An updated review on lumpy skin disease: a perspective of Southeast Asian countries
Abstract
Recently, Lumpy Skin Disease (LSD) has been portrayed as a terrifying threat to cattle in Southeast Asia. A lump like nodules in the external skin and mucous membrane with fever and swollen lymph nodes are the preliminary noticeable clinical signs of this devastating disease. It is commonly an arthropod-borne contagious illness, correspondingly the non-vector spreading through body discharge and infected fomites. The incubation period ranges from one to four weeks leading to viremia. A pronounced socio-economic collapse is driven by reduced quantity and quality of milk, udder infection, thinness, low quality hides, loss of draught power, abortion, infertility, limitation to meat ingestion, higher morbidity, etc. Animals of any age and gender are susceptible to the disease. The morbidity rate varies according to the immune status of animals and frequency of mechanical vectors. Primarily the disease was endemic in most Sub-Saharan regions of Africa, consequently extent to Middle East, Europe, and Asia. In the South-Eastern part of Asia, the disease has first been introduced in Bangladesh in July 2019 followed by China, India, Nepal, Bhutan, Vietnam, Hong Kong and Myanmar. Bangladesh recorded the maximum attack rate in Chattogram whereas at Cuttack in India. Particular vulnerable locations of other countries are yet to be confirmed. There is no epidemiological proceeding considering the present LSD situation report from rest of Asia. Strict quarantine, vector control, and prophylactic vaccine might be the best remedy for limiting the risk factors of the disease. Future studies should be directed towards determining the true burden of LSD on livestock and its potential risk factors with the perspective of geographic distributions.
INTRODUCTION
Lumpy Skin Disease (LSD) is an infectious disease in cattle caused by Lumpy Skin Disease Virus (LSDV) under the family Poxviridae. Currently the disease has been emerged as a devastating threat for the large domesticated ruminants in Asia, Europe and the Middle East [1]. The disease is enlisted by the OIE due to its capacity for fast trans-boundary spread [2,3].
In endemic areas, LSD is a re-emerging transmissible infection that results significant socio-economic impairment to small-scale and courtyard agrarians [4]. Considering the disease burden, morbidity and mortality cattle are found as more sensitive to the illness compared to buffalos and other ruminants [5]. Despite the practice of mixed herd farming in many countries consisting of cattle, sheep, and goats, it is not yet evidenced that small ruminants act as reservoirs for LSDV except for few laboratory experimental inoculation reports [2,5]. Nodular dermatitis is a common feature of LSD in high yielding cattle and Asian water buffalos in comparison to aboriginal Asian and African ruminants [2,6]. The disease is devastating because it causes a dramatic decline in milk yield, abortion, poor coat condition and sterility in bulls [7]. LSDV can spread large distance, even from one continent to another, if infected animals are moved across farms and quarantine protocols are eased [8]. Notably, there are no epidemiological evidence that the disease is zoonotic [9]. Until 1988, the disease was cramped into greater Africa with a gradual spread to the Middle-East, then Eastern Europe, and the Federation of Russia afterward [10]. The outbreak then spread further, with new cases being reported in South and East Asia in 2019 [1,11]. According to an OIE report, Bangladesh was identified as the first hotspot in South Asia, with the first incident occurring on July 14, 2019 [12]. However, during the current study, there is no existing scientific case report of LSD in buffalo in the country. Later, a considerable number of LSD cases has been reported subsequently in China, India, Nepal, Bhutan, Vietnam, Hong Kong and Myanmar [13].
Despite the economic importance of LSD, limited number of studies are accessible on this extremely devastating arthropod-borne disease in South and East Asian states [11,14]. Recurrent outbreak and re-appearance of the disease in various parts of the world pointed out the importance of re-evaluation of the disease biology, viral transmission mechanism and updated preventive and adaptive control techniques. Considering the above-mentioned facts, a systematic review on LSD has been conducted, focusing predominantly on the South-Eastern part of Asia.
MATERIALS AND METHODS
This review was attempted during the concurrent outbreaks of LSD in South-East Asian states. Newly affected countries were often monitored, and the reported data were immediately incorporated with our repository. To retrieve data, a comprehensive investigation of recently published scientific literatures was performed through PubMed and Web of Science databases using different key words like LSDV, LSD, Southeast Asia, Bangladesh, cattle, vaccine. In addition to this, more information’s regarding LSD epidemics in recent days were documented based on the OIE situation report of this zone. However, the study did not consider the reported statistics of local newspapers due to lack of laboratory validations.
BIOLOGY OF LSDV
The virus that causes LSD is an enveloped, linear, ovoid, double-stranded DNA virus under the family Poxviridae and genus Capripoxvirus [15]. The sole serotype of LSDV; “Neethling” was first identified in South Africa and represented similar antigenic properties with goat and sheep pox virus [16]. The virus is characteristically impervious to many physical and chemical agents and remains constant between pH 6.6 and 8.6, but is predisposed to higher alkaline environment [16]. It undergoes an exclusive survival capability in necrotic skin nodules (33 days), desiccated crusts (35 days), sunlight protected infected tissue (6 months) and air-dried hides at room temperature (minimum 18 days) [17]. Resistance to heat is flexible but most isolates are disabled at 55ºC for couple of hours, or 65°C for 30 minutes [18]. The virus is susceptible to highly alkaline or acidic solutions, and detergents containing lipid solvents [19]. The organism becomes defenseless in daylight while inactivated with ultraviolet rays and at 55 °C for one hour [20]. Moreover, LSDV shows susceptibility to 20% chloroform, 1% formalin, ether, 2% phenol, 2–3% sodium hypochlorite, 0.5% quaternary ammonium compounds, iodine compounds dilution and the detergents containing lipid solvents [21].
EPIDEMIOLOGY OF LSD
Geographic distribution
LSD has been reported in a wide range of locations around the world. It was initially discovered in Zambia in 1929, but it went unnoticed [17]. The disease was considered as a case of poisoning or hypersensitivity reaction for insect bites as per the abundance of biting insects at that time of year. The degree of infectiousness was first documented when it struck Zimbabwe, Botswana, and the Republic of South Africa from 1943 to 1945 [19]. The disease was constrained to Sub-Saharan Africa till 1986. Outside this region, the first LSD outbreak occurred in Egypt in 1988, followed by Israel in 1989 [22]. The disease hit the Middle Eastern countries since 1990 including Kuwait (1991), Lebanon (1993), Yemen (1995), United Arab Emirates (2000), Bahrain (2003), and Oman (2010) [11,19]. Subsequently, outbreaks were reported in Jordan, Iraq, and Turkey in the year 2013, and Iran, Cyprus, and Azerbaijan in 2014 [23]. In 2016, along with Saudi Arabia, Russia, Armenia, Georgia, and Kazakhstan, LSD was also pronounced in South-Eastern European countries, namely Greece, Bulgaria, North Macedonia, Serbia, Kosovo, Albania and Montenegro [24]. In Russia, LSD appeared for the first time in 2015 and continued until 2019. Recently devastating effects of the disease has been reported in significant number of Asian countries and the initial source of the virus spread has yet to be determined.
Contemporary state of LSD in Southeast Asia
Currently, a substantial part of South-East Asian animal is becoming affected at a fast pace by the highly contagious disease, LSD. The first land in the continent of Asia to report an occurrence of LSD was Bangladesh. According to the situation report of OIE and recent scientific articles, there are eight countries in this defined region reporting the outbreak of the disease including Bangladesh, China, India, Nepal, Bhutan, Vietnam, Hong Kong and Myanmar until the investigation is conducted. The Republic of Bangladesh is the eighth most populated state in the world, and is terrestrially encircled by India from the east, west, and north, the Bay-of-Bengal from the south and Myanmar from the south-east. Approximately 24 million cattle along with 1.5 million buffaloes are documented in this land [25]. On a regular basis, a great number of animals are imported from India and travelled inland to supply the high demand for beef in the country, as well as in China. In addition, import of zoo animals from different parts of the world may make LSDV easier to enter the country. Because of its first emergence in three upazillas, Anowara, Karnophuli, and Patia in Chattogram in July 2019, LSD has been classified an exotic disease in Bangladesh (Figure 1). There were initially 66 cattle identified among 360 susceptible (18.33%) from these regions on 22nd July, 2019 presenting with external clinical signs suggestive of LSD [12]. Later a true scenario of LSD outbreak had been revealed by Central Disease Investigation Laboratory (CDIL), DLS on 3rd December 2019, while performed real-time PCR. Chattogram has still been found as the highest prevalent area in Bangladesh reporting 23% morbidity among cattle. The study also claimed 1.42%, 0.87%, 0.21%, 0.06% and 0.05% morbidity in cattle in Gazipur, Naryanganj, Dhaka, Satkhira and Pabna respectively (Table 1).
Moreover, phylogenetic analysis exposed the existing strain of LSDV in Bangladesh closely related to LSDV NI-2490, LSDV KSGP-0240, and LSDV Kenya [26]. Another piece of recent molecular study from Chattogram just been reported 10% overall farm level prevalence of LSD that proposed the addition of newly purchased animal into herd as an important risk factor [27]. Besides, the morbidity rates of 41.06% and 21% in Dinajpur Sadar and Barishal were also documented depending on the external clinical signs and skin scrapping [28,29].
On the 3rd of August 2019, China became the second country in Southeast Asia to have an epidemic. There were 65 animals infected in the Ili Kazak region, which is located in the northwestern Xinjiang province bordering Kazakhstan and is home to 4 million cattle, as proven by QPCR [1]. Since then, a total of nine discrete outbreaks have been documented throughout seven provinces of China that figured out the rate of morbidity 19.5% (156 out of 801) and mortality 0.9% (7 out of 801) [30]. The spread of disease has tremendously increased from western to eastern part of China within a year and even beyond the continental to Taiwan Island.
According to the OIE, India faced three primary outbreaks of LSD at Mayurbhanj district in the state of Odisha, followed by one incursion each at four more districts, bringing the total number of outbreaks in the Eastern share of the country. There were 182 clinically affected among 2539 susceptible animals accounted for the apparent morbidity rate 7.1% with no recorded mortalities. In terms of districts affected, Cuttack displayed the highest morbidity rate of 38.34%, and Kendrapara showed 0.75% [11]. Almost after a year pause, Nepal encountered its first outbreak of LSDV at June, 2020 in some adjoin cattle farms at Morang bordered by India. Consequently, few other districts were affected throughout July. All the external nodule samples (34 samples) reacted positive to RT-PCR and no information available of animal death [31].
Based on OIE situation portal, four more states in South-East Asia namely Bhutan, Vietnam, Hong Kong and Myanmar had been attacked by the LSDV. No scientific publications are available regarding the specific affected locality, morbidity and mortality in these lands except the OIE situation reports. A scant of incomplete information’s are gathered in Table 1.
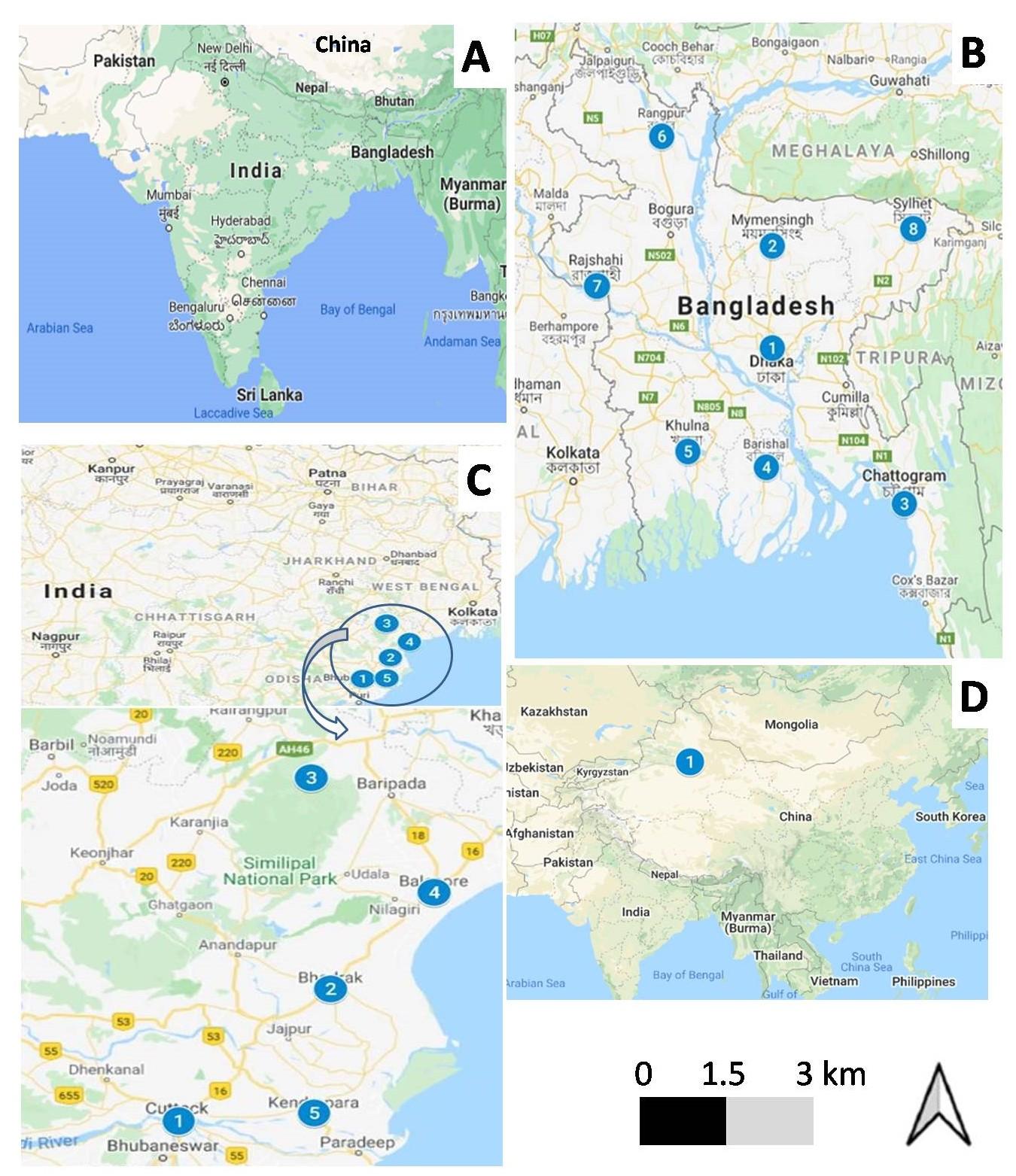
Table 1. Courses and extents of Lumpy Skin Disease in Cattle at Southeast Asian countries from 2019 to 2020.
Table 2. Potential risk factors of Lumpy Skin Disease.
Risk factors
The risk factors for the severity of LSD are identified in 3 basic categories. All the factors along with their states are listed in Table 2.
Host associated factors
LSD is a host-specific disease affecting severely the cattle and Asian water buffalos (Bubalus bubalis) [32]. Buffalo have a substantially lower morbidity rate than cattle [22]. Cattle of both sexes are susceptible to the virus, regardless of their age. The degree of disease severity is determined by the hosts’ susceptibility and immunological condition [33]. Indigenous (Bos indicus) breeds are less vulnerable to clinical disease compared to the Bos Taurus [11,17]. Moreover, young animals exhibited higher susceptibility and severity than the aged cattle [17]. The role of wildlife as a possible viral reservoirs must be clarified [34]. Giraffe (Giraffa camelopardalis) and impala (Aepyceros melampus) showed susceptibility to LSDV in experimental inoculations [22].
Agent related factors
LSDV is remarkably stable under varying environmental conditions. It is resistant to drying and inactivation, can survive in desiccated scabs and also withstand icing and thawing [19]. The virus was reported to be shed in nasal, lachrymal, and pharyngeal exudations of diseased animals, and likewise in saliva, blood, milk, and semen. In the infectious cattle blood, the virus has been isolated within around 8.8 days and viral DNA within 16.3 days [35]. It can last for up to 22 days in semen and 11 days in saliva in a suitable environment [11,36]. Existence for a longer time in fomites, clothing, and equipment has been proved but no indication has been found in insects exceeding four days [17].
Environment and management factors
LSDV can infect, persist, and develop within susceptible host while gets a proper environment. Warm and humid climatic conditions that favor higher proliferation of mosquitoes, flies, and ticks are reported as important environmental risk factors [22]. The disease is mostly seen during wet seasons when there is an abundance of blood-sucking insects in surroundings [11,33]. Few studies reported the higher morbidity in intensive large farms compared to the backyard small farms [11,37]. Common grazing and watering points may facilitate virus circulation through the transmission of vectors [33]. Moreover, the entry of new animals in herds without observing proper quarantine periods was reported as risk factor for LSD [17,22,33].
Transmission of LSDV
The mechanism of LSDV transmission is useful in evaluating the epidemiology of the virus, thus contribute towards progressive control strategy and extinction of the disease [1,38]. An epitome of possible modes of transmission of LSDV is shown in Figure 2.
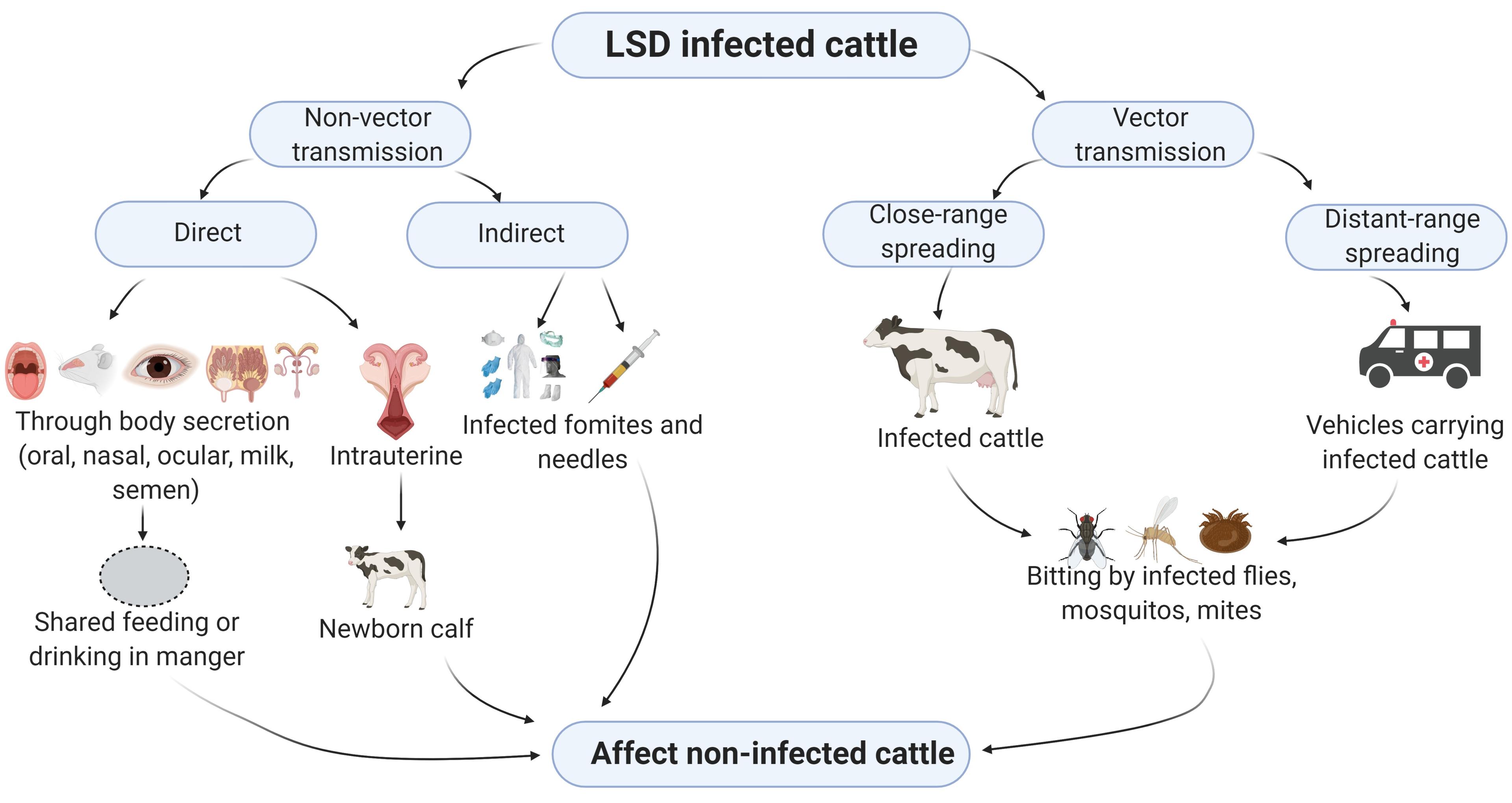
Non-vector transmission
Although ineffective, non-vectored LSD transmission happens when clinically afflicted animals come into contact with contaminated materials, without the need of biological or mechanical vectors. Infectious LSDV is excreted in saliva, nasal and ocular discharges, contaminating communal eating and drinking areas and spreading the disease [17,32,39]. Transmission through contaminated needles during vaccination, dispersion through infected semen during coitus, ingestion of milk, and intrauterine transmission may also act as a sources of infection [17,36,40].
Vector transmission
The role of arthropod vectors in the transmission of this virus was experimentally confirmed [41,42]. Several blood-sucking hard ticks, for instance, Rhipicephalus appendiculatus (brown ear tick), Rhipicephalus decoloratus (blue tick), and Amblyomma hebraeum, mosquito Aedes aegypti and flies Stomoxys calcitran, Haematobia irritans and Musca domestica have been implicated in the spreading of LSDV in sub-Saharan Africa [38–40]. In the tick host, LSDV is trans-stadially [41,42] and transovarially transmitted during cold temperatures [43,44]. The virus may spread in short distances of a few kilometers [45], and even cover longer-distance due to unrestricted animal movements across international borders [20,33].
PATHOGENESIS
LSD is manifested by prompt explosion of multiple circumscribed cutaneous nodules and accompanied by a febrile reaction [46]. The spread of viral particles takes place through blood and form generalized lymphadenitis [47]. Viremia occurs after the early febrile condition for almost 4 days. Following skin lesions due to the replication of the virus in certain cells such as fibroblasts, pericytes, and, endothelial cells of lymphatic and blood vessels lesions are produced in those sites [16,20]. Histopathological changes in acute skin injuries include lymphangitis, vasculitis, thrombosis, infarction, edema and necrosis [19]. Nodules might be found in subcutaneous tissues and muscle fascia [20]. Neighboring tissue of epidermis, dermis, and core musculature reveal hemorrhages, congestion, and edema with distended lymph nodes [22]. A special structure called ‘sit-fasts’ (necrotic cores detached from the adjacent skin) [17] is usually seen indifferent parts of the body, which may ulcerate [48]. The host immunological status exposes the lower rate of lymphocyte diffusion and phagocytic motion during the subsequent fourteen days of post infection [49].
CLINICAL SIGNS
The incubation period of the disease varies from 1 to 4 weeks and then develop fever and downheartedness after viral entry, which continues about 4 to 14 days [15]. The clinical courses of LSD may vary, and these are acute, sub-acute, or in-apparent. Typical LSD is characterized by high body temperature (>40.50C) and skin nodules (10-50 mm diameter) that usually undergo necrosis, affecting the cranium, internal ear, eyelids, muzzle, neck, udder, limbs, perineum, genitalia, and so on [20]. Additional clinical signs comprise lachrymation and nasal expulsion, enlarged subscapular and pre-femoral lymph nodes, and reduced milk yield [17]. Moreover, abortion, prolonged fever, infertility, emaciation, and lameness, may occur in infected animals.
HEALTH AND ECONOMIC IMPACT
The socio-economic impact of LSD can be direct or indirect and has been registered by several major sectors and industries. The sharp drop in milk production is the fast and foremost visible effect directly associated with LSD in the South-Asian region which harbored 21% of the world’s dairy farm animals [50]. According to a Turkish investigation, an impacted cow’s average milk yield fell by 159L each lactation [51]. However, meat from LSD infected cattle is not prohibited from entering the food chain, despite the possibility of the meat having secondary bacterial infection. An estimated 1.2% and 6.2% reduction in beef production per annum among local breeds and Friesian cattle was reported in Ethiopia respectively, due to LSDV infection [52]. Besides, any breaches, scars, or lesions in the raw cattle hides or skin may deteriorate the value of leather, as in the case of severely LSD affected animal hides [53]. Bangladeshi leather is highly admired for its good quality and 56% of leather is generated from cattle [54], that contributed 3.5% of the country’s annual exports [55]. Similarly, having the global exporting position of ninth, India earns annual revenue of US$ 8,500 million for its leather and leather products [56]. Pyrexia and lameness hamper the use of animals for draught purposes. LSD can be transmitted to breeding stock through artificial insemination with infected bull semen, resulting in a lower rate of pregnancy [36]. What is more, several health complications including mastitis, orchitis, abortion, and infertility in bulls also cause huge economic losses for farm owners.
The indirect economic impact of LSD is counted for trade restriction, immunization, quarantine and treatment costs, feed and labor costs, stamping out, maintenance of farm biosecurity, etc. Farm owners need to pay additional cost of feed supplement for sick animals during the period of recovery along with the prolonged duration for fattening [57]. The expenses for LSD in Jordan that involved medication of the affected cattle with broad-spectrum antibiotic and anti-inflammatory drugs was estimated at US$ 35.04 [58]. Sometimes a large number of affected animals have to be stamped out, as was done in Greece [59] and Bulgaria where Bulgaria faced the highest economic disaster of around US$ 8000 per herd [60]. As a trans-boundary infectious disease, the probability of rapid spread of LSD by means of production and marketing channel is high [61]. A risk assessment study for LSD conducted on an Ethiopian bull market estimated the financial loss of US$ 6,67,785.6 considering the culling rates, and the sum of bulls at risk [62]. In a peripheral farming scheme, it is not always rational to adopt quarantine cost-effectively. An estimation figure of quarantine budget in USA including manual labor, feedstuff, diagnostic testing, discarding test positives, and other apprehensive expenses accounted for $145,000 (2010 US$) [63]. Israel paid nearly US$ 750,000 for controlling the initial outbreak of LSD by discarding every suspected animals in the locality and executing the ring vaccination [64,65].
DIAGNOSIS OF LSD
Clinical history, clinical signs, and symptoms of infected animals can be used to make a presumptive LSD diagnosis. During the nodular skin lesion appearance stage, a confirmatory laboratory diagnosis is conducted. There is no diagnostic test tool on the market [22]. The confirmatory tests are mostly in the form of conventional or real-time polymerase chain reaction (PCR) specific for Capri poxvirus [66]. Primers used to diagnose LSD in South-East Asian countries are listed in Table 3. Samples obtained from the skin lesions yield more positive results in PCR than the blood or those collected from septic viscera due to the greater load of viral particles sheltered in the nodule [34]. Fluids like saliva, nasal swab, or whole blood can be collected from clinically infested animals for viral isolation and molecular testing [67].
Additionally, the disease can be detected using serological tests using Enzyme-linked Immunosorbent Assay (ELISA), Indirect Fluorescent Antibody test (IFAT), Indirect Immunofluorescence test, Virus Neutralization Test (VNT) and Serum Neutralization Test (SNT) [68,69]. However, the ELISA has been confirmed experimentally showing higher sensitivity and specificity in comparison with IFTA or VNT [70]. A fairly new assay called Immuno-peroxidase Monolayer Assay (IPMA) has been identified for potential use in LSD diagnosis. It is a cheap and convenient test, adapted to low biosafety levels, and has higher sensitivity and specificity than VNT and commercial ELISA [71]. In autopsy, small nodules alike pox knob can be noticed in the mucous membrane of multiple viscera and cavities such as tongue, oro-nasal cavities, trachea, pharynx, lungs, testis, and urinary bladder, etc. [16].
Table 3. PCR primers available to diagnose LSD in Southeast Asian countries.
DIFFERENTIAL DIAGNOSIS
In animals, LSD is identified by lumpy nodules on the external body coat, mouth, tongue, cornea, oral, and ocular mucus membrane. Almost identical clinical indications have been seen in other disorders, leading to LSD suspicions. Although it has a shorter clinical course, Pseudo-Lumpy Skin Disease, occurred due to the bovine alpha herpes virus, creates nodule-like skin swellings and can be confused with LSD [72]. Allergic symptoms like urticaria and bug bites can resemble bovine LSD in some situations. Pseudocowpox, besnoitiosis, demodicosis, vaccinia virus, bovine papular stomatitis, dermatophilosis, vesicular stomatitis, cutaneous tuberculosis, photosensitization, onchocercosis, and ringworm are all deliberated as the differential diagnoses for LSD [17].
TREATMENT AND CONTROL STRATEGIES
Prophylactic actions of LSD is hardly attempted in epidemic situations other than the symptomatic and supportive treatment like wound repair sprays and antibiotic drugs to restrain the secondary bacterial infections of the skin abrasions [2,73]. Anti-inflammatory drugs and intravenous fluid therapy might be administered to upsurge the appetite although it has no prolific feedback [2]. Literally, no precise antiviral drugs are available for the treatment of LSD, thus prevention through vaccination is the only effective way of restraining the disease [2].
Prophylactic immunization with homologous (Neethling strain) or heterologous live attenuated vaccine (Sheep/Goat pox vaccine) is the best medical prophylaxis for LSD [19,32]. Recently, Bangladesh procured “Lumpyvax”, a commercially available vaccine from MSD Animal Health (https://www.msd-animal-health.co.za/products/lumpyvax/020 product_details.aspx.) for immediate control of the current and seemingly rampant LSD outbreaks in the country. In addition to medical prophylaxis, several other zoo sanitary prophylactic measures are helpful in the control of LSD in domestic animals. These include movement control, restricted grazing [2,32], stamping out of severely affected animals, apposite disposal of infected carcass [74], washing with disinfectant of contaminated premises [75], use of pest repellents [2], strict quarantine [17] and finally, disease awareness campaigns targeting veterinary students and professionals, farmers, herdsmen, animal traders, truck drivers, and artificial inseminators.
CONCLUSIONS AND RECOMMENDATIONS
To recapitulate, this review summarizes eight virgin hotspots and their extent for the Lumpy Skin Disease (LSD) in South-East Asian cattle. The disease has become an extreme threat for marginal farmers. Until nineteenth century, the disease was endemic in greater Africa, which then outstretched into the Middle East, Eastern Europe, and the Russian Federation and recently in Asia. The recurrent assault by LSD in vulnerable areas has stricken the attention of the scientific community. Hence, it is needless to say, this is the high time to anticipate emergency preparedness to limit this trans-boundary disease from spreading enormously. Attention should be concentrated on vector control, movement restriction, harsh quarantine, improved vaccination programs, proper veterinary care, and overall farm sanitary management to avoid incursion and spread of the contagion. Thus, the study encourages future scholars to focus on identifying the source of infection, molecular detection and characterization of the causal agent, and finally, the epidemiology and ecology of LSDV in Southeast Asia.
ACKNOWLEDGEMNENTS
The authors are thankful to Shobhan Das, Faculty of Veterinary, Animal and Biomedical Sciences, Sylhet Agricultural University, Sylhet-3100, Bangladesh for generating the map.
AUTHOR CONTRIBUTIONS
Conceptualization, M.D. and M.M.R.; Methodology, M.D and M.M.R.; Formal analysis, M.D., S.A. and M.M.R.; Data curation, M.D., M.M.R. and M.M.R.; Writing-original draft preparation, M.D., M.S.R.C, S.A. and A.K.M.; Writing- review and editing, M.D, M.S.R.C., S.A., A.K.M., M.J.U, M.M.R. and M.M.R.; Visualization, M.D., M.M.R, M.J.U, and M.M.R.; Supervision, M.M.R. and M.M.R.; Critical revisions and writing, M.M.R. and M.M.R. All authors have read and agreed to the published version of the manuscript.
CONFLICTS OF INTEREST
The authors declare no conflict of interest.
References
- [1]Calistri P, De Clercq K, Gubbins S, Klement E, Stegeman A, Cortiñas Abrahantes J, et al. Lumpy skin disease epidemiological report IV: data collection and analysis. EFSA Journal. 2020;18(2):6010.
- [2]Tuppurainen ESM, Babiuk S, Klement E. Lumpy skin disease. Springer international Publishing, USA, 2018, pp 47-51.
- [3]Swiswa S, Masocha M, Pfukenyi DM, Dhliwayo S, Chikerema SM. Long-term changes in the spatial distribution of lumpy skin disease hotspots in Zimbabwe. Tropical Animal Health and Production. 2017;49:195–199.
- [4]Negesso G, Hadush T, Tilahun A, Teshale A. Trans-Boundary Animal Disease and Their Impacts on International Trade: A Review. Academic Journal of Animal Diseases. 2016;5:53–60.
- [5]Kardjadj M. Capripoxviruses : Transboundary Animal Diseases of Domestic Ruminants. Annals of Virology and Research. 2016;2(3):1024.
- [6]Borisevich S V., Sizikova TE, Petrov AA, Karulin A V., Lebedev VN. Nodular Dermatitis: Emergence of Novel Poxviral Infection in Russia. Problems of Particularly Dangerous Infections. 2018;1:5–11.
- [7]Angelova T, Yordanova D, Krastanov J, Miteva D, Kalaydhziev G, Karabashev V, et al. Quantitative and qualitative changes in milk yield and cheese-making properties of milk in cows vaccinated against lumpy skin disease. Macedonian Journal of Animal Science. 2018;8(2):89-95.
- [8]Sameea Yousefi P, Mardani K, Dalir-Naghadeh B, Jalilzadeh-Amin G. Epidemiological Study of Lumpy Skin Disease Outbreaks in North-western Iran. Transboundary and Emerging Diseases. 2017;64:1782–1789.
- [9]Gelaye E, Lamien CE. Sheep and goat pox. Transboundary Animal Diseases in Sahelian Africa and Connected Regions, Springer International Publishing, USA, 2019, pp 289–303.
- [10]Calistri P, DeClercq K, Gubbins S, Klement E, Stegeman A, Cortiñas Abrahantes J, et al. Lumpy skin disease: III. Data Collection and Analysis. EFSA Journal. 2019;17(3):5638.
- [11]Sudhakar SB, Mishra N, Kalaiyarasu S, Jhade SK, Hemadri D, Sood R, et al. Lumpy skin disease (LSD) outbreaks in cattle in Odisha state, India in August 2019: Epidemiological features and molecular studies. Transboundary and Emerging Diseases. 2020;67:2408-2422.
- [12]OIE World Animal Health Information System Event summary : Lumpy skin disease, Bangladesh. 2020;3:31742.
- [13]FAO/OIE (2020). GF-TADs Lumpy Skin Disease Regional Situation Update. https://rr-asia.oie.int/wp-content /uploads/2021/01/agenda-presentations-1.pdf.
- [14]DLS (2019). Situation Report: Lumpy Skin Disease in Bangladesh Background 3–4. https://fscluster.org/sites/ default/files/documents/sitrep_lsd_20191210.pdf.
- [15]Yilmaz H. Lumpy Skin Disease: Global and Turkish Perspectives. Approaches in Poultry, Dairy & Veterinary Sciences 2017;1:11-15.
- [16]Abdulqa HY, Rahman HS, Dyary HO, Othman HH. Lumpy Skin Disease. Reproductive Immunology: Open Access. 2016;01:1–6.
- [17]Ali A, Gumbe F. Review on lumpy skin disease and its economic impacts in Ethiopia. 2018;7:39–46.
- [18]Kreindel S, Masiulis M, Skrypnyk A, Zdravkova A, Escher M, Raizman E. Emergence of lumpy skin disease in Asia and Europe. FAO. 2016;360:24–26.
- [19]Aber Z, Degefu H, Gari G, Ayana Z. Review on Epidemiology and Economic Importance of Lumpy Skin Disease. International Journal of Basic and Applied Virology. 2015;4:8–21.
- [20]Hailu B, Tolosa T, Gari G, Teklue T, Beyene B. Estimated prevalence and risk factors associated with clinical Lumpy skin disease in north-eastern Ethiopia. Preventive Veterinary Medicine. 2014;115:64–68.
- [21]Mulatu E, Feyisa A. Review: Lumpy Skin Disease. Journal of Veterinary Science & Technology. 2018;09:1-8.
- [22]Tuppurainen ESM, Oura CAL. Review: Lumpy Skin Disease: An Emerging Threat to Europe, the Middle East and Asia. Transboundary and Emerging Diseases. 2012;59:40–48.
- [23]Panel E, Health A. Urgent advice on lumpy skin disease. EFSA Journal 2016;14:4573.
- [24]EFSA 2017. Lumpy skin disease: I. Data collection and analysis. EFSA Journal. 2017;15(4):4773.
- [25]Rahman AKMA, Islam SKS, Sufian MA, Talukder MH, Ward MP, Martínez-López B. Foot-and-mouth disease space-time clusters and risk factors in cattle and buffalo in Bangladesh. Pathogens. 2020;9:423.
- [26]Badhy SC, Chowdhury MGA, Settypalli TBK, Cattoli G, Lamien CE, Fakir MAU, et al. Molecular characterization of lumpy skin disease virus (LSDV) emerged in Bangladesh reveals unique genetic features compared to contemporary field strains. BMC Veterinary Research. 2021;17:61.
- [27]Hasib FMY, Islam MS, Das T, Rana EA, Uddin MH, Bayzid M, et al. Lumpy skin disease outbreak in cattle population of Chattogram, Bangladesh. Veterinary Medicine and Science. 2021;00:1-9.
- [28]Sarkar S, Meher MM, Parvez MMM, Akther M. Occurrences of Lumpy skin disease (LSD) in cattle in Dinajpur sadar of Bangladesh. Research in Agriculture Livestock and Fisheries. 2020;7:445–455.
- [29]Khalil M, Sarker MFR, Hasib Y. Outbreak investigation of lumpy skin disease in dairy farms at Barishal, Bangladesh. Turkish Journal of Agriculture – Food Science and Technology. 2021;9:205-209.
- [30]Lu G, Xie J, Luo J, Shao R, Jia K, Li S. Lumpy skin disease outbreaks in China, since 3 August 2019. Transboundary and Emerging Diseases. 2021;68:216–9.
- [31]Acharya KP, Subedi D. First outbreak of lumpy skin disease in Nepal. Transboundary and Emerging Diseases 2020;67:2280-2281.
- [32]OIE (2021). Lumpy skin Disease (LSD). Situation in Bhutan https://rr-asia.oie.int/wp content/uploads/2021/01/1-presentation-on-lsd-outbreaks-in-bhutan-2020.pdf.
- [33]OIE (2021).Technical meeting on lumpy skin disease (LSD). LSD situation in Viet Nam. https://rr-asia.oie.int/wp-content/uploads/2021/01/4-201221_lsd_vietnam_update_oie_meeting.pdf.
- [34]OIE (2021). Lumpy skin disease in Hong Kong. https://rr-asia.oie.int/wp-content/uploads/2021/01/3-lumpy-skin-disease-in-hong-kong-18-12-2020-afcd.pdf.
- [35]OIE (2021). Current LSD outbreak situation and control measures implemented in Myanmar. https://rr-asia.oie.int/wp-content/uploads/2021/01/2-lsd ppt_20122020_mtmmyanmar.pdf.
- [36]Tuppurainen, E., Alexandrov, T. & Beltrán-Alcrudo, D. Lumpy skin disease field manual – A manual for veterinarians. FAO Animal Production and Health Manual. 2017;20:1-60.
- [37]Gari G, Waret-Szkuta A, Grosbois V, Jacquiet P, Roger F. Risk factors associated with observed clinical lumpy skin disease in Ethiopia. Epidemiology and Infection 2010;138:1657–1666.
- [38]Zeynalova S, Asadov K, Guliyev F, Vatani M, Aliyev V. Epizootology and molecular diagnosis of lumpy skin disease among livestock in Azerbaijan. Frontiers in Microbiology 2016;7:1022.
- [39]Gubbins S. Using the basic reproduction number to assess the risk of transmission of lumpy skin disease virus by biting insects. Transboundary and Emerging Diseases. 2019;66:1873–1883.
- [40]Annandale CH, Holm DE, Ebersohn K, Venter EH. Seminal transmission of lumpy skin disease virus in heifers. Transboundary and Emerging Diseases. 2014;61:443–448.
- [41]Kasem S, Saleh M, Qasim I, Hashim O, Alkarar A, Abu-Obeida A, et al. Outbreak investigation and molecular diagnosis of Lumpy skin disease among livestock in Saudi Arabia 2016. Transboundary and Emerging Diseases. 2018;65:e494–500.
- [42]Sprygin A, Pestova Y, Wallace DB, Tuppurainen E, Kononov A V. Transmission of lumpy skin disease virus: A short review. Virus Research. 2019;269:197637.
- [43]Ali H, Ali AA, Atta MS, Cepica A. Common, Emerging, Vector-Borne and Infrequent Abortogenic Virus Infections of Cattle. Transboundary and Emerging Diseases. 2012;59:11–25.
- [44]Rouby S, Aboulsoud E. Evidence of intrauterine transmission of lumpy skin disease virus. Veterinary Journal. 2016;209:193–195.
- [45]Lubinga JC, Tuppurainen ESM, Stoltsz WH, Ebersohn K, Coetzer JAW, Venter EH. Detection of lumpy skin disease virus in saliva of ticks fed on lumpy skin disease virus-infected cattle. Experimental and Applied Acarology. 2013;61:129–138.
- [46]Lubinga JC, Tuppurainen ESM, Coetzer JAW, Stoltsz WH, Venter EH. Evidence of lumpy skin disease virus over-wintering by transstadial persistence in Amblyomma hebraeum and transovarial persistence in Rhipicephalus decoloratus ticks. Experimental and Applied Acarology. 2014;62:77–90.
- [47]Lubinga JC, Tuppurainen ESM, Mahlare R, Coetzer JAW, Stoltsz WH, Venter EH. Evidence of transstadial and mechanical transmission of lumpy skin disease virus by Amblyomma hebraeum ticks. Transboundary and Emerging Diseases. 2015;62:174–82.
- [48]Lubinga JC, Tuppurainen ESM, Coetzer JAW, Stoltsz WH, Venter EH. Transovarial passage and transmission of LSDV by Amblyomma hebraeum, Rhipicephalus appendiculatus and Rhipicephalus decoloratus. Experimental and Applied Acarology. 2014;62:67–75.
- [49]Hendrickx G, Gilbert M, Staubach C, Elbers A, Mintiens K, Gerbier G, et al. A wind density model to quantify the airborne spread of Culicoides species during north-western Europe bluetongue epidemic, 2006. Preventive Veterinary Medicine. 2008;87:162–81.
- [50]Vorster JH. Pathology Lumpy skin disease. vol. 1. 2008.
- [51]Chihota CM, Rennie LF, Kitching RP, Mellor PS. Mechanical transmission of lumpy skin disease virus by Aedes aegypti (Diptera: Culicidae). Epidemiology & Infection. 2001;126:317–21.
- [52]RADOSTITS O, GAY C, Gay C, Hinchcliff K, Constable P. A Textbook of the Diseases of Cattle, Horses, Sheep, Pigs and Goats: veterinary medicine. Veterinary Medicine. 2007;10:2045–50.
- [53]Neamat-Allah ANF. Immunological, hematological, biochemical, and histopathological studies on cows naturally infected with lumpy skin disease. Veterinary World. 2015;8: 1131–6.
- [54]Siddiky M. Dairying in South Asian region: opportunities, challenges and way forward. SAARC Journal of Agriculture. 2017;15: 173–87.
- [55]Şevik M, Doğan M. Epidemiological and Molecular Studies on Lumpy Skin Disease Outbreaks in Turkey during 2014-2015. Transboundary and Emerging Diseases. 2017;64: 1268–79.
- [56]Gari G, Bonnet P, Roger F, Waret-Szkuta A. Epidemiological aspects and financial impact of lumpy skin disease in Ethiopia. Preventive Veterinary Medicine. 2011;102: 274–83.
- [57]Green HF. Lumpy skin disease: its effect on hides and leather and a comparison on this respect with some other skin diseases. Bull Epizootic Dis of Africa. 1959;7:63–79.
- [58]Paul HL, Antunes APM, Covington AD, Evans P, Philips PS. Bangladeshi leather industry: An overview of recent sustainable developments. Journal of the Society of Leather Technologists and Chemists. 2013;97:25–32.
- [59]Hong SC. Developing the leather industry in Bangladesh. Asian Development Bank. 2018;102:1–8.
- [60]Dwivedi A, Agrawal D, Madaan J. Sustainable manufacturing evaluation model focusing leather industries in India: A TISM approach. Journal of Science and Technology Policy Management. 2019;10: 319–59.
- [61]Tadesse Degu BM, Fesseha H. Epidemiological Status and Economic Impact of Lumpy Skin Disease-Review. Int J Rec Biotech. 2020;8:1–15.
- [62]Abutarbush SM, Hananeh WM, Ramadan W, Al Sheyab OM, Alnajjar AR, Al Zoubi IG, et al. Adverse Reactions to Field Vaccination Against Lumpy Skin Disease in Jordan. Transboundary and Emerging Diseases. 2016;63:213–219.
- [63]Agianniotaki EI, Tasioudi KE, Chaintoutis SC, Iliadou P, Mangana-Vougiouka O, Kirtzalidou A, et al. Lumpy skin disease outbreaks in Greece during 2015–16, implementation of emergency immunization and genetic differentiation between field isolates and vaccine virus strains. Veterinary Microbiology. 2017;201:78–84.
- [64]Casal J, Allepuz A, Miteva A, Pite L, Tabakovsky B, Terzievski D, et al. Economic cost of lumpy skin disease outbreaks in three Balkan countries: Albania, Bulgaria and the Former Yugoslav Republic of Macedonia (2016-2017). Transboundary and Emerging Diseases. 2018;65:1680–1688.
- [65]Rossiter PB, Hammadi N Al. Living with transboundary animal diseases (TADs). Tropical Animal Health and Production. 2009;41:999–1004.
- [66]Alemayehu G, Zewde G, Admassu B. Risk assessments of lumpy skin diseases in Borena bull market chain and its implication for livelihoods and international trade. Tropical Animal Health and Production. 2013;45:1153–1159.
- [67]Peck D, Bruce M. The economic efficiency and equity of government policies on brucellosis: Comparative insights from Albania and the United States of America. OIE Revue Scientifique et Technique. 2017;36: 291–302.
- [68]Molla W, de Jong MCM, Gari G, Frankena K. Economic impact of lumpy skin disease and cost effectiveness of vaccination for the control of outbreaks in Ethiopia. Preventive Veterinary Medicine. 2017;147: 100–107.
- [69]Brenner J, Haimovitz M, Oren E, Stram Y, Fridgut O, Bumbarov V, et al. Lumpy skin disease (LSD) in a large dairy herd in Israel, June 2006. Israel Journal of Veterinary Medicine. 2006;61:73.
- [70]Alexander S, Olga B, Svetlana K, Valeriy Z, Yana P, Pavel P, et al. A real-time PCR screening assay for the universal detection of lumpy skin disease virus DNA. BMC Research Notes. 2019;12:1-5.
- [71]Bedeković T, Šimić I, Krešić N, Lojkić I. Detection of lumpy skin disease virus in skin lesions, blood, nasal swabs and milk following preventive vaccination. Transboundary and Emerging Diseases. 2018;65: 491–496.
- [72]Möller J, Moritz T, Schlottau K, Krstevski K, Hoffmann D, Beer M, et al. Experimental lumpy skin disease virus infection of cattle: comparison of a field strain and a vaccine strain. Archives of Virology. 2019;164: 2931–2941.
- [73]Babiuk S, Bowden TR, Parkyn G, Dalman B, Manning L, Neufeld J, et al. Quantification of lumpy skin disease virus following experimental infection in cattle. Transboundary and Emerging Diseases. 2008;55: 299–307.
- [74]Milovanović M, Dietze K, Milicévić V, Radojičić S, Valčić M, Moritz T, et al. Humoral immune response to repeated lumpy skin disease virus vaccination and performance of serological tests. BMC Veterinary Research. 2019;15:1-9.
- [75]Haegeman A, De Leeuw I, Mostin L, Van Campe W, Aerts L, Vastag M, et al. An Immunoperoxidase Monolayer Assay (IPMA) for the detection of lumpy skin disease antibodies. Journal of Virological Methods 2020;277: 113800.