Prophylactic effects of vitamin E and selenium on di (n-butyl) phthalate-induced testicular damage in prepubertal rats
Abstract
Di (n-butyl) phthalate (DBP), a potential endocrine disruptor, adversely affects male reproductive function via activation of oxidative stress. This study was conducted to investigate the protective effects of vitamin E (Vit E) and selenium (Se) on DBP-induced testicular damage. Prepubertal rats were given 500 mg DBP/kg alone and in combination with Vit E (250 mg/kg) & Se (1 mg/kg) by oral gavage for 7 days and sacrificed at day 1 (D1), D30 and D45 after administration. Testicular section of DBP-treated rats showed markedly distorted seminiferous tubules, no spermatids and a reduction in the thickness of their epithelial lining, compared to the control. In contrast, co-administration of Vit E & Se protected the seminiferous tubules and regained its normal architecture to the control level. To clarify whether spermatogenic cells differentiate into mature spermatids in the treated testes at the end of first wave of spermatogenesis, immunostaining for Hsc 70t, a specific marker for spermatids, was carried out. As a result, the increase in maturation of spermatids in Vit E & Se+DBP-treated testes, compared to the DBP-treated, was demonstrated. For example, the most advanced spermatids in the tubules from rats in the DBP-treated groups were steps 8-9 at D45 of recovery, while those of the DBP+Vit E & Se-treated were steps 14-19 that were more or less similar to the control group. These results show for the first time that prepubertal administration of Vit E & Se have protective effects on DBP-induced testicular damage and restoration of normal spermatogenesis.
INTRODUCTION
Phthalates are widely distributed industrial chemicals that are used as plasticizers to soften polyvinyl chloride-based products. These are ubiquitous environmental contaminants due to large-scale production, widespread use, and their high concentration in cosmetics and personal care products, toys, pharmaceuticals, medical devices, food packaging, vinyl flooring, and building materials [1]. Upon dietary exposure, phthalates are rapidly metabolized in the gut and other tissues by non-specific esterases and correspondingly produce monoester and alcohol [2]. Human exposure occurs by ingestion of contaminated food, water, drugs and inhalation of indoor air or dermal contact. Phthalates including di(n butyl) phthalate (DBP) have been shown to induce testicular atrophy due to depletion of spermatogenic cells via apoptosis in rodents [3-8].
Increased oxidative damage of protein, lipid and DNA is one of the underlying mechanisms of DBP-induced testicular toxicity [9]. Oxidative stress is the imbalance between formation of reactive oxygen species (ROS) and antioxidant defense mechanisms. Oxidative stress and ROS play an important role in the modulation of several important physiological functions that lead to cellular damage and cell death [10]. This oxidative stress has also been linked to spermatogenic cell apoptosis and to infertility [11].
Vitamin E (Vit E) is a well-known antioxidant that present mainly in cell membranes, plays an important role in scavenging free oxygen radicals [12] and prevents cell membrane damage [13]. A number of studies have shown that, Vit E supplementation significantly protects testis against oxidative damage [14-16]. Also, it decreases lipid peroxidation produced in oxidative stress and enhances the motility and fertilization potential of sperm [17]. Selenium (Se) is another antidoxidant and it is involved in modulation of intracellular redox equilibrium with its several forms of cellular selenoproteins. Se takes part in fundamental biological processes ranging from cellular antioxidant defense to the protection and repair of DNA and apoptosis [18, 19]. Se is also essential for the production of normal spermatozoa and thus plays a critical role in reproduction [20]. As far as we are aware, there are no published reports regarding the effects of both Vit E and Se on the testes exposed to DBP.
MATERIALS AND METHODS
Animals and treatments
Forty prepubertal (3-week-old) Sprague-Dawley (SD) male rats were selected for this study. The rats were housed three to four per plastic cage, maintained on a 12 h light/dark cycle at a constant temperature (22ºC± 1ºC) with a humidity (45% to 70%), and provided water and rodent pellets ad libitum. Animals were maintained and handled humanely in accordance with the guidelines on animal experiments of the Institutional Animal Care and Use Committee (IACUC) of Bangabandhu Sheikh Mujibur Rahman Agricultural University, Gazipur, Bangladesh. The rats were randomly divided into four groups (I–IV) with ten (n=10) rats per group which were treated daily by gavage for 7 days using curved needle to be introduced directly into the stomach. Group I (Control): Rats in this group were administered the same amount of vehicle (mixture of 5% ethanol and 95% corn oil). Group II: Rats in this group were administered 500 mg DBP/kg (Sigma-Aldrich) which was dissolved in the vehicle. Group III: Rats in this group were administered 250 mg Vit E/kg + 1 mg Se/kg in the form of alpha-tocopherol acetate and sodium selenite respectively (E-Sel Powder, Square Pharmaceuticals Limited, Bangladesh) about 2 h prior to administration of DBP as group II. Group IV: This group received only Vit E+Se as group III. Four rats (n=4) in each group were sacrificed under diethyl ether anaesthesia at 24 h (D1) after last administration, three rats (n=3) were at D30 (58-day-old) and rest three rats (n=3) were at D45 (73-day-old) after treatment, and their testes were collected and subjected to histopathology. The first spermatogenic cycle occurs about 7 to 8 weeks after birth in rats; the experimental period was extended 10 weeks of postnatal life to achieve a maximum effect throughout the first wave of spermatogenesis. The dose of DBP (500 mg/kg) was based on our previous reports [4, 5] testicular toxic for rats and Vit E & Se on the basis of previous report [21].
Hematoxylin and eosin staining
The testes were excised and immersed in 10% neutrally buffered formalin for 48 h at room temperature. Then, the samples were washed in 1X PBS for 3 h, dehydrated through a graded series of ethanol, cleared in xylene, and embedded in paraffin. The paraffin blocks were cut into 5 µm. Sections were then stained with Meyer’s hematoxylin and eosin and/or periodic acid–Schiff (PAS)–hematoxylin.
Morphometrical study
The seminiferous tubules diameter and seminiferous epithelial height were measured by a computer-assisted system using Scion Image software (Scion Co., Frederick, MD, USA). From each rat, 5 microscopic fields were used. At least 50 seminiferous tubules diameter were measured from each rat. Precautions were taken to use round or nearly round tubules. The tubular diameter was measured and expressed in μm at 100X magnification with a semi-automatic system which is formed of a trinocular microscope (Olympus Corporation, Japan). Measurements were calibrated in term of micrometer using Nikon micrometer slide before performing any measurements. Seminiferous epithelial height was measured from the basement membrane to the latest stage of germinal cell (spermatids). Data were expressed as mean ± S.E.M.
TUNEL analysis
In order to quantitatively assess the incidence of apoptotic spermatogenic cells after treatment, in situ terminal deoxynucleotidyl transferase-mediated digoxigenin-dUTP nick-end-labeling (TUNEL) assay (TaKaRa, Tokyo, Japan) was performed by using an Apoptotic Detection Kit according to the manufacturer’s instructions. Briefly, the testes sections were deparaffinized and digested with 10 µg/ml proteinase K at 37ºC for 15 min. After being washed three to five times with PBS (pH 7.4), they were treated with terminal deoxynucleotidyl transferase (TdT) enzyme and Labeling Safe Buffer, which were included in the kit. The TdT reaction was conducted at 37ºC for 90 min. After further washing three to five times with PBS, they were incubated with horseradish peroxidase (HRP) goat anti-biotin at 37ºC for 30 min. The localization of HRP sites was determined by application of diaminobenzidine-hydrogen peroxide (DAB) solution. The sections were then counterstained with methyl green and mounted. Images of seminiferous tubules were obtained by using an OLYMPUS (BX50) light microscope connected to a digital camera. Under the microscope of 200X magnification, 3 fields in each section were randomly selected. The area of seminiferous tubules in all fields was measured by a computer assisted system using Scion Image software (Scion Co., Frederick, MD, USA). Then, TUNEL-positive (brown-stained) spermatogenic cells in all selected areas were counted. The number of TUNEL-positive cells was calculated as described in our previous study [4]. Data were obtained from 4 rats in each group and were given as mean ± S.E.M.
Immunohistochemistry
The testes sections were deparaffinized, rinsed in PBS, treated with blocking solution (10% bovine serum albumin in PBS) and then incubated with anti-Hsc70t antibody (gift from Dr. Naoki Tsunekawa, 1:3000 dilution) in blocking solution at 4ºC overnight. After washing in PBS, the sections were incubated with biotinylated goat anti-rabbit IgG (BRL Gaithersbursg, MD, USA) at room temperature for 2 h. They were rinsed again and treated with Avidin Biotinylated Horseradish Peroxidase complex system (Elite ABC Peroxidase Kit, Vector Laboratories, Burlingame, CA, USA). The reaction was stain with DAB. They were counterstained with periodic acid-Schiff (PAS)-hematoxylin.
Statistical analysis
Statistical analysis was performed using Scion Image software (Scion Co., Frederick, MD, USA). All results are represented as the mean ± S.E.M. For the comparison of apoptotic spermatogenic cell index, one-way analysis of variance (ANOVA) was carried out followed by Fisher’s PLSD as a post-hoc test. Student’s t-test was used to compare the mean values of testis weight, seminiferous epithelial height and the tubular diameter. Differences were considered to be statistically significant when the p value was less the 0.05.
RESULTS
Histopathology
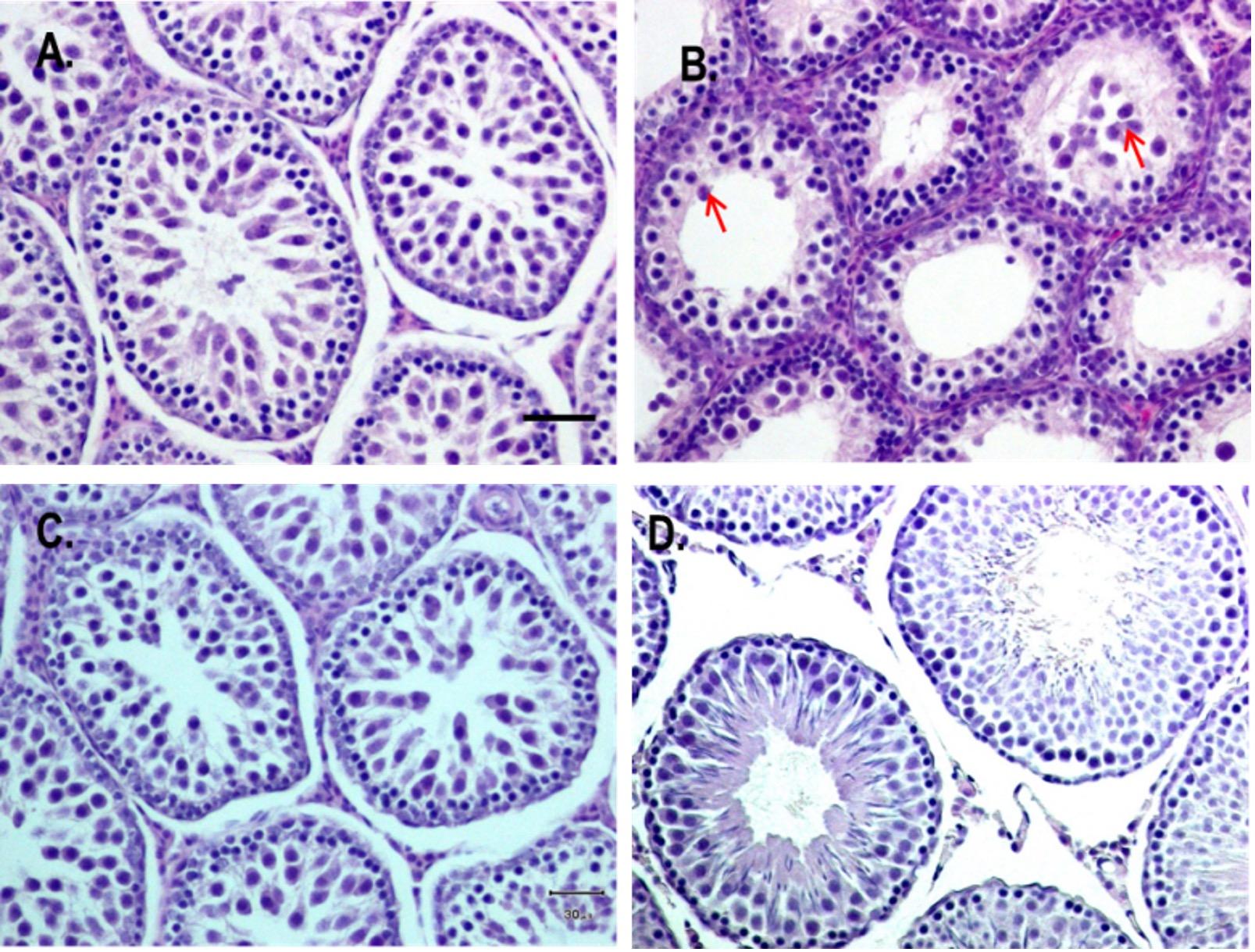
Testicular section from DBP-treated rats exhibited markedly distorted seminiferous tubules with very wide lumen, no spermatids and a reduction in the thickness of their epithelial lining, compared to the control (Figure 1A, B). In addition, sloughing of spermatogenic cells into the lumen were also observed in some tubules (Figure 1B). In contrast, co-administration of Vit E & Se regained its normal architecture to the control level (Figure 1C). In Vit E & Se-treated rats, the histological profile was more or less comparable to that of control, group I (Figure 1D).
Histomorphometry
The data given in Table 1 show that epididymis weight was almost same in all the treated groups. However, the testes weight decreased in DBP- and DBP+Vit E & Se-treated group, compared to the control. The decrease was significant only in DBP-treated group (P<0.05). A significant reduction of seminiferous tubular diameter and their epithelial height were also observed in DBP-treated group, compared to control. Co-administration of Vit E & Se resulted in considerable increase in the tubular diameter and epithelial height, as well as testes weight gain as compared to the DBP administration alone. Thus, Vit E & Se protected testes from the DBP-induced toxicity and regained its normal architecture approximately close to control level.
Table 1. Testes and epididymis weights, and testes histomorphometry after treatment
Parameters Treatments. Data represented as mean ± SEM, *P<0.05 and **P<0.001 compared with the control.
Spermatogenic cell apoptosis
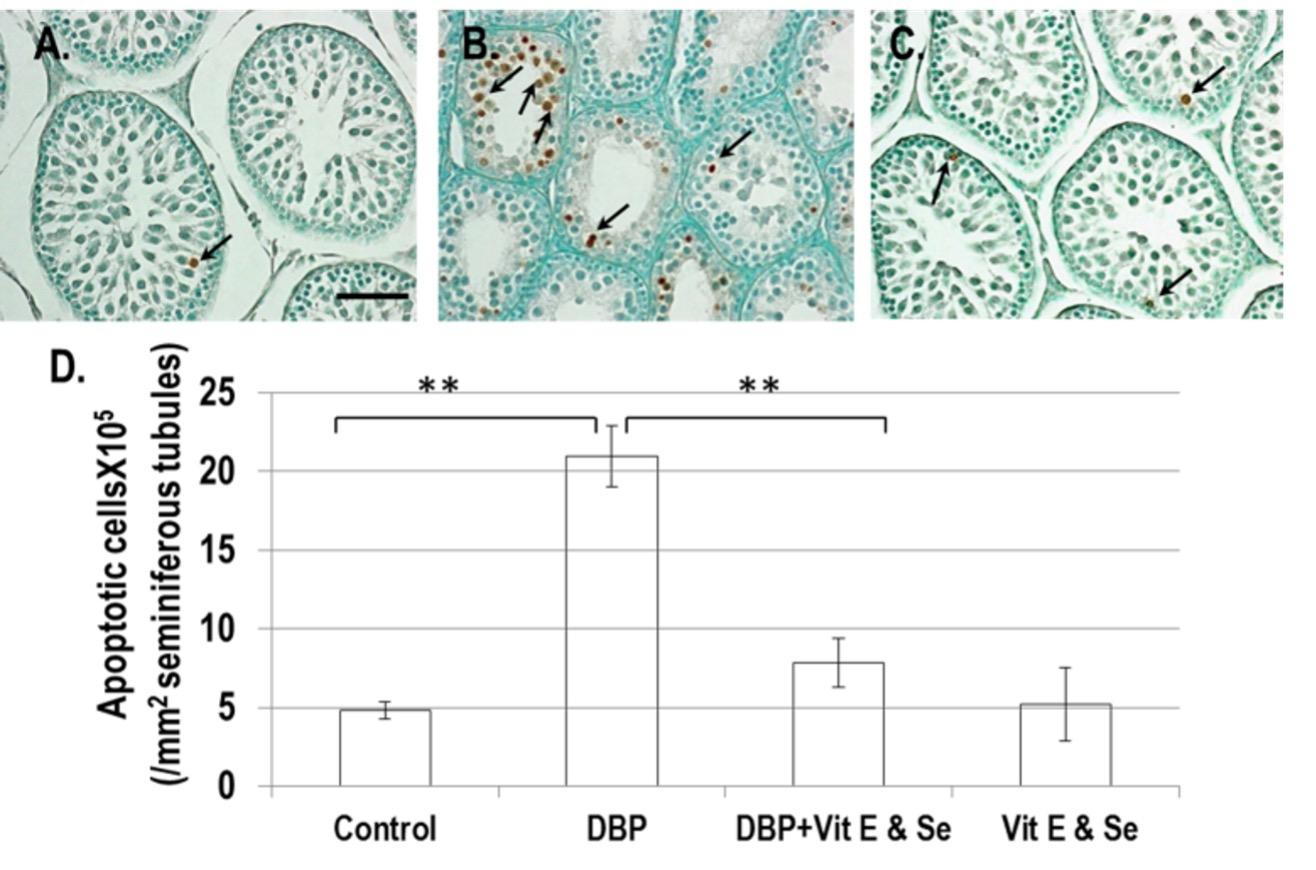
A significant increase number of apoptotic spermatogenic cells were observed in DBP-treated group compared to that of the control group (Figure 2A). In contrast, a significant decrease number of the apoptotic spermatogenic cells was observed in DBP+Vit E & Se-treated group compared to the only DBP-treated group (Figure 2), and thus Vit E & Se administration was found to be protective against DBP-induced testicular damage.
Effects on maturation of spermatogenic cells
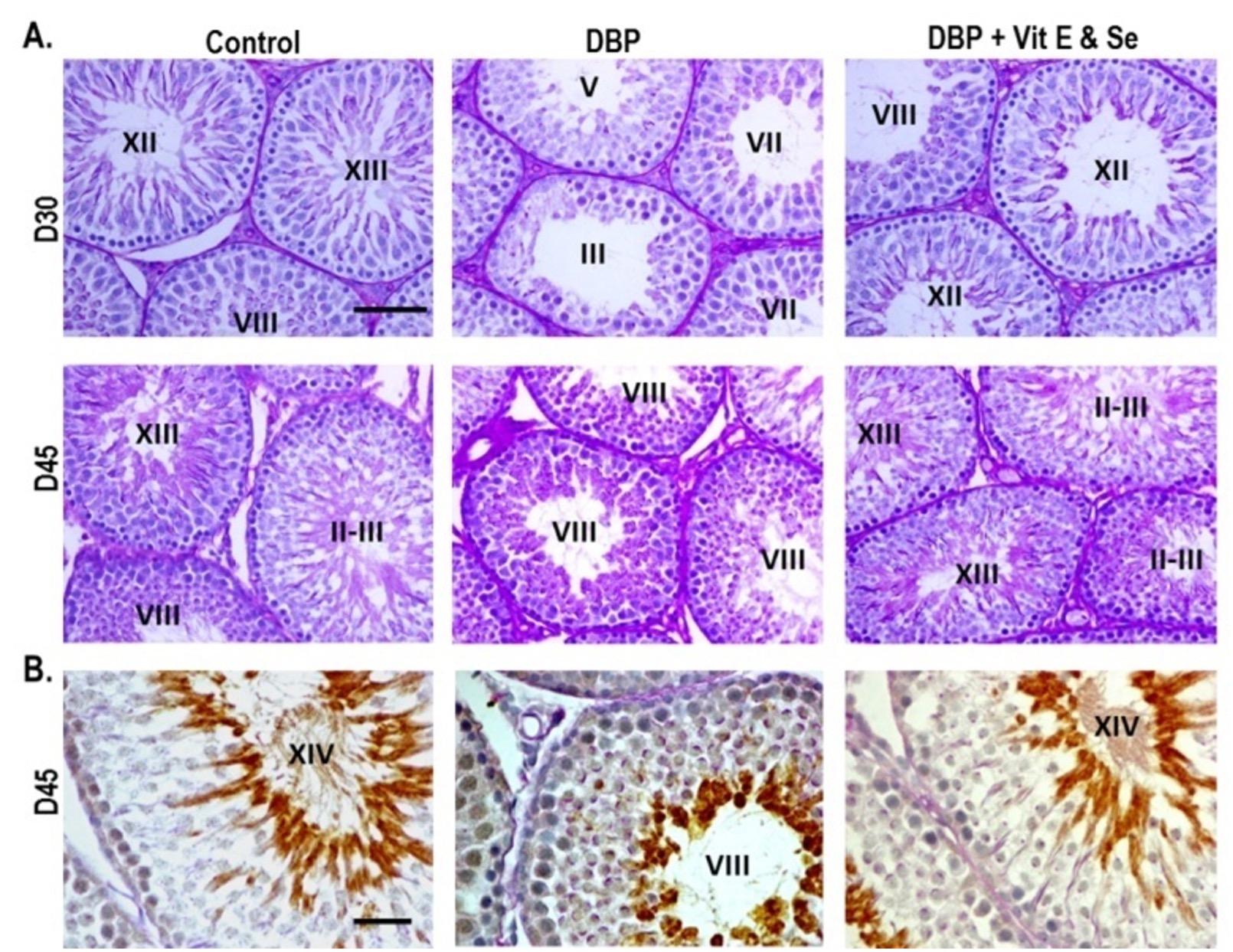
In order to clarify whether spermatogenic cells differentiate into mature spermatids in the DBP- and DBP+Vit E & Se-treated testes, we performed PAS-hematoxylin and immunohistochemical staining of the testicular sections using Hsc 70t antibody. Hsc 70t protein is expressed in the cytoplasm of step 9 to the final step of spermatids, but not in less differentiated spermatogenic cells [22]. Testicular sections of control rats contained the seminiferous tubules with fully matured spermatids, steps15-19. However, exposure to DBP delayed the maturation of spermatogenic cells in the tubules, the most advanced spermatids were steps 2-6. Moreover, loosely arranged spermatogenic cells, and wider tubular lumen were found in the tubules at D30 after treatment. At D45, development of spermatogenic cells in the DBP-treated rats proceeded further, and the most advanced spermatids were steps 8-9, while the co-administration of Vit E & Se showed better maturation of spermatids, compared with only DBP administration, the most advanced spermatids in the tubules were steps 14-19 that almost similar to the control group (Figure 3A). As shown in figure 3B in the control, a strong Hsc 70t immunological reaction was found in the elongated spermatids, step 14 in the adluminal region of tubule. The reaction was observed in rounded spermatids, steps 8-9 in the only DBP-treated rats testes, while combined DBP and Vit E & Se-treated rats testes showed strong immunoreaction in the elongated spermatids, step about 14 that was more or less comparable to that of control, indicating that Vit E and Se administration could alleviate DBP-induced testicular injuries.
DISCUSSION
DBP was particularly chosen for this study due to its potential toxicity on human reproduction and the male reproductive system is the main target organ of the toxicity [23]. In addition, it is used in a variety of consumer products, such as cosmetics, printing inks, and pharmaceutical coatings and can induce testicular atrophy [4, 24]. We used prepubertal rats in the present study and observed the testicular alterations throughout the first wave of spermatogenesis. The first wave of spermatogenesis in rats occurs from the first to 7th/8th weeks of postnatal life, and is a key process in sexual maturation. During the first wave of spermatogenesis, a massive number of spermatogenic cells undergo apoptosis, and the highest rate of apoptosis occurs at 22-26 days of age to maintain an optimal Sertoli and spermatogenic cells ratio [25]. In the present study, we have chosen 3-week-old rat as an animal model, because prepubertal stage is much more sensitive than adults [26], and rats are more sensitive to phthalates than mice [8].
In the present study, rats treated with DBP showed markedly distorted seminiferous tubules with very wide lumen, no spermatids and a reduction in thickness of their epithelial lining. These findings were concordant with our previous report [4] where detachment of spermatogenic cells, loss of stratification and disorganization of lining epithelium of the seminiferous tubules, and reduction in the tubular diameter were observed in 500 mg/kg DBP-treated rats, and complete loss of spermatocytes in the tubules was seen in 1000 mg/kg. However, the tubular epithelial lesions induced by DBP could be significantly improved by co-administration with Vit E & Se. This shows that Vit E & Se can protect testes from the DBP-induced toxicity and regain its normal architecture more or less similar to the control level. To our knowledge, this is the first investigation of prevention of DBP-induced testicular damage by Vit E & Se.
DBP is a known endocrine disruptor. The most prominent effect of DBP is testicular atrophy. To date, several mechanisms have been proposed to explain the induction of testicular atrophy by DBP, such as the depletion of zinc [27], activation of estrogen receptors and reduction of intratesticular testosterone level [4], alteration of Sertoli cell vimentin filament organization [5] and activation of oxidative stress [9]. There have been some attempts at prevention of di(2-ethylhexyl) phthalate (DEHP), a compound quite similar to DBP-induced testicular atrophy. Co-administration of zinc, testosterone or gonadotrophin-releasing hormone failed to afford protection against DEHP-induced testicular atrophy in young rats [16, 17, 28]. Prevention of DEHP-induced testicular disturbance was first achieved by administration of Vit B12 in rats [29]. It has also been reported that co-administration of Vit C and E protected rat spermatogenesis from DEHP-induced toxicity, and that exert a preventive effect through their antioxidant activities [30]. In another study, DEHP exposure caused disruption of vimentin filaments and significantly induced apoptotic death of spermatogenic cells, Se deficiency exacerbated the toxicity, whereas Se supplementation provides protection [31]. Thus, like the Vit B12, Vit C and Vit E, Se is also essential for normal spermatogenesis judging from evidence that animals deficient in the Se exhibits spermatogenic disorders [31]. In the present study, we demonstrated that administration of Vit E & Se significantly improved the spermatogenic status of DBP-treated rats. Although further study is needed to determine which agent, Vit E or Se is more important for the prevention of DBP-induced toxicity, both these have often been simultaneously used in clinical and experimental medicine, because these synergistically exert a potent antioxidant action which affords protection against mammary carcinogenesis [21]. Ebeid, [32] reported that the simultaneous administration of Vit E & Se synergistically increased the antioxidative status by minimizing lipid peroxidation in seminal plasma of the domestic fowl, which enhanced sperm motility and number. Obainime and Roberts, [33] showed that supplementation with Vit C, E and Se caused a reversal of the cadmium-induced biochemical, hormonal and histological toxicities of the liver, kidney and testes in rats.
Mammalian spermatogenesis is one of the most dynamic processes of cell proliferation, differentiation and morphogenesis and involves numerous cellular and molecular steps. In rats, spermatogenesis contains 14 stages and 19 steps of spermatids, and its completion requires about 7 to 8 weeks after birth. In the present study, at D30 (58-day-old) after treatment, the most advanced spermatids in the tubules from rats in the DBP-treated group were steps 3-7 and at D45 (73-day-old), the most advanced spermatids in tubules were steps 8-9, whereas combined exposure of DBP and Vit E & Se significantly improved the maturation of spermatids, the most advanced spermatids in the tubules were steps 14-19 that almost similar to the control group (Figure 3). These results show for the first time that prepubertal administration of Vit E & Se have protective effects on DBP-induced testicular damage and restoration of normal spermatogenesis.
ACKNOWLEDGEMENT
We thank to Dr. Naoki Tsunekawa, the University of Tokyo for giving anti-Hsc70t antibody. This work was supported in part by Grants from RMC/UGC implemented by BSMRAU (to M.Shah Alam).
AUTHOR CONTRIBUTIONS
M.S Alam designed and performed the experiments and wrote the manuscript, N. Hoque analyzed the data.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
References
- [1]Hernandez-Diaz S, Mitchell AA, Kelley KE, Calafat AM, Hauser R. Medications as a potential source of exposure to phthalates in the US population. Environ. Health Perspect 2009; 117: 185–189.Hernandez-Diaz S, Mitchell AA, Kelley KE, Calafat AM, Hauser R. Medications as a potential source of exposure to phthalates in the US population. Environ. Health Perspect 2009; 117: 185–189.
- [2]Wittassek M, Angerer J. Phthalates: metabolism and exposure. Int. J. Androl 2008; 31: 131–138.
- [3]Alam MS, Kurohmaru M. Butylbenzyl phthalate induces spermatogenic cell apoptosis in prepubertal rats. Tissue Cell 2016; 48: 35-42.
- [4]Alam MS, Ohsako S, Matsuwaki T, Zhu XB, Tsunekawa N, Kanai Y, Sone H, Tohyama C, Kurohmaru M. Induction of spermatogenic cell apoptosis in prepubertal rat testes irrespective of testicular steroidogenesis: a possible estrogenic effects of di(n-butyl) phthalate. Reproduction 2010; 139: 427-437.
- [5]Alam MS, Ohsako S, Tay TW, Tsunekawa N, Kanai Y, Kurohmaru M. Di(n-butyl) phthalate induces vimentin filaments disruption in rat Sertoli cells: A possible relation with spermatogenic cell apoptosis. Anat. Histol Embryol 2010; 39: 186-193.
- [6]Tay TW, Andriana BB, Ishii M, Choi EK, Zhu XB, Alam MS, Tsunekawa N, Kanai Y, Kurohmaru M. Phagocytosis plays an important role in clearing dead cells caused by mono(2-ethylhexyl) (MEHP) phthalate administration. Tissue Cell 2007; 39: 241-246.
- [7]Tay TW, Andriana BB, Ishii M, Choi EK, Zhu XB, Alam MS, Tsunekawa N, Kanai Y, Kurohmaru M. An ultrastructural study on the effects of mono(2-ethylhexyl) phthalate on mice testes: Cell death and sloughing of spermatogenic cells. Okajimas Folia Anat. Jpn 2007; 83: 123-130.
- [8]Zhu XB, Tay TW, Andriana BB, Alam MS, Choi EK, Tsunekawa N, Kanai Y, Kurohmaru M. Effects of di-iso-butyl phthalate on testes of prepubertal rats and mice. Okajimas Folia Anat. Jpn 2010; 86: 129-136.
- [9]Aly HA, Hassan MH, El-Beshbishy HA, Alahdal AM, Osman AM. Dibutyl phthalate induces oxidative stress and impairs spermatogenesis in adult rats. Toxicol. Ind. Health 2016, 32: 1467-1477.
- [10]Droge W. Free radicals in the physiological control of cell function. Physiol. Rev 2002; 82: 47–95.
- [11]Kaushal N, Bansal MP. Dietary selenium variation-induced oxidative stress modulates CDC2/cyclin B1 expression and apoptosis of germ cells in mice testis. J. Nutr. Biochem 2007; 18: 553–564.
- [12]Gurel A, Coskun O, Armutcu F, Kanter M, Ozen O. Vitamin E against oxidative damage caused by formaldehyde in frontal cortex and hippocampus: Biochemical and histological studies. J. Chem. Neuroanat 2005; 29: 173-178.
- [13]Ozden S, Catalgol B, Gezginci-Oktayoglu S, Arda-Princci P, Bolkent S, Alpertunga B. Methiocarb induced oxidative damage following subacute exposure and the protective effects of vitamin E and taurine in rats. Food Chem. Toxicol 2009; 47: 1676-1684.
- [14]Murugesan P, Muthusamy T, Balasubramanian K, Arunakaran J. Studies on the protective role of vitamin C and E against polychlorinated biphenyl (Aroclor 1254)- induced oxidative damage in Leydig cells. Free Radic. Res 2005; 39: 1259-1272.
- [15]Erat M, Ciftci M, Gumustekin K, Gul M. Effect of nicotin and vitamin E on glutathione reductase activity in some rat tissues in vivo and in vitro. Eur. J. Pharmacol 2007; 554: 92–97.
- [16]Yousef MI, Abdallah GA, Kamel KI. Effect of ascorbic acid and vitamin E supplementation on semen quality and biochemical parameters of male rabbits. Anim. Reprod. Sci2003; 76: 99–111.
- [17]Naseem M, Goh YM, Hafandi A, Amal NM, Kufli CN, Rajion MA. Effect of vitamin E and soybean oil supplementation on sperm parameters in male sprague-Dawley rats. Trop. Biomed 2007; 24: 45–48.
- [18]Fischer JL, Lancia JK, Mathur A, Smith ML. Selenium protection from DNA damage involves a Ref1/p53/Brca1 protein complex. Anticancer Res 2006; 26: 899–904.
- [19]Ganther DE. Selenium metabolism, selenoproteins and mechanisms of cancer prevention: complexities with thioredoxin reductase. Carcinogenesis 1999; 20: 1657–1666.
- [20]Flohe L. Selenium in mammalian spermiogenesis. Biol. Chem 2007; 388: 987–995.
- [21]Horvath PM, Ip C. Synergistic effect of vitamin E and selenium in the chemoprevention of mammary carcinogenesis in rats. Cancer Res 1983; 43: 5335-5341.
- [22]Tsunekawa N, Matsumoto M, Tone S, Nishida T, Fujimoto H. The HSP70 homolog gene, Hsc 70t, is expressed under translational control during mouse spermatogenesis. Mol. Reprod. Dev 1999; 52: 383-391.
- [23]Howdeshell KL, Rider CV, Wilson VS, Gray LEJr. Mechanisms of action of phthalate esters, individually and in combination, to induce abnormal reproductive development in male laboratory rats. Environ. Res 2008; 108: 168–176.
- [24]Zhang Y, Jiang X, Chen B. Reproductive and developmental toxicity in F1 Sprague–Dawley male rats exposed to di-n-butyl phthalate in utero and during lactation and determination of its NOAEL. Reprod. Toxicol 2004; 18: 669–676.
- [25]Yan W, Suominen J, Samson M, Jegou B, Tappari J. Involvement of Bcl-2 family proteins in germ cell apoptosis during testicular development in the rat and pro-survival effect of stem cell factor on germ cells in vitro. Mol. Cell. Endocrinol 2000; 165: 115-129.
- [26]Kondo T, Shono T, Suita S. Age-specific effect of phthalate ester on testicular development in rats. J. Pediatr. Surg 2006; 41: 1290–1293.
- [27]Oishi S, Hiraga K. Testicular atrophy induced by phthalic acid monoesters: effects of zinc and testosterone concentrations. Toxicology 1980; 15: 197–202.
- [28]Oishi S. Enhancing effects of luteinizing hormonereleasing hormone on testicular damage induced by di-(2-ethylhexyl) phthalate in rats. Toxicol. Lett 1989; 47: 271-277.
- [29]Oishi S. Prevention of di(2-ethylhexyl) phthalate- induced testicular atrophy in rats by co- administration of the vitamin B12 derivative adenosylcobalamin. Arch. Environ. Contam. Toxicol 1994; 26497-26503.
- [30]Ishihara M, Itoh M, Miyamoto K, Suna S, Takeuchi Y, Takenaka I, Jitsunari F. Spermatogenic disturbance induced by di-(2-ethylhexyl) phthalate is significantly prevented by treatment with antioxidant vitamins in the rat. Int. J. Androl 2000; 23: 85-94.
- [31]Erkekoglu P, Zeybek ND, Giray B, Asan E, Hincal F. The effects of di (2-ethylhexyl) phthalate exposure and selenium nutrition on sertoli cell vimentin structure and germ-cell apoptosis in rat testis. Arch. Environ. Contam. Toxicol 2012; 62: 539-547.
- [32]Ebeid TA. Vitamin E and organic selenium enhances the antioxidative status and quality of chicken semen under high ambient temperature. Br. Poult. Sci 2012; 53: 708-714.
- [33]Obianime AW, Roberts II. Antioxidants, cadmium-induced toxicity, serum biochemical and the histological abnormalities of the kidney and testes of the male Wistar rats. Niger. J. Physiol. Sci 2009; 24: 177-185.