracts of leaves and fruits to determine the presence of distinct chemical groups in the compounds. Then a qualitative screening was performed to determine flavonoids, alkaloids, coumarins, anthraquinones, and saponins [10].
Preparation of dilutions
A 200 mg of each dried extract, fruits and leaves were dissolved in 10 ml of distilled water (DW) or phosphate buffer (PB) to prepare a solution with a concentration of 20 mg/ml. Phosphate buffer was used to have a pH similar to blood pH 7.4. A series of different dilutions (2-4-6-8-10-12-14-16-18-20) mg/ml was prepared for each extract. So that we had 4 standard series of dilutions: leaves extract in distilled water, leaves extract in phosphate buffer, fruits extract in distilled water, and fruits extract in phosphate buffer.
Preparation of streptokinase (SK)
Streptokinase suspension of 100 μl (30,000 IU) was used in this study, prepared by adding 5 mL of distilled water or phosphate buffer to a commercially available lyophilized SK vial with a concentration of 1,500,000 IU [11].
Collection of the blood
Whole blood was drawn from healthy human volunteers without a history of oral contraceptives or anticoagulant therapy. Every 1 ml of blood was transferred to a previously weighted sterile Eppendorf tube. The ethical approval number is (1-9-2020 ph. F-Damascus-Un). The informed consent was obtained orally.
Determination of thrombolytic activity
Eppendorf tubes were incubated at 37°C for 45 minutes. After thrombus (clots) formation, the serum was completely removed without destroying the thrombus. Then each tube was weighed again to determine the weight of the thrombus (Thrombus weight = weight of the tube containing the thrombus – weight of the empty tube). A 100 μl of extracts of each concentration previously prepared was added to Eppendorf tubes containing thrombus.
Positive control: 100 μl of prepared streptokinase in distilled water.
100 μl of prepared streptokinase in phosphate buffer.
Negative control: 100 μl of distilled water, 100μl of phosphate buffer.
All tubes were incubated at 37 °C for 90 min. The fluid was removed from the tubes and weighed. The weight difference as a percentage was calculated.
Thrombolysis percentage % = (the difference in the weight of the thrombus between before and after the addition of the extracts/weight of the thrombus before the addition) X 100 [11].
Determination of total phenolic content
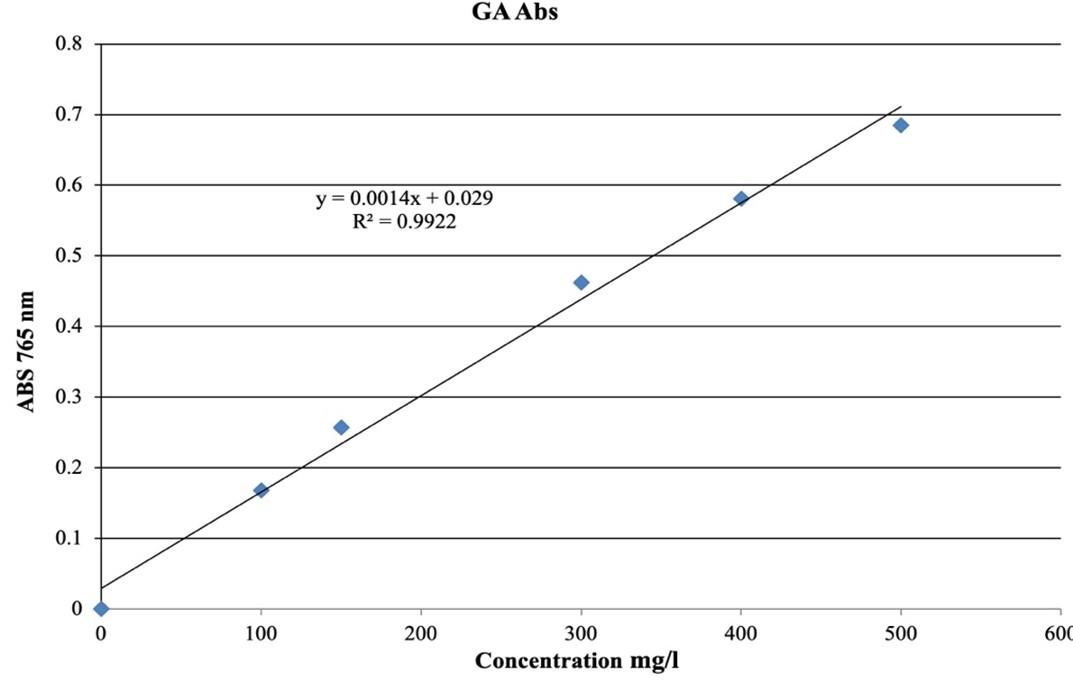
The Folin-Ciocalteu method was used to evaluate the total phenol in methanolic extracts of the fruits and leaves. A 100 μl of Folin-Ciocalteu reagent was mixed with a 20 μl of extracts solution sample in a test tube. Then 1.58 ml of distilled water was added with shaking and incubation in the shade for 9 min at room temperature. Next, a 300 μl of sodium carbonate (20%) was added to the test tube that was kept in the shade for 45 min. The absorbance of all samples was measured at 765 nm with a UV-visible spectrophotometer, and the total phenolic content of extracts was expressed as mg of Gallic acid Equivalent (GAE) per 1g of dried plant. (Gallic acid was used as standard) [12].
Determination of total flavonoids content
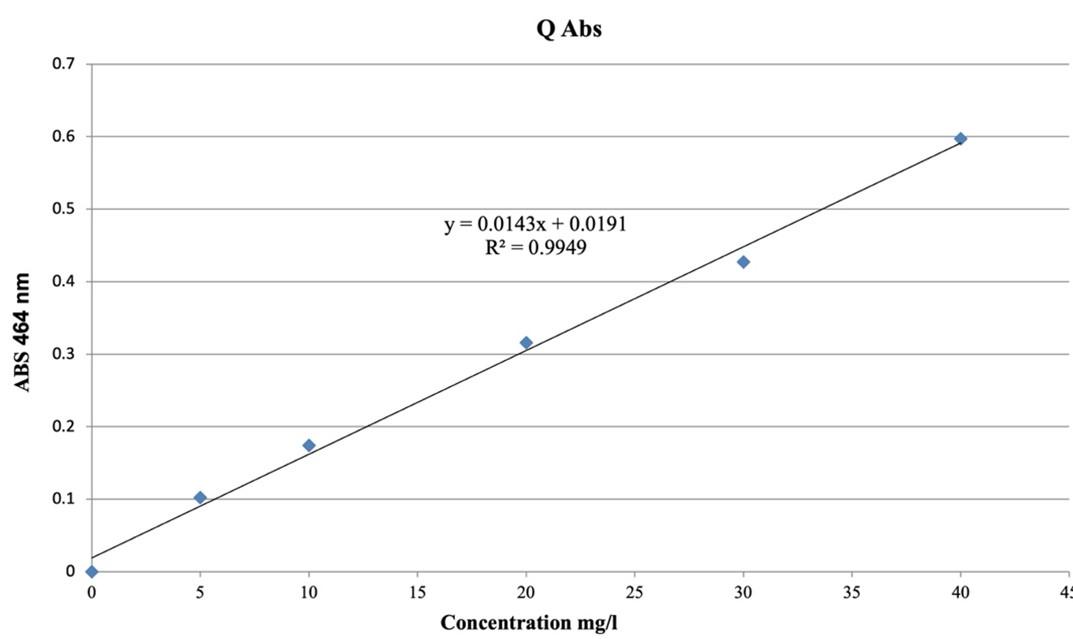
The total flavonoid content of methanolic extracts for fruits and leaves was determined by the aluminium chloride colorimetric method. In the test tube, 1ml of aluminium chloride (2%) was added to 1ml of the sample. Then the tube was shaken and closed to incubate in the shade for 30 min. The absorbance of all samples was measured at 464 nm with UV-visible spectrophotometer and using methanol as a blank. The total flavonoid content in each sample was expressed in mg of quercetin equivalent per 1g of dried extracts (quercetin was used as standard) [13].
Statistical analysis
The statistical study was conducted using GraphPad Prism 8.0.1 statistical program. The numerical data were expressed as the mean and standard deviation and were evaluated by one-way analysis of variance (ANOVA).
RESULTS
The thrombolytic activity for leaves and fruit extracts
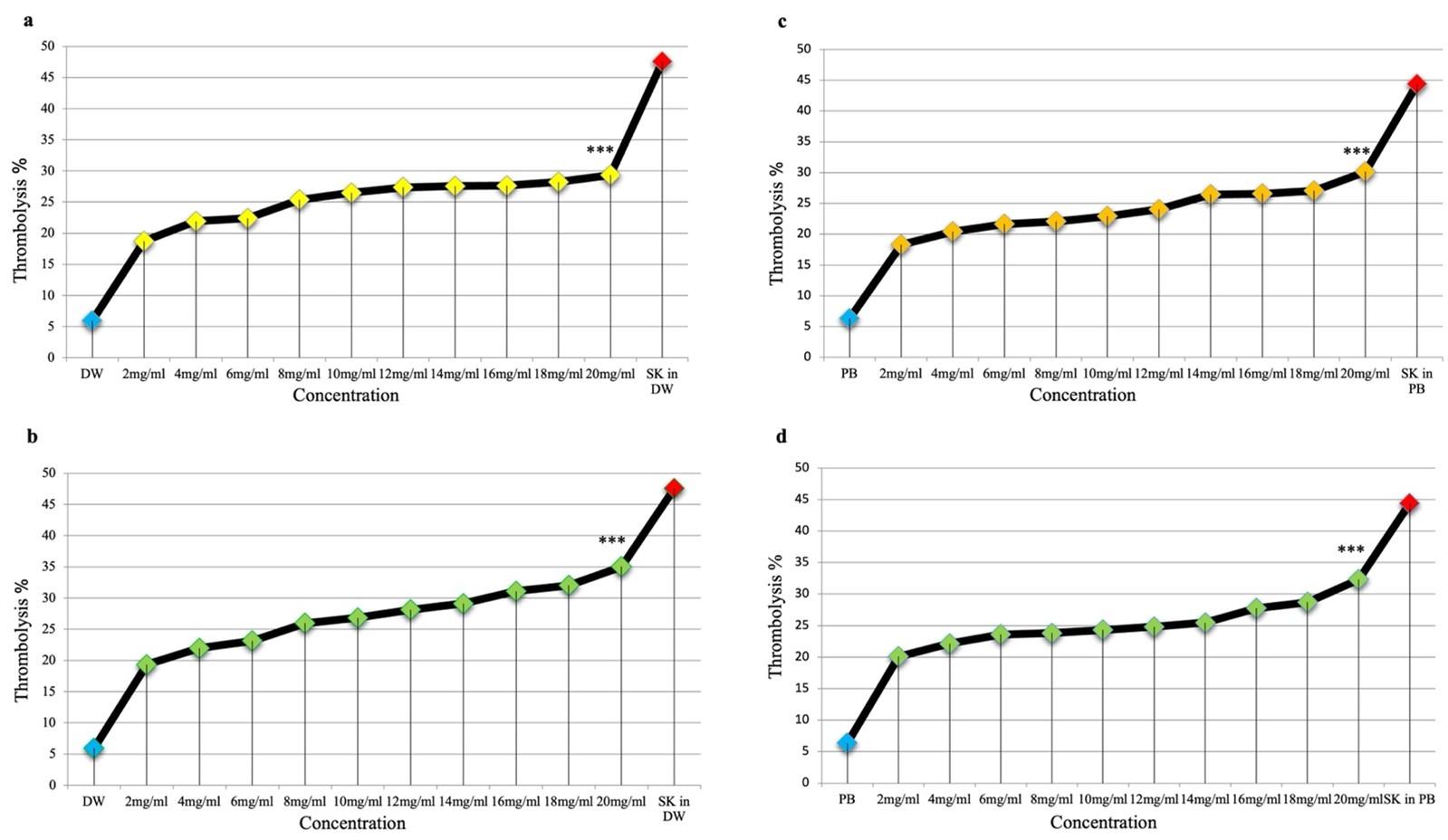
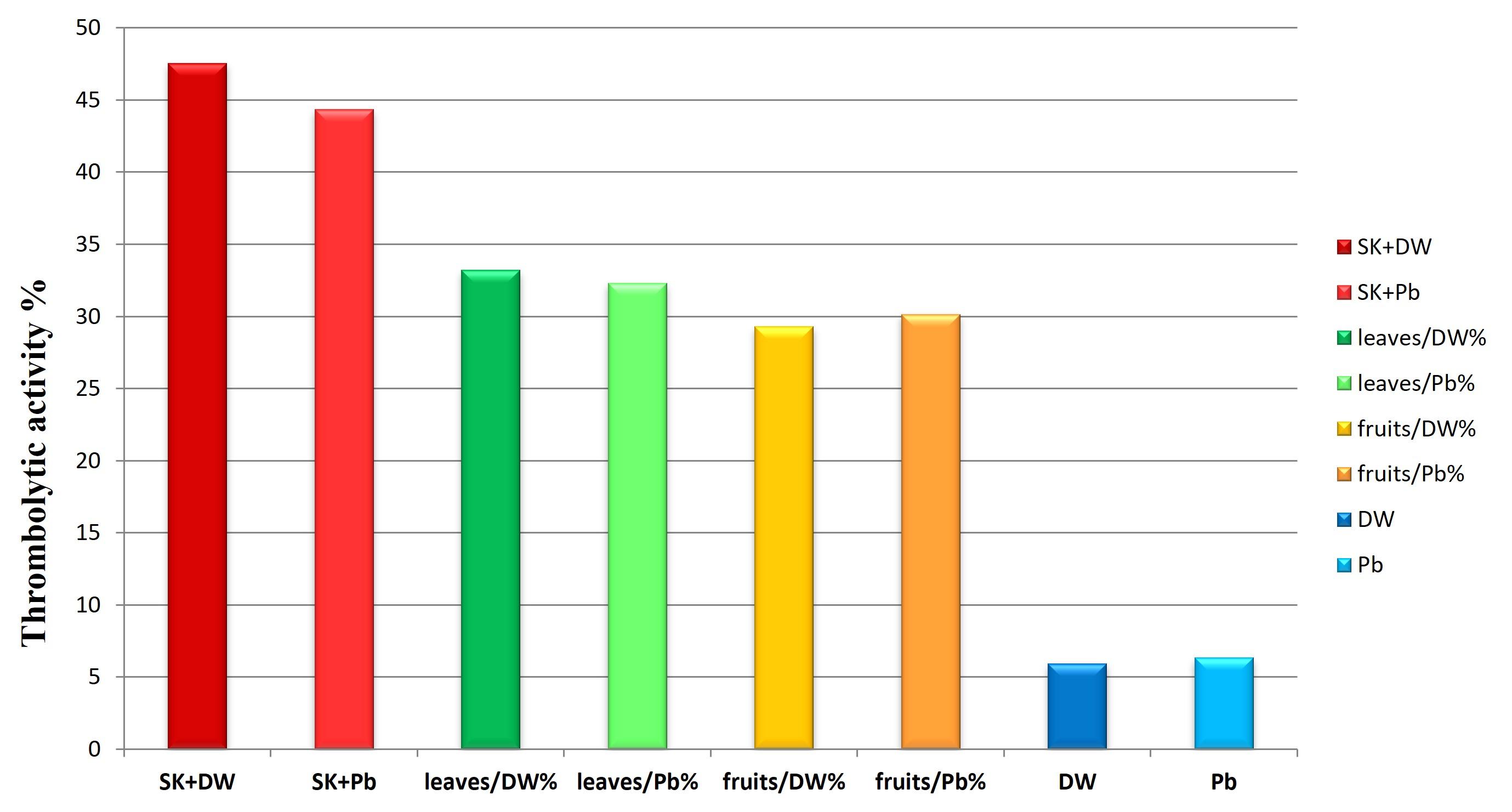
In DW, the fruits, and leaves extracts showed moderate thrombolytic activity: 29.34+0.20% for fruits extract and 35.03+0.38% for leaves extract, compared to streptokinase (47.54+0.26%). In PB, the thrombolytic effect was 30.15+0.6% for fruit extract and 32.32+1.6% for leaves extract compared to streptokinase’s (44.36+0.91%). All results showed in Figures 1a-d and Figure 2.
There was no significant difference neither between leaves extract in DW and PB (P value = 0.069), nor between fruit extracts in DW and PB (P value = 0.5).
Comparing the results of the four series, a significant difference was found only between leaf extract in DW and fruit extract in DW (P-value = 0.002) and a significant difference between leaf extract in DW and fruit extract in PB (P-value = 0.005). Whereas the extract of leaves in DW had the best thrombolytic effect, the remaining three series (leaves in PB, fruits in DW, and fruits in PB) were all close in effect, and no significant difference was observed between them.
Moreover, comparing the highest percentages of thrombolysis reached by each prepared series at a concentration of 20 mg/ml with the positive controls, streptokinase in distilled water or phosphate buffer, we found P-value < 0.001. Therefore, there is a significant difference, which means that the prepared concentrations could not reach the thrombolytic effect of streptokinase (Figure 1 a-d).
The phytochemical screening
The phytochemical screening indicated the presence of coumarins and flavonoids in leaves extract and only flavonoids in fruit extract.
Total phenol and flavonoids content in leaves and fruits
The determination of total phenol was based on the calibration curve of gallic acid used as standard (Figure 3), and of flavonoid was based on the calibration curve of quercetin (Figure 4). It was found that the amount of phenol and flavonoid was higher in leaves extract than in fruit extract (Table 1).
Table 1. Total phenolic content, and total flavonoids content of Melia azedarach fruits and leaves.
DISCUSSION
This study was conducted to evaluate the in vitro thrombolytic effect of methanolic extracts of Melia azedarach fruits and leaves as part of discovering new thrombolytic drugs.
There were no differences between PB and DW extracts in thrombolytic effects, so they can be used as extracts to give the same effects.
The phytochemical screening confirmed the presence of flavonoids in the methanolic extract of fruits and leaves, as well as coumarins in leaves extract. The total phenolic and flavonoid content was higher in leaves extract. These outcomes explain why leaves have a better thrombolytic effect than fruits. These results agreed with a study by Guguloth et al. 2021, which attributed the thrombolytic effect to the flavonoids [13], and the suggested mechanism is weakening platelets adhesion [1]. In addition, a study by Alamgeer et al. 2018 attributed the thrombolytic effect to coumarins, whereby coumarins inhibit calcium activity [14].
For total phenolic content, comparing our study with other studies, Munir et al. 2012 found that the content of phenols reached 82.54 mg GAE/1g and 66.89 mg GAE/1g in leaves and fruits, respectively. In comparison, the flavonoid content reached 16.99 mg QE/1g and 10.67 mg QE/1g in leaves and fruits, respectively [15]. However, we found lower values in our study, which were 15.78 mg GAE/1g in leaves and 3.64 mg GAE/1g in fruits for phenols; 0.813 mg QE/1g in leaves and 0.17mgQE/1g in fruits for flavonoids.
Moreover, the results of Mwamatope et al. were close to ours, where the concentration of phenols and flavonoids in the leaves, respectively, reached 7.828 mg GAE/1g and 0.532mgQE/1g [16].
The differences between these values and ours may be due to different methods of extraction, polar stores, geographical locations in which the plant was studied, and the different harvesting periods, which affected the chemical content of the plant [16].
CONCLUSION
Based on the above results, it is clear that this plant may have significant implications in the thrombolytic field. The extracts can be used in water and blood pH without significant differences. In addition, the leaves extract gave the best thrombolytic effect among the prepared extracts, which may be attributed to flavonoids and coumarins.
This study also indicated the possibility of developing novel drugs in the thrombolytic field from Melia azedarach. This is only a preliminary study, and the extract should be thoroughly investigated phytochemically and pharmacologically to confirm its medicinal and pharmaceutical potential.
ACKNOWLEDGEMENTS
This work was supported by Faculty of Pharmacy, Damascus University.
AUTHORS CONTRIBUTION
MIHA supervised the overall research work, revised the manuscript. critically reviewed the manuscript. HH performed the research work, wrote the manuscript and analyzed the data, revised the manuscript. All authors revised and approved the final version of the manuscript.
CONFLICTS OF INTEREST
There is no conflict of interest among the authors.
References
- [1]Lichota A, Szewczyk EM, Gwozdzinski K. Factors affecting the formation and treatment of thrombosis by natural and synthetic compounds. Int J Mol Sci. 2020; 21: (1–54).
- [2]Furie B, Furie BC. In vivo thrombus formation. J Thromb Haemost. 2007; 5: (12–7).
- [3]Mackman N. Triggers, targets, and treatments for thrombosis. NIH. 2008; 451: (914–918).
- [4]Smilowitz NR, Subashchandran V, Yuriditsky E, Horowitz JM, Reynolds HR, Hochman JS, et al. Thrombosis in hospitalized patients with viral respiratory infections versus COVID-19. Am Heart J. 2021; 231: (93–95).
- [5]Sayeed MA, Kabir H, Mamun M, Rashid U, Bhuiyan FA, Rashid MA. Thrombolytic activity of methanolic extracts of Desmodium paniculatum (L.) and Sarcochlamys pulcherrima (Roxb.). Bangladesh Pharm J. 2014; 17: (67–69).
- [6]Sherwani SK, Bashir A, Haider SS, Shah MA. Thrombolytic potential of aqueous and methanolic crude extracts of Camellia sinensis (Green Tea): In vitro study. J Pharmacogn Phytochem. 2013; 2: (125-129).
- [7]Sultana S, Asif HM, Akhtar N, Waqas M, Rehman SU. Comprehensive review on ethanobotanical uses, phytochemistry and pharmacological properties of melia azedarachlinn. AJPRHC. 2014; 6: (1-7).
- [8]Nadeeka N N A, Kim M S, Chathuranga K, Ma J Y, Kim H, Lee JS. Melia azedarach extract exhibits a broad spectrum of antiviral effect in vitro and in vivo. J Biomed Transl Res 2020;21:125-136..Wang W, Xia Z, Tian Z, Jiang H, Zhan Y, Liu C, et al. Chemical constituents from the fruits of Melia azedarach (Meliaceae). Biochem Syst Ecol. 2020; 92: (94-104).
- [9]Agha MI, Monjed H. Pharmacognosy chemistry. Damascus University; 1997.
- [10]Tabassum F, Chadni SH, Mou KN, Hasif KI, Ahmed T, Akter M. In-vitro thrombolytic activity and phytochemical evaluation of leaf extracts of four medicinal plants of Asteraceae family. J Pharmacogn Phytochem. 2017; 6: (1166-1169).
- [11]Duraisankar M, Ravichandran V. Antipyretic potential of polyherbal ayurvedic products. Asian J Pharm Clin Res. 2012; 5: (146–150).
- [12]Katib R, Sultan N. Evaluation of total phenolic content, total flavonoids content and free radical scavenging activity of lepidium sativum l. seeds and leaves. Bull Pharm Sci. 2021; 44: (377–385).
- [13]Guguloth SK, Malothu N, DSNBK P. GC-MS analysis and thrombolytic property of methanolic leaf extracts of Terminalia pallida Brandis against carrageenan instigated tail thrombosis Model in Mice. Pharmacognosy Res. 2021; 14: (53–60).
- [14]Alamgeer A, Tarar M, Hasan UH, Saleem M. Evaluation of anticoagulant and thrombolytic activity of Berberis orthobotrys in animal model. Bangladesh J Pharmacol. 2018; 13: (196–202).
- [15]Munir A, Sultana B, Babar T, Bashir A, Amjad M, Hassan Q. Investigation on the antioxidant activity of leaves, fruit and stem bark of Dhraik (Melia azedarach). Eur J Appl Sci. 2012; 4: (47–5).
- [16]Mwamatope B, Tembo D, Chikowe I, Kampira E, Nyirenda C. Total phenolic contents and antioxidant activity of Senna singueana, Melia azedarach, Moringa oleifera and Lannea discolor herbal plants. Sci African . 2020; 9: e00481.