Ameliorating effects of black seed oil on bisphenol A-induced abnormality of blood, hormone profile and gonadal histology of female mice
Abstract
Bisphenol- A (BPA) is a multi-functional chemical produced in large volumes that is extensively used primarily in the manufacture of polycarbonate plastics and epoxy resins. Black seed oil (BSO) has been employed for many century as one of the main sources of nutrition and healthcare both for humans and animals. The study was carried out to investigate the effects of BPA and BSO on blood, hormone profile, and histopathological changes of ovary in female mice. Total 15 female Swiss Albino mice, aged between 6-8 weeks with an average body weight of 27.4±1g were randomly divided into 3 groups (5 mice/group). Group A act as vehicle control. Group B was administered 50 mg BPA /kg bw daily, while group C received both BPA and BSO and dose rate was 50 mg BPA /kg/day and 1ml BSO/kg/day respectively. Results revealed that Hb concentration, TEC and PCV (p<0.01) were significantly reduced while such difference was absent in other hematological values in BPA treated group compared to control. Inclusion of BSO along with BPA prevented or improved the alterations of those values. Hormonal study showed that, serum thyroxine (T4) and Estradiol level were significantly (p<0.01) reduced by BPA and BSO counteracted the negative effects of BPA. Histopathological study of ovary demonstrated that there were degenerative changes in the lining of granulosa cells of ovarian follicles upon treatment with BPA. From the present study it could be concluded that subjection to BPA is accompanied with anemia, hormonal homeostasis disturbances and granulosa cells degeneration.
INTRODUCTION
There is growing concern about the impact of various endocrine disrupting chemicals (EDCs) on human and animal reproduction, that can affect the normal physiology of the endocrine and reproductive systems by inhibiting the endogenous hormone biosynthesis, metabolism, or modulating the action of hormones [1,2]. Bisphenol-A (BPA), is widely known as an EDC that has been produced on large quantity and extensively utilized industrial chemical for manufacturing many products such as thermal printer paper, electronic equipment, water pipes, sports safety equipment, medical devices, plastic containers, lining of cans for food and beverages [3]. Initially BPA was manufactured as a synthetic estrogen which was used to enhance the rapid growth of poultry and cattle [4]. Human and animal may be exposed to BPA through ingestion, inhalation and dermal exposure. It is reported that daily exposure to BPA to a human body about 10μg/ day [5]. BPA can be detected in urine, serum, placental tissue and even in the fetal liver [6]. Environmental exposure to BPA is associated with many clinical manifestations in humans including immune system dysfunction [7], heart failure and kidney diseases [8]. BPA can suppress the normal reproductive action by inhibiting the function of hypothalamic-pituitary-gonadal axis (HPG axis) [9]. BPA also causes early start of adolescence, birth defects, breast cancer, miscarriages and effects on ability to reproduce [10].
The use of traditional medicine increases every day and have more acceptance than prescribed drugs because it was mostly attributed to being safer than drugs, also patients believe that by using this type of medication there was no need for a physician [11]. One of these alternatives medicinal plant, Nigella sativa commonly known as black cumin, belonging to the family Ranunculaceae and it is native plant from the Mediterranean area used for culinary and medical purposes [12]. The black seed oil is reported to be beneficial for reproductive system as its seeds contain essential oils, unsaturated fatty acids, proteins, steroids, vitamins and minerals [13]. The use of 1ml/kg/day of BSO restored the sexual hormones secretion that led to increase the protein synthesis, white blood cells count and decrease the serum cholesterol concentration [14]. Nigella sativa seeds extract has role in the expulsion of fetal membrane and return of uterus to normal condition, without interfere with the progesterone hormone level during the first cycle in cows treated 100 mg/kg body weight. [15]. The black seed is considered as antidiabetic, anthelmintic, anti-hyperlipidemic, analgesic, immunomodulatory, hypotensive, anti-oxidative, anti-cancer and anti-bacterial effects [16].
So far, in Bangladesh few studies were performed in relation to BPA and black seed oil influence on blood and reproductive organs in mice. The present research work was done with a view to fulfilling the objectives, a) to assess some selected hematological parameters (Hb, PCV, TEC and Erythrocyte indices), b) to observe the action of BPA and BSO on Thyroxine and Estradiol level, and c) to evaluate ovarian histopathology.
MATERIALS AND METHODS
Experimental animals
The mice were purchased from ICDDR’B, Dhaka and housed in a compartmentalized square wooden cages wrapped with wire mesh under controlled conditions of temperature (26-30) °C and relative humidity of 70-80% with natural day light.
Experimental chemicals
Bisphenol-A (BPA) was purchased from Sigma-Aldrich Company, USA. The pure black seed oil with high quality (BSO) was bought from local market in Mymensingh.
Ethical approval
The present study and all experimental procedures were approved and performed according to the guidelines for the care and use of animals as established by Animal Welfare and Experimentation Ethics Committee, Bangladesh Agricultural University, Mymensingh [AWEEC/BAU/2019(36)].
Experimental design
The experiment was conducted during the period from February to April 2018 and totals15 apparently healthy female Swiss albino mice (Mus musculus); aged between 6-8 weeks with an average body weight of 27.4±1g was used. At first, the mice were randomly divided into three groups, each consisted of five mice. Group A (control group) received only basal mouse pellet feed mixed with sunflower oil. Group B was administered Bisphenol-A (BPA) @ 50 mg / kg bw daily, formulated in sunflower oil (as vehicle for BPA), while group C received BPA and BSO @ 50 mg/kgbw/day and 1ml/kgbw/day, respectively.
Management practices
The diet was calculated on daily basis, diet and water were provided ad libitum in all groups. Feeds were kept in air tight poly packed sac to prevent spoilage. During the experimental period, effective hygienic and sanitary measures were followed and also mice cage were cleaned regularly.
Collection of blood and serum preparation
At the end of the experiment (12th week), blood samples were collected by sacrificing the mice. The mice were kept fasting overnight. Then the mice were placed one by one in an airtight container containing diethyl ether pre-soaked cotton. They were being looked over for insensibility and taken out from the airtight vessel and blood was collected directly from heart by a sterile syringe. About 1 to 1.5 ml blood was collected and transferred 0.5-0.75 ml blood into anticoagulant containing tube and the remaining half of blood was transferred to another tube without anticoagulant for serum preparation. As per conventional method serum was separated by centrifugation and stored at – 20 °C until tested.
Hematological parameters
Hemoglobin (Hb), packed cell volume (PCV) and Total Erythrocyte Count (TEC) were performed as per standard method [17]. The RBC indices measure the size, shape, and physical characteristics of the RBCs. It provides information about the hemoglobin content and size of erythrocytes that can be determined by calculating the values obtained from TC of RBC in million/cubic mm, Hb concentration in gm% and PCV in %.
Hormonal assay
The following hormonal parameters: Serum Estradiol and Serum thyroxine (T4) were determined by using Estradiol radioimmunoassay kit and T4 radioimmunoassay kit at the Institute of Nuclear Medicine & Allied Sciences (INMAS), Mymensingh Medical College.
Histopathology
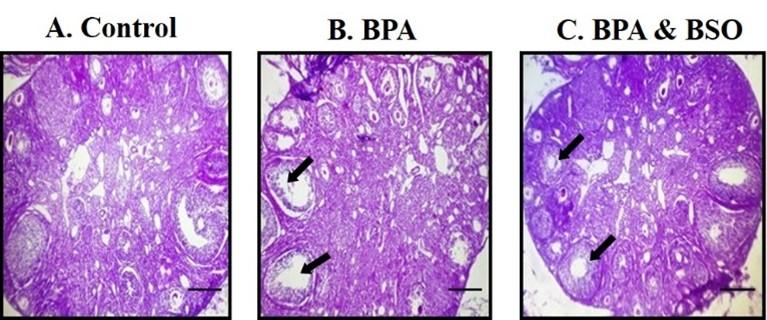
The ovary from each group of mice were collected after complete removal of blood by perfusion with phosphate buffered saline and kept in 10% neutral buffered formalin for 15 days. The well-fixed tissues were processed, sectioned and stained with Hematoxylin and Eosin (H & E) for histo-pathological study [18]. The stained slides were observed under Optka Vision Lite 21 and photographs of the characteristic findings were put down. Histopathologically, the degenerative lesions of ovary in all groups of experimental mice were graded as mild (+), moderate (++) and severe (+++).
Statistical analysis
All data were placed and stored in Microsoft Excel- 2007 and imported to the software IBM SPSS Statistics 20 for analysis by one-way ANOVA followed by post-hoc Turkey’s test. Because of using multiple comparisons, the corrected p value was calculated and adjusted at 0.01 and 0.05 considered for level of significance.
RESULTS
Effects of BPA and BSO on hematological parameters in mice
Effects of BPA and BSO on hematological parameters including Hb, TEC, PCV and Erythrocyte Indices (MCV, MCHC and MCH) in different groups of mice were presented in Table 1. Data revealed that Hb concentration, TEC and PCV (p<0.01) were significantly reduced while such difference was absent in MCV, MCHC and MCHC values in BPA treated group compared to control. Supplementation of BSO in BPA treated mice improved hematological parameters which was non-significant compare to control group.
Table 1. Effect of bisphenol-A (BPA) and black seed oil (BSO) treatment on hematological parameters in mice at 12th week.
Effects of BPA and BSO on hormonal assay in mice
Effects of BPA and BSO on serum thyroxine (T4) and estradiol concentration in different treated groups of mice were shown in Table 2. BPA treated mice showed significantly (p<0.01) decreased the level of T4 and estradiol concentration compared to the control group. Addition of BSO in BPA treated mice restored those level of T4 (p<0.01) and Estradiol (p<0.05) concentration near to normal level.
Table 2. Effect of bisphenol-A (BPA) and black seed oil (BSO) treatment on Thyroxine (T4) and Estradiol concentration in mice at 12th week.
Effects of BPA and BSO on patho-physiological alterations in ovary
To see the toxic effects of BPA directly in the reproductive organs, histology of ovary had been carried out. Section of ovary of non-treated mice showed normal tissue structures and no remarkable changes were found. BPA treated group showed severed degenerative (+++) changes in the lining of granulosa cells of ovarian follicles. On the contrary, it was noticed that restoration of granulosa cells (++) after treated with BSO than the BPA treated group. (Figure 1.)
DISCUSSION
The study was conducted to assess the effect of BPA and BSO on hematological parameters, hormonal assay and histotexture of ovary in female Swiss albino mice. In hematological study, we observed that Hb concentration, TEC and PCV (p<0.01) were significantly reduced in BPA treated mice and the present findings are closely similar with the [19, 20]. They reported that BPA induced a significant decrease in TEC, Hb concentration and PCV. The decrease in the red blood cells may indicate a disruption of erythropoiesis. BPA may interfere with the Hb synthesis or the hemolysis of RBC or react with Hb making a complex, which may affect the physiological functions of Hb [21]. One study reported that, black seed oil significantly elevated Hb, TEC and PCV and decreased MCH and MCHC [22]. The other study found that, BSO restoration the Hb, TEC and PCV value to normal level in ribavirin treated female albino rats [23]. Black seed oil may increase the erythrocyte count and haemoglobin concentration by stimulating the bone marrow and accelerating the effect of it’s on the cellular respiratory mechanism [24]. In case of hormonal assay, BPA treated mice showed significantly (p<0.01) reduced the T4 and estradiol concentration and BSO in BPA treated mice restored those values. The findings of the study are partially consistent with [25-27] stated that BPA can interfere in estradiol level by agonism with estrogen nuclear receptors as well as steroid biosynthesis inhibition. BPA has a direct effect on thyroid follicle and inhibit thyroid hormones synthesis via T3 pathways arrest and suppression of thyroid hormone receptor (TR) transcription [28]. BSO has a positive increased effect on estrogen concentration due to it’s content thymoquinone and unsaturated fatty acids [29]. The treatment with oral administration of black seed oil increased T4 levels in rabbits [30]. Nigella sativa oil used against hypothyroidism due to having its antioxidant effects and thymoquinone constituent that improved thyroid profile [31].
Concerning histopathological study, degenerative changes were found in granulosa cells of BPA treated group and BSO restoration, these changes near to normal ovarian histotexture. One study reports that exposure of BPA reduced the granulosa cells of preantral follicles in cultured mouse [32]. Other experiment showed that BPA may be the causal factor for the death of granulosa cells of ovary and antral follicles via inhibiting cell cycle and disturbances of the apoptotic pathway [33].
CONCLUSIONS
The findings of the present study suggested that exposure to BPA is a potential threat for development of anemia by decreasing Hb concentration, TEC and PCV level. BPA can suppress the sexual behavior and risk factor for developing hypothyroidism, which might be attributed to decrease in thyroxine (T4) and Estradiol levels and granulosa cells degeneration. On the other hand, ingestion of black seed oil may have a positive impact on hematology and reproductive function in female mice by accelerating cellular respiratory mechanisms and regulating the hypothalamo-pituitary-gonadal hormone axis respectively which are connected with the chemical composition of oil. This study seems to suggest a prophylactic activity of black seed oil in the blood and reproductive system of female mice. Further extensive research on this aspect is required to know the possible potential sources and disorders of BPA exposure and to pick up the active ingredients of black seed oil which are actually responsible for combating the BPA induced harmful effects.
ACKNOWLEDGEMENT
The authors thank the authority of the Institute of Nuclear Medicine & Allied Sciences (INMAS) and Department of Pathology, Mymensingh Medical College for hormonal analysis and histo- pathological study of ovary, respectively. This research work was supported by the NST fellowship of Bangladesh for MS degree to the first author of this article (Grant No. 1217/359/2017-2018).
AUTHOR CONTRIBUTIONS
MKI designed the experiment. KMS performed the experiment and wrote the draft; EH and MIH analyzed the data; MKI, MAM and AM critically revised the manuscript.
CONFLICTS OF INTEREST
The author declares that no conflict of interest exists.
References
- [1]Dang VH, Choi KC, Jeung EB. Tetrabromodiphenyl ether (BDE 47) evokes estrogenicity and calbindin-D9k expression through an estrogen receptor-mediated pathway in the uterus of immature rats. Toxicol. Sci. 2007; 97:504–511.
- [2]Choi KC,Jeung EB. The biomarker and endocrine disruptors in mammals. J REPROD DEVELOP. 2003; 49: 337–345.
- [3]Geens T, Goeyens L, Covaci A. Are potential sources for human exposure to bisphenol-A overlooked? Int. J. Hyg. Environ. Health. 2011;214:339–347.
- [4]Singh S, Li SS. Epigenetic effects of environmental chemicals bisphenol A and phthalates. Int. J. Mol. Sci. 2012; 13(8):10143-10153.
- [5]Geens T, Aerts D, Berthot C, Bourguignon JP, Goeyens L, Lecomte P. A review of dietary and non-dietary exposure to bisphenol-A.Food Chem. Toxicol. 2012; 50:3725–3740.
- [6]Kasper-Sonnenberg M, Koch HM, Wittsiepe J, Bruning T, Wilhelm M. Phthalate metabolites and Bisphenol A in urines from German school-aged children: Results of the Duisburg Birth Cohort and Bochum Cohort Studies. Int. J. Hyg. Environ. Health. 2014; 217:830-838.
- [7]Holladay SD, Xiao S, Diao H, Barber J, Nagy T, Ye X,GogalJr RM. Perinatal BisphenolA exposure in C57B6/129svj male mice: Potential altered cytokine/chemokine production in adulthood. Int. J. Environ. Res. Public Health. 2010; 7:2845-2852.
- [8]Shankar A, Teppala S, Sabanayagam C. Bisphenol A and peripheral arterial disease: Results from the NHANES. Environ. Health Perspect. 2012; 120: 1297-1300.
- [9]Manfo FP, Jubendradass R, Nantia EA, Moundipa PF, Mathur PP. Adverse effects of Bisphenol A on male reproductive function. REV ENVIRON CONTAM T. 2014; 228: 57-82.
- [10]Al-Hiyasat AS, Darmani H, Elbetieha AM. Leached components from dental composites and their effects on fertility of female mice.Eur J Oral Sci.2004; 112:267-272.
- [11]Heber D. Herbal preparation for obesity: are they useful? Primary Care. 2003; 30: 441-463.
- [12]amal A, Arif JM, Ahmad IZ. Potential of Nigella sativa L. seed during different phases of germination on inhibition of bacterial growth. J. Biotechnol. Pharm. Res.2010; 1(1): 09-13.
- [13]Ali BH, Blunden G. Pharmacological and toxicological properties of Nigella sativa.Phytother. Res.2003; 17:299–305.
- [14]Juma FT, Abdulrahman HMA .The effects of Nigella sativa oil administration on some physiological and histological values of reproductive aspects of rats. Iraqi J. Vet.Medi.2011; 35(2): 52 – 60.
- [15]El-GaafarawyAM, Zaki AA, El-SedfyR,El-Khenawy KHI. Effect of feeding Nigella sativa cake on digestibility, nutritive value and reproductive performance of Frisian Iranian cows and Immuno activity of their offspring. Egyptian Journal of Nutrition Feeds.2003;6: 539-549.
- [16]Ayed AL, Talal Z. Long-term effects of Nigella sativa L. oil on some physiological parameters in normal and streptozotocin-induced diabetic rats. Journal of Diabetes Mellitus. 2011;1(3): 46-53.
- [17]Ghai CL. A Textbook of Practical Physiology. Jaypee Brothers Medical Publishers (p) Ltd: New Delhi, India, 2013.
- [18]Banchroft J, Stevens A, Turner D.Theory and practice of histological techniques churchillivingstone. New York, London, San Francisco, Tokyo, 1996.
- [19]Uluta OK, Yldz N, Durmaz E, Ahbab MA, Barlas N, Çok A. An in vivo assessment of the genotoxic potential of bisphenol -A and 4-tert-octylphenol in rats. Arch. Toxicol. 2011; 85: 995-1001.
- [20]Yamasaki K and Okuda H. Comparison of endocrine-mediated effects of two bisphenol – A related compounds, 2,2-bis (4-cyanatophyenyl) propane and 4,4´ cyclohexylidenebisphenol, based on subacute oral toxicity studies using rats. Toxicol. Lett. 2012; 208:162- 167.
- [21]Fang X, Cao S, Liu R.Interaction of Bisphenol A with bovine hemoglobin using spectroscopic and molecular modeling methods. Appl. Spectrosc.2011; 65.
- [22]Mohamed MA, Awad SM. Effect of Nigella sativa oil on some hematological values in aluminum-treated rats.Aust. J. Basic & Appl. Sci2008; 2(4): 1157-1164.
- [23]Abd-Elmonem HA. Effect of black seed oil supplementation on selected immunological, hematological and Iron status parameters in ribavirin treated female albino rats.Pak J Pharm Sci. 2018;31 (3):777-783.
- [24]Ebaid H, Dkhil MA, Zahran WS, El-FekiMA,Gabry MS. Role of Nigella sativa in ameliorating chloramphenicol induced tissue damage in rats. J. Med. Plant Res. 2011; 5:280-288.
- [25]Parhizkar S, Latiff LA, Rahman SA, Dollah MA, Parichehr H. Assessing estrogenic activity of Nigella sativa in ovariectomized rats using vaginal cornification assay. Afr. J. Pharm. Pharmacol.2011;5(2):137-142.
- [26]Jasima WK, Hassana MS, Keam GG. Study the effect of Nigella sativa on thyroid function and reproductive hormone of female rat. J. contemp. med. sci.2016;2(6): 67–69.
- [27]Caserta D, Di-Segni N, Mallozzi M, Giovanale V, Mantovani A, Marci R, Moscarini M. Bisphenol A and the female reproductive tract: an overview of recent laboratory evidence and epidemiological studies.Reprod. Biol. Endocrinol. 2014; 12:37.
- [28]Sheng ZG, Tang Y, Liu YX, Yuan Y, Zhao BQ, Chao XJ. Low concentrations of bisphenol A suppress thyroid hormone receptor transcription through a nongenomic mechanism. Toxicol. Appl. Pharmacol. 2012; 259(1):133–42.
- [29]Liu M, Xu X, Rang W, Li Y, Song Y. Influence of ovariectomy and 17-estradiol treatment on insulin sensitivity, lipid metabolism and post-ischemic cardiac function.Int. J. Cardiol.2004; 97(3): 485-493.
- [30]Sharif SH, Elmahdi BM, Ali Mohammed AM, Mohammed AH. The effects of Nigella sativa L. Ethanolic extract on thyroid function in normal and alloxan-induced diabetic rats. Thyroi. Res. &Prac.2012; 9:48–54.
- [31]Bacak GE and Avci G. Effects of thymoquinone on plasma leptin, insulin, thyroid hormones and lipid profile in rats fed a fatty diet. J. Fac. Vet. Med. KafkasUniver. 2013;19(6):1011–1016.
- [32]Lenie S, Cortvrindt R, Eichenlaub-Ritter U, Smitz J. Continuous exposure to bisphenol A during in vitro follicular development induces meiotic abnormalities. Mutat Res. 2008;651:71–81.
- [33]Peretz J, Craig ZR, Flaws JA. Bisphenol A inhibits follicle growth and induces atresia in cultured mouse antral follicles independently of the genomic estrogenic pathway. BiolReprod. 2012;87:63.