Long term administration of gentamicin affects hemato-biochemical parameters and liver architecture of Swiss Albino mice
Abstract
Gentamicin is most frequently used aminoglycoside antibiotic. Despite its wide use, the effects of gentamicin have not been clearly studied in relation to alteration of hemato-biochemical parameters and liver injury. In the present study, to evaluate the effects of gentamicin on behavioral, hematological, biochemical and morphological parameters of liver, Swiss albino mice were divided into 4 experimental groups (group A: control; group B: pharmacological dose; group C: pharmacological dose rate with chronic treatment, and group D: high dose with chronic treatment). All the mice from group C and D showed dullness, fearness, roughness of the body coat, anorexia and weakness. Liver weight and size were increased significantly in the mice of group C and D than that of group A or group B. Similarly, in hematological study, Total erythrocyte count (TEC), Total leukocyte count (TLC) and hemoglobin (Hb) % values were decreased significantly, whereas, Alanine amino transferase (ALT) values were increased significantly in the mice of group C and D. In addition, congestion and dark coloration with hepatomegaly were found in treated group C and D. Histological study revealed that the liver parenchyma showed central vein congestion, lymphocytic infiltration, irregular size of hepatocyte and dilatation of sinusoids of treated group C and D. Taken together, our current study suggests that although pharmacological dose of Gentamicin has no adverse effect on liver but chronic pharmacological dose or chronic high dose has serious adverse effect on liver. These observations lead us to postulate that gentamicin induces liver tissue damage after long term treatment.
INTRODUCTION
Liver is the largest gland in our body and plays most important functions not only in the storage and release of nutrients but also in the neutralization and elimination of a variety of toxic substances. Antibiotics are one of the most widely used choices of drugs. These drugs are used for prevention of many problems caused by infections. However, antibiotics have side effects and can damage various body organs including liver, kidney, brain, blood, skin, eyes and mouth [1]. Among the antibiotics, gentamicin is the most widely studied aminoglycoside antibiotic used to treat severe infections of Gram-negative bacteria [2]. The aminoglycoside antibiotic, gentamicin is synthesized by Micromonospora purpurea [3]. The use of gentamicin has tremendously increased in human and veterinary practice due to their greater effectiveness against human, livestock and poultry diseases [4]. But sometimes people of Bangladesh, particularly rural people are not concerned about many legal issues due to limited literacy. They purchase antibiotics without any prescription from physician or even when the practice is not legal. For treatment purposes, they use overdose of antibiotic for a long time which may cause adverse effects in human beings. In rural part of Bangladesh, 95% of the people consume drugs without any prescription and purchase drugs from local pharmacies; only 8% of them consume drugs according to the prescription from physicians [5].
Gentamicin is a heat stable antibiotic that remains active even after autoclaving, thus making it useful in the preparation of certain microbiological growth media. The action on bacteria is bactericidal and gentamicin has increased activity at alkaline pH. In case of gentamicin, oral absorption is minimal and for systemic use gentamicin must be given by the parenteral route. Uptake is rapid after intramuscular injection and it has a serum half – life of 75-110 minutes [6]. The distribution of aminoglycosides antibiotic after an IV injection is virtually complete within 1 hour. The penetration of aminoglycosides across membranous barriers by simple diffusion is very limited due to polycationic nature of these antibiotics. Therefore, very low concentrations of aminoglycosides are found in cerebrospinal fluids or respiratory secretions [7].
Gentamicin is mainly used for clinical practice. The most frequently reported side effects associated with gentamicin therapy are ototoxicity, nephrotoxicity and hepatotoxicity [8]. These forms of toxicity occur more frequently in patients who experience prolonged exposure to serum gentamicin concentrations of greater than 2 mcg/mL [9].
After the use of gentamicin in cells, increased production of Reactive Oxygen Species (ROS) is effective in inducing toxic impacts of this drug on the structure and function of tissues [10]. Gentamicin enhanced the production of superoxide anion, hydrogen peroxide and hydroxyl radicals by mitochondria [11]. Free radicals cause Peroxidation of phospholipids membrane, DNA strand breakage, protein denaturation. These effects induce changes in membrane fluidity, thus the membrane gets permeable even to molecules as large as enzymes [12]. Additionally, blood chemical investigation was conducted for more elucidation of the effects of tissue damage which could be provoked by gentamicin. Therefore, the effects of gentamicin must take into account as problem relating to human beings, animals and birds. Despite its wide use, gentamicin has not been definitively linked to instances of clinically apparent liver injury, but in the present study we investigated morphological and hemato-biochemical effects of gentamicin on liver in mice.
MATERIALS AND METHODS
Chemicals
Gentaren 10% (Reneta, Bangladesh), 100 ml bottle is an aminoglycoside antibiotic preparation. Gentamicin commonly known as a broad-spectrum antibiotic was purchased from local market.
Animals and treatments
The experimental male Swiss albino mice were collected from International Center for Diarrheal Disease Research (icddr’b), Mohakhali, Dhaka, Bangladesh. All the mice were possessed good health and devoid of any external deformities certified by the registered veterinarian from icddr’b. After procurement, all the mice were kept under close observation in order to acclimatize to the new environment for a period of one week prior to commencement of the experiment. All mice were raised under confinement as an intensive system. All experimental protocols were approved by the Animal Welfare and Ethical Committee, Faculty of Veterinary Science, Bangladesh Agricultural University. All efforts were made to minimize the number of mice used and their sufferings. Twenty male mice, aged 5-6 weeks old weighing 25-30 g were used for this experiment. The rats were housed four per one plastic cage, maintained on a 12h light/dark cycle at a constant temperature (70-740F) and humidity (45-60%) and provided water and rodent pellets ad libitum. For each individual, under study a record sheet with full details of each parameter were maintained. For the experimental purpose, the mice were randomly divided into four groups: Group A (Control), without any treatment; Group B (5mg/kg for 7 days), pharmacological dose; Group C (5mg/kg for 30 days), pharmacological dose rate with chronic treatment; Group D (10mg/kg for 30 days), high dose rate with chronic treatment. Each group contained five mice. Body weights of all mice were recorded before starting the treatment. After administration of Gentamicin all the mice were kept under close observation for entire 35days (30 days of treatment period and 5 days of post treatment).
Clinical examination
During the tenure of the treatment, the animals were carefully observed for behavioral study.
Blood chemical analysis
Each animal was euthanized by using chloroform before 2ml of blood was taken in 5 ml disposable syringe by cardiac puncture for estimation of various blood chemical parameters such as- TEC, TLC, Hb%, ALT. The blood sample was allowed to stand for one hour and centrifuged at 3000 rpm for 15 minutes. Eppendrof tubes were used for collection of serum and stored in freeze at -200C. ALT was measured by using commercially available kits. The number of RBC and WBC were calculated accordingly (Number of RBC= Number of counted ×1000 and express the results in millions per cu. Mm; Number of WBC= Number of counted×50 and the result is expressed in thousand per cu. Mm).
Gross and histology
After sacrifice of each animal sequentially, liver was collected from each animal and examined for gross study. For gross study, color, weight and size of liver was taken into consideration.
For histological observation, 5 mm pieces were collected from different side of liver and immersed in 10% formalin for 48 hours. Then, the sample was washed in 10% phosphate buffer solution for 3 hours, dehydration was done by passing the tissue in the ascending grade of alcohol, such as 70, 80, 90, 95, 100% (1), and 100% (2) each for 2 hour and finally 100% (3) for overnight, cleared in xylene and embedded in paraffin. Sections from the paraffin blocks were cut in 5 μm in thickness by using rotatory microtome. Then, the sections were stained with Meyer’s Hematoxylin and Eosin (H&E). The sections were protected by a thin cover slip attached to the slide with a mounting medium ‘DPX’ [13]. The samples were studied with the aid of light microscope.
Photomicrographs
Photomicrographs were taken using ZEISS Axiocam ERc5s microscope (Germany) camera. All images were transferred to Adobe Photoshop Elements 15 (Adobe Systems, CA, USA). Only brightness and contrast were adjusted and no other adjustments were made.
Data analysis
A statistical software package (SPSS, version 20) was used for data analysis. The descriptive data is given as mean ± standard deviation (SD). Chi-squared test was used for the analytical assessment. The differences were considered statistically significant when P values were less than 0.05 and 0.01.
RESULTS
Behavioral changes
Mice of group A were healthy and active without any abnormal signs during the whole experimental period. Mice of group B treated with recommended dose (5 mg/kg) were apparently normal without any abnormal sign up to 7 days of intramuscular administration of gentamicin. Mice of group C (5 mg/kg for 30 days) showed fear with less appetite, roughness of the body, apathy and weakness. However, in group D (10 mg/kg for 30 days), all the mice produced irritable behavior, roughness of the hair coat, dullness, less appetite and weakness. Mortality of the animals (12% and 50%) was found in groups C and D respectively (Table 1), but, the highest concentration was found in group D treated with 10 mg/kg for 30 days.
Table 1. Behavioral effects in the control and gentamicin treated mice.
Hematological and biochemical changes
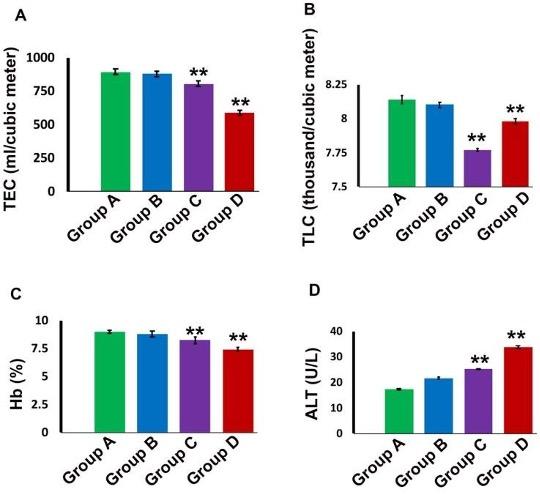
Hematologically, in group A (Control), the mean value of TEC, TLC and Hb % was 891.80±1.304 ml/cubicmeter, 8.14±0.018 thousand/cubicmeter and 8.99±0.013respectively (Figure 1). The value of TEC, TLC and Hb% were decreased significantly (P<0.01) in group C (5 mg/kg for 30 days) and group D (10 mg/kg for 30 days) compared to control group (Figure 1A, B, C). Biochemically, in group A (Control), the mean value of ALT was 17.28±0.130 U/L. This value was increased significantly (P<0.01) in group D (10 mg/kg for 30 days) (Figure 1D).
Gross architectural changes of Liver
Reddish color with normal liver was found in Group A (control). In group B (5 mg/kg for 7 days) liver was normal after the treatment period. Whereas, congestion, dark coloration and hepatomegaly was found in group C and group D (data not shown).
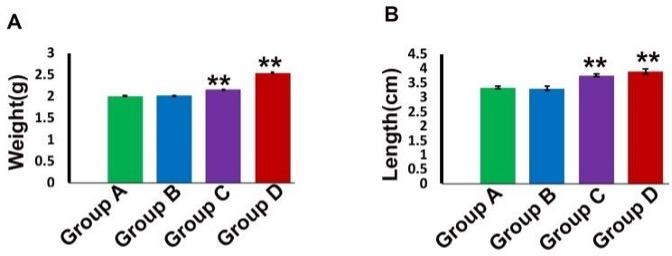
The mean weight of liver of control group was 2.00±0.010 g. Whereas, in group C and group D these parameters were 2.03±0.001** and 2.54±0.039** g respectively. The weight of liver was increased significantly (p<0.01) in group C (5 mg/kg for 30 days) and more significantly (p<0.01) in group D (10 mg/kg for 30 days) (Figure 2).
Microscopic architectural changes of Liver
In liver of group A (control) and group B (pharmacological dose 7 days treatment) liver parenchyma appeared as centrally located central vein and many hepatocytes surrounding the central vein many hepatocytes (Figure 3A, 3B). Long term administration of Gentamicin with pharmacological dose (group C) induced marked congestion around central vein and lymphocytic infiltration around bile canaliculi. (Figure 3C, 3D). In addition to these alterations, in case of higher dose with chronic treatment (group D), dilatation of sinusoids, appearance of inclusion bodies in hepatic parenchyma, tissue destruction and irregularity of the size of hepatocytes were also found in the liver (Figure 3E, 3F, 3G).
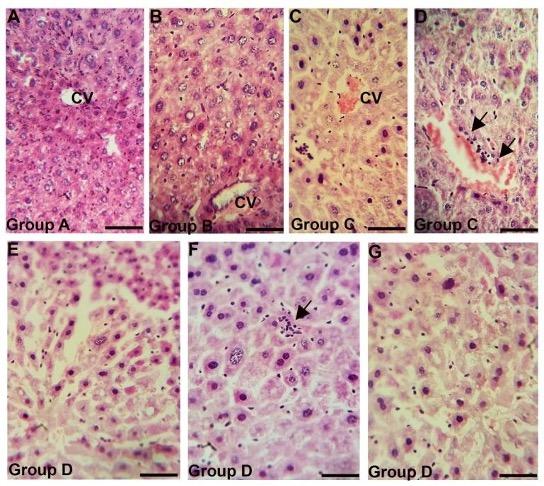
(A) showing intact liver parenchyma with centrally located central vein. (B) also showing intact liver parenchyma. (C-D) showing congestion around central vein and lymphocytic infiltration around bile canaliculi (black arrows), (E-G) showing dilatation of sinusoids (E), inclusions bodies in hepatic parenchyma (black arrow) (F) and tissue destruction and irregularity of liver hepatocytes (G). CV, central vein. Scale bar = 20 µm.
DISCUSSION
In our present study, swiss albino mice were used to observe the morphological and blood chemical alterations of liver after Gentamicin administration.
Gentamicin is commonly used as a therapeutic agent against infections. But long-term exposure of gentamicin in 30% cases may induce hepatorenal toxicity [2, 8]. In case of liver, hepatocytes are hexagonal liver cell that contains many metabolic enzymes. Liver damage may exert to pour these enzymes into plasma and can be useful for the determination of liver damage. After use of Gentamicin is responsible for increased production of reactive oxygen species ROS associated with an increase in lipid peroxidation which takes place in the cell membranes or tissues. Lipid peroxidation is an oxidative stress whereas increased production of ROS decreased antioxidants which lead to an imbalance between oxidant and antioxidant status and ultimately leading to cellular damage [8, 14].
Intramuscular administration of Gentamicin in higher doses showed, roughness of the body, apathy, loss of appetite and weakness. Other groups were observed similar findings when 10 dogs received Gentamicin 10 mg/ kg IM 3 times a day for 14 days [15; 16]. However, they also found diarrhea and vomition following administration of Gentamicin in dog. In our present study, highest concentration of mortality was found in group D (10mg/kg for 30 days). Whereas death of new born in rabbit was also reported following low dose (20mg/kg) of intramuscular injection of Gentamicin during gestation period [17].
Various blood chemical parameters were performed for evaluation of the functions of the organ such as TEC, TLC, Hb% and ALT. In the present study, a significant increase of Alanine amino transferasewas observed and the increased ALT is essential indicator of initial hepatocellular damage [18]. It was postulated that Gentamicin treatment caused elevation in serum urea, creatinine concentration and ALT (Alanine amino transferase) activity associated with pathological changes in liver and kidney [19]. Pathological lesions in the organs and chemical changes in the blood were more severe in diabetic gentamicin treated rats. It was showed that gentamicin could induce renal toxicity and significant increase in the level of ALT [20]. The recorded increased level of ALT indicates functional disorders of the liver as postulated by another researcher [21]. Increased level of ALT due to Gentamicin treatment induced oxidative injury causing tissue damage. This finding is in accord with that of [22] who also reported similar results. In the present study, intramuscular administration of Gentamicin in 3 different doses (5mg/kg for 7 days, 5mg/kg for 30 days and 10 mg/kg for 30 days) significantly reduced the TEC, TLC and Hb%. Similar findings were reported by [23] that long term exposure of Gentamicin in high dose affects the haemopoietic cells in the bone marrow and decrease erythrocyte production. From this study, congestion, dark coloration and hepatomegaly was found in treated group (5 mg/kg for 30 days and 10 mg/kg for 30 days). Gentamicin induced hepatomegaly with decrease the hepatic function in treated animal after intramuscular administration of 75 mg gentamicin /kg for 15 days in rabbits [24].
Histomicrograph study reveals that mice treated with gentamicin with high doses for long term were showed congestion in central vein, lymphocytic infiltration in liver parenchyma and destruction of tissue architecture particularly dilatation of sinusoids. The tissue changes seen in the present work confirmed with the findings of previous work [19; 25]. The cellular organization of mouse liver was studied using light and electron microscopy. Gentamicin treated mouse showed that approximately 35% of the hepatocytes contained two nuclei; none of the Kupffer or Ito cells were double nucleated. The presence of canaliculi and a bile duct system appear similar to that reported for other mammalian species [26; 27].
In conclusion, our present study clarified that long-term treatment of gentamicin with either pharmacological or high dose in swiss albino mice showed a fair degree of reduced food intake, increased mortality, induced significant increase of alanine aminotransferase and caused derangement of liver function with concomitant changes in the histological structures of that organ. The present study suggests that we should have conscious about taking of antibiotic in major or minor issues. The present study may be considered as an experimental base of the relevant human studies.
ACKNOWLEDGEMENT
The authors extend their appreciation to the Ministry of Science and Technology, Bangladesh (MoST; Project no. BS 228/2015-16) for funding the research works.
AUTHOR CONTRIBUTIONS
NJ and MRI designed the experiment. NJ, TA and NS performed the experiments; NJ, MRJ and MRI analyzed the data; NJ wrote the draft, MRJ and MRI critically revised the manuscript; MRI supervised the study.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
References
- [1]Ayatollahi J. Evaluation of knowledge and activities of medical students in the last two yearsof their education about chemoprophylaxis following contact with infectious diseases.Int J Clin Implant Dent. 2005;9 (26): 54-9.
- [2]Stojiljkovic N, Stoiljkovic M. Micromorphological characteristics of the liver andbiochemical analyses in the blood of rats treated by gentamicin and verapamil. Acta MedMedianae. 2006;45 (2): 5-9.
- [3]Gilbert DN, Mandell GL, Bennett & Dolin R. Aminoglycosides in principles and practice of infectious diseases. 5th edition. New York. 2000, pp 307-36.
- [4]Craig W. Pharmacokinetic/pharmacodynamic parameters; rational for antibacterial dosing of mice and men. Clinical Infectious Diseases. 1998, pp 261-10.
- [5]Hossain MM, Glass RI, Khan MR. Antibiotic use in a rural community in Bangladesh.Int J of Epidemiol.1982;11:402-5.
- [6]Gonzelman GM. Pharmacotherapeutics of aminoglycosides antibiotics. Am JRenal Med. 1980; 5:1076-1078.
- [7]Rieviere JE, Coppoc GL. Determination of cerebrospinal fluid Gentamicin in the beagleusing an indwelling cerebral ventricular cannula. Chemotherapy.1981;27:309-312.
- [8]Masakazu K, Yoshiko E, Masashi E. Acquired resistance of Listeria monocytogenes in andescaped from liver parenchymal cells to gentamicin is caused by being coated with their plasma membrane. Microb Infect. 2014; 16 (3):237-243.
- [9]Zimmerman HJ. Aminoglycosides. Hepatic injury from the treatment of infectious and parasitic diseases. In, Zimmerman HJ. Hepatotoxicity: the adverse effects of drugs and other chemicals on the liver, 2nd ed. Philadelphia, Lippincott. 1999, pp 589.
- [10]Wojciech L, Vincent LP. Ternary Complexes of Gentamicin with Iron and Lipid CatalyzeFormation of Reactive Oxygen Species. Chem Res Toxicol. 2005;18(12):357- 364.
- [11]Yang C, Du X, Han Y. Renal cortical mitochondria are the source of oxygen free radicalsenhanced by gentamicin. Ren Fail. 1995; 17:21-26.
- [12]May Y H, Jochen S. Formation of a cytotoxic metabolite from gentamicin by liver.Biochem Pharmacol.1990; 40(11): 11-14.
- [13]Luna L G. Manuals of histologic staining methods of the armed forces institute ofpathology, 3rd edition, McGraw Hill Book Company, New York. 1968.
- [14]Nayma S, Sadia CSM, Tanveer HP, Jesmine A. Effects of Ashwagandha (Withaniasomnifera) root extract on some serum liver marker enzymes (AST, ALT) In Gentamicinintoxicated rats. J Bangladesh Soc Physiol. 2012; 7(1):1-7.
- [15]Dantas AFM, Kommers GD, Hennemann CRA. Experimental gentamicin toxicosis in dogs. Cienc Rural. 1997; 27(3): 451-456.
- [16]Aguiar HCR, Silva CF, Schoenau W, Kommers GD, Silva PA, Leitzka MRM, DE-AguirHCR, De-Silva PA. Urinary gammaglutamyltranspeptidase activity,urinalysis, BUN and Creatinine serum dosage as an auxiliary diagnostic means in dog nephrotoxicity induced byaminoglycosides. Ciencia- Rural. 1997; 27(2): 237-244.
- [17]Lichthorn M. Clinical study on the safety of parentral antibiotic treatment for growing,pregnant and lactating rabbits. 1985, pp:163.
- [18]Michalowicz J, Duda W. Phenols- sources and toxicity. Pol J Environment Stud. 2007; 16 (3):347-362.
- [19]Atef M, Arbid MS, Hanafy MSM. Comparison of gentamicin toxicity in normal and diabetic rats. Acta vet Hung. 1992; 40(1-2):107-111.
- [20]Kadkhodaee M, Khastar H, Faghihi M, Ghaznavi R, Zahmatkesh M. Effect of cosupplementation of vitamin E and C on gentamicin induced nephrotoxicity in rats. Exp Physiol. 2005;90(4):571-576.
- [21]Mayne PD. Clinical Chemistry in Diagnosis and Treatment, Sixth ed. New York,USA.1994, pp:478.
- [22]Lipsky JJ, Cheng L, Sacktor B, Leitman PS. Gentamicin uptake by renal brush bordermembrane vesicles. J. Pharmacol. Clin. Ther. 1980; 215:390-3.
- [23]Lijana RC, Williams MC. The effects of antibiotics on hemolytic behavior of red blood cell.Cell Biophys. 1986;8(4): 223-42.
- [24]Barza M, Ioannidis J, Cappelleri. Single or multiple daily doses of aminoglycoside forinterpretation of renal and medullary concentration. A meta-analysis. 1996, pp:338-345.
- [25]Mahmood DH, Waters A. Comparative study of uranyl nitrate and cisplatin induced renalfailure in rat. European Journal of Drug Metabolism and Pharmacology.1994; 91: 327-336.
- [26]Blouin A, Bolender RP, Weibel ER. Distribution of organelles and membranes betweenhepatocytes and nonhepatocytes in the rat liver parenchyma; A stereological study. J Cell Biol.1977; 72:441–455.
- [27]Bouwens L, Baekeland M, DeZanger R, Wisse E. Quantitation, tissue distribution andproliferation kinetics of Kupffer cells in normal liver.Hepatology.1986;6:718–722.Hepatology.1986;6:718–722.