Qualitative phytochemicals and pharmacological properties analysis of methanol extract of Tabernaemontana pauciflora leaves
Abstract
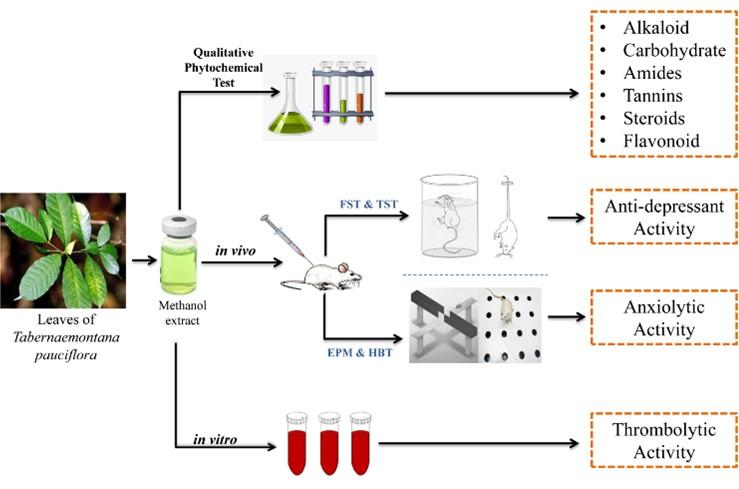
The methanol extract of the leaves of Tabernaemontana pauciflora (METP) was screened to investigate the qualitative phytochemical, in vivo anti-depressant, anxiolytic and in vitro thrombolytic activities. The in vivo antidepressant activity of METP was examined by force swimming test (FST) and tail suspension test (TST), whereas anxiolytic activity was examined by using hole-board test (HBT) and elevated plus maze test (EPM) on mice model. Additionally, qualitative phytochemical assay was performed to identify some secondary metabolites, namely alkaloids, carbohydrates, flavonoids. The in vitro thrombolytic model was used to evaluate the clot lysis effect of METP. In the antidepressant assay, METP (200 and 400 mg/kg) exhibited significant anti-depression activity (P < 0.001) on FST (45.64% and 58.31%) and TST (38.67% and 63.62%) whereas the notable anxiolytic effect was observed in EPM (67.67% and 56.06%) and HBT (35.53% and 50%) in dose dependent manner which compared with control. In in vitro thrombolytic assessment of METP exposed 22.66% inhibition of clot. The current results proposed that METP might be effective in neuropharmacological and thrombolytic response due to the presence of alkaloids, tannins and flavonoids, which required further studies to investigate the mechanism.
INTRODUCTION
Neurological disorders are the common phenomena. Currently an estimated 14.7M neurological cases, which according to 2019 is equivalent to at least 1 in 6 individuals living with one or more sign of neurological problems [1, 2]. Benzodiazepines are synthetic drugs to prescribe frequently for their anxiolytic, sedative-hypnotic and anticonvulsant actions [3]. Besides, depression is a common and serious dominant mental disorder that negatively affects how you feel on character, apathy, loss of energy, interruption of thinking and activity, as well as profound feelings of frustration, despair and suicidal ideation [3]. Instead of the availability of antidepressant drugs like TCA (Tricyclic antidepressant), selective reversible inhibitors of monoamine oxidase-A (MAO-A), selective serotonin reuptake inhibitors (SSRIs) and selective noradrenalin reuptake inhibitors (SNRIs) [4]. Depression is still denoted as a major medical problem associated with major side effects [5]. Not only associated with physical dependence and tolerance providing side effects, these psycho-neural drugs also cause for harmful effects on respiratory system, digestive system and immune systems of the human organ [6].
The depression is often conducted by irritability, anxiety, confusion, insomnia and behavioral disorders. As a result, the sequential use of an anxiolytic with an antidepressant agent is often necessary to recover the disease [7]. Selective serotonin reuptake inhibitors (SSRIs) are currently used as the first-choice therapy for depression, but insomnia, irritability and anxiety sometimes occur in the early stages of treatment. As these symptoms may result in early dropout or delays in the anti-depressive response, the concomitant use of an anxiolytic is considered helpful. A new anxiolytic and antidepressant agents that are why to search with the lowest adverse effect is still researchers have high interest of this area [8]
Atherothrombotic, the formation of a blood clot within an artery as a result of atherosclerosis named cardiovascular disease are characterized by the existence of great influences of thrombus in the blood vessel [9]. The formation of complex form by connecting among platelets to platelets, also to leucocytes by activated platelets results in plaque formation and development of thrombosis. A neutral agent called plasmin triggers clot lysis by crumble up fibrinogen & fibrin contained clot [10]. Additional plasminogen agent can easily convert to plasmin by streptokinase enzyme which forms a 1:1 stoichiometric complex with plasminogen [11].
T. pauciflora of the family of Apocynaceae locally named as Bunut, Malul-ulat, Rodok. It is distributed in Chittagong hill tracts area, Bangladesh, Vietnam, Cambodia, peninsular Malaysia (widespread), Myanmar, Borneo. Traditionally the different part of T. pauciflora is used to cure headaches, to remedy various syphilitic afflictions effect and to treat eruptions of the skin [12]. Based on their ethno botanical uses the present study aimed to investigate the qualitative phytochemical analysis, anxiolytic, antidepressant and thrombolytic activities of methanol extract of T. Pauciflora leaves.
MATERIALS AND METHODS
Chemicals
The chemicals were used for the experiments as methanol (Sigma Chemicals, USA). Diazepam, fluoxetine (Eskayef Banlgadesh Ltd; Tongi, Bangladesh) and lyophilized streptokinase (SK) vial (Square Pharmaceuticals Ltd. Bangladesh). All other chemicals were obtained from local trader through Taj Scientific Ltd, Chittagong, Bangladesh.
Animals
The study was accompanied on Swiss Albino mice were purchased from Jahangirnagar University, Dhaka, Bangladesh. They were five to six weeks of age of male-female, weighing about 20 – 30 g, which were kept in housed colony cages (six mice per cages) at ambient temperature of 25-27°C with 12 hours light and dark cycles having with proper ventilation in the room [13]. The study was conducted according to the guidelines of P&D Committee of Department of Pharmacy, International Islamic University Chittagong (Pharm/AEC/138/13-’19, 22/12/2019).
Collection and preparation of plant extract
Fresh leaves of T. pauciflora were collected from a local village Chittagong, Bangladesh and authenticated by Dr Shaikh Bokhtiar Uddin, Professor, Department of Botany, University of Chittagong. The leaves were shaded dried for a multi-week time and ground into a fine powder utilizing a process machine. The ground leaves (1000g) were absorbed adequate measure of methanol (3L) for ten days at room temperature with incidental shaking and mixing at that point separated through with cotton plug followed by Whitman channel paper No. 1. After filtration, the filtrated was subjected for the evaporation in water bath. The obtained semisolid yield extract was 10.06% which was refrigerated at 4 degree centigrade for further use.
Qualitative phytochemical analysis
The METP tried for the screening of bioactive mixes by subjective way utilizing keeping standard method to assess the alkaloids, carbohydrates, flavonoids, terpenoids, tannins, polyphenol, phenols, steroids, amides and glycosides [13-15].
Evaluation of anxiolytic activity
Elevated plus maze (EPM) test
The EPM test is used mainly to assess exploratory behaviors in a mouse model. The EPM apparatus consisted of two open arms (5×10 cm2) separated by two closed arms (5×10×15 cm3), which raised to a height of 25 cm in a dimly light space [16, 17]. The maze floor and walls were constructed from dark opaque wood. Mice (n = 5) were treated with extract (200 and 400 mg/kg). After 30 min, each mouse placed individually in the center of the EPM facing towards closed arm and recorded the % of time spent in the open arms for 5 min.
% time spent in open arm = (time spent in open arm)/(time spent in open arm + time spent in closed arm) ×100
Hole-board test (HBT)
The hole-board apparatus was applied for this experiment as the method stated earlier. The tool consisted of a wooden box (40 cm × 40 cm.) in diameter with 16 holes uniformly distributed on the surface. The apparatus elevated to the 25 cm height. In brief, mice were tested after the 30 minutes of dosing with METP (200 and 400 mg/kg) and 15 minutes after the administration of diazepam (1 mg/kg), whereas each mice was allowed to move on the platform and the number of head dips in the holes was counted for 5 minutes [18, 19].
Evaluation of anti-depressant activity
Force swimming test (FST)
Force swimming test is the most widely used assay described by Porsolt et. al. frequently used behavioral models for screening antidepressant activity. The animals were divided into four groups with 5 in each and prepared the test dose 400 mg/kg and 200 mg/kg along with control dose (0.9% saline water; 10 ml/ kg orally) and fluoxetine hydrochloride (10 mg/kg). Then take the mice’s weight and administrate dose according to their weight. After 30 minutes each mouse was individually forced to swim in open glass chamber (25 × 15 × 25 cm) containing fresh water to a height of 15 cm and maintained at 26° ± 1°C. It has to be ensured that mice could not get any support by touching the bottom of the sidewalls of the chamber. Each of the mice shows vigorous movement during the initial 2 minutes period of the test. The duration of immobility was manually recorded during the next 5 minutes of the total 7 min testing period [20, 21].
Tail suspension test (TST)
This procedure test commonly employed behavioral model for screening antidepressant like activity in mice was first given by Stern et al. The animals are divided into four groups with 5 in each. At first, we have to prepare the test dose 400 mg/kg and 200 mg/kg along with control dose (0.9% saline water, 10 ml/kg orally) and Fluoxetine hydrochloride (10 mg/kg). Then take the mice’s weight and administrate dose according to their weight. After 30 minutes of dose administration individually each mouse was suspended to the edge of a table, the table was 50 cm above the floor and tape was placed approximately 1 cm from the tip of the tail. The total period of immobility was recorded manually for 7 min [22, 23].
Evaluation of thrombolytic activity
The thrombolytic effect of METP justified through slide modification of method which described by Daginawala and Kawsar et al. [24, 25], where streptokinase was used as standard. Streptokinase vial (15, 00,000 I.U) which are available in drug market was accepted and prepared solution with 5 ml distilled water. Later, streptokinase 100 μl dose from this stock solution were used for in vitro analysis. Blood was derived from five healthy students except consideration of previous antithrombotic therapy. After that, collected blood was assigned into pre-weighed micro centrifuge tube for allowing clot formation. The following formula was applied for the determination of clot lysis:
Clot lysis (%) = (weight of released clot/clot weight) × 100
Statistical analysis
The values were showed in mean ± standard error mean (SEM). **P< 0.001 and *P< 0.01 statistically significant, which was carried by one-way ANOVA (Dunnett’s test) using GraphPad Prism (version 8.4.) software.
RESULTS
Qualitative phytochemical assay
Phytochemicals are present and the total content of METP is alkaloids, carbohydrates, tannins, flavonoids, steroids and amides presented in the Table 1. It was found to be absent of glycosides, polyphenol, phenols and terpenoids in the METP.
Table 1. Qualitative phytochemical screening of T. pauciflora (leaves).
Effect of methanol extract of T. pauciflora on anxiolytic activity
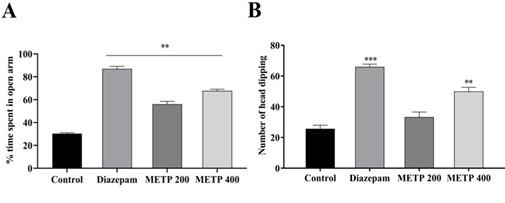
The METP displayed increased percentage of time spent into open arms, exposed in Figure 1 (A). The METP displayed a significant percentage of time spent in open arm (67.67 ± 1.45 and 56.06 ± 2.44) by 400 and 200 mg/kg respectively compared with control (55.06 ± 2.77), whereas the standard drug diazepam exhibited (87.02 ± 2.08). Additionally, HBT is an exploratory technique applied in animal to measure anxiety, stress and emotion. There was dose depended effect increases when mice treated with METP 400 mg/kg and 200 mg/kg, represented in Figure 1 (B). The METP 400 mg/kg dose exhibited significant number of head dipping (50 ± 2.65) compared to control (25.67± 2.33), whereas the diazepam showed (66 ± 1.73) number of head dipping in hole-board apparatus.
Effect of methanol extract of T. pauciflora on antidepressant activity
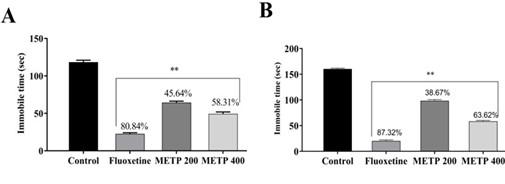
Possible antidepressant effect of METP was tested in the FST after oral administration. In this study, animals treated with two doses of METP (200 and 400 mg/kg) reported decreases in their time of immobility, which was important for METP at (49.33 ± 2.60 s and 64.33 ± 2.03 s), compared to control (118.33 ± 2.60 s and 22.67 ± 1.45 s). The result presented in Figure 2 (A). Similarly, as predicted, animals were treated with fluoxetine (10 mg/kg) showed a substantial decrease (22.6 s) in the immobility period. The mice was treated with two doses of METP 200 and 400 mg/kg in this FST, revealed decreases in their immobility times, which was (98.33 ± 0.88 s and 56 ± 0.88 s) for methanol extract of T. pauciflora (leaves) when compared to negative control (160.33 ± 1.45 s), shown in Figure 2 (B). Similarly, mice treated with standard fluoxetine (10 mg/kg) as expected, manifested a remarkable decrease in the immobility time (20.66 ± 1.45 s).
Effect of methanol extract of T. pauciflora on thrombolytic activity
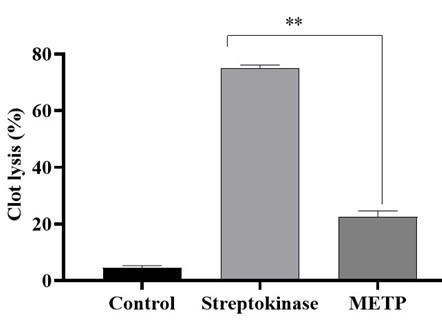
The in vitro thrombolytic model was used to measure the clot lysis of the METP compared with streptokinase, and normal saline (control) expressed in Figure 3. The METP exposed significant (P < 0.001) percentage of clot lysis activity (22.66 ± 1.96%) compared to control (4.66 ± 0.63%), whereas standard drug streptokinase evinced (75.0 ± 1.14%) clot lysis action.
DISCUSSION
The drug discovery system is the process in which new candidate medications are discovered. Medicinal plant drug discovery continuously increasing day by day to provide new and significant leads against pharmacological targets [26]. Phytochemical investigation of the crude extract was done to find out the uncovered presence of compound which might be responsible for the therapeutic uses [27, 28]. The phytochemical (qualitative) studies demonstrated that the methanolic extracts of T. pauciflora contained alkaloids, tannins, flavonoids, steroids, and amides. This phytochemical group present in the METP may help us to find out the specific compound and also help us to estimate how much bioactive in this plant extract.
Depressant causes a slowing motion in the physiological functions. There is various leading cause falling in depression such as oxidative stress, long time inflammation, and several chronic disorders, trauma hemorrhage, difficulties within the family, which causes a slowing motion in the physiological functions [29, 30]. Antidepressants are those medications that help relieve symptoms of mood disorders by repairing neurotransmitter chemical imbalances in the brain. The tail suspension test (TST) is done to predict the antidepressant pharmacological activity in animal model [31]. The total immobility time after induced by tail suspension of METP was measured according by the Steru et al. [31, 32]. In this test animal were treated with two doses of METP 200 and 400 mg/kg, results are represented as mean ± SEM, where the METP 400 mg/kg exhibited significant decreases in the immobility times as (58.33 ± 1.76 s) compared with the control as (160.33 ± 1.45 s), whereas the standard drug fluoxetin showed (20.33 ± 1.76 s) immobility time [33]. Force swimming test is commonly used to screen antidepressant activity and other types of psychotropic actions in animal [34]. The term immobility are considered to possess the behavioral desperation analogous to that seen in human depression and it is well endowed that antidepressant ligands are able to inhibit the duration of immobility in animal [35]. The both dose of METP displayed remarkable reduction of immobility time (49.33 ± 2.60 s and 64.33 ± 2.03 s) compared with the control (118.33 ± 2.60 s). The METP 400 mg/kg displayed significant decreases of number of head dipping (50 ± 2.65) compared with control (25.67± 2.33) 10 ml/kg whereas standard (Diazepam) 1 mg/kg showed (66 ± 1.73). From above discussion it can be suggested that the methanol extract of T. pauciflora (precisely METP 400 mg/kg) has significant anti-depressant effect.
Anxiety denotes a normal reaction to stress. Anxiolytics relieve anxiety disorders associated with rapid heart rate, palpitations and chest pain and also can increase the risk of high blood pressure and cardiac disease [36, 37]. The Elevated Plus Maze (EPM) test has been used in a model of anxiety [38]. In the EPM test, the METP 400 mg/kg dose revealed crucial inhibition of number of time spent (67.67 ± 1.45) compared to control (1% tween-80 solution) dose. In this test diazepam is the reference control or positive control with anxiolytic effect mainly used in the chronic treatment [39, 40]. The HBT is an experimental system which was designed 1970s as open arm test and done in animal to evaluate anxiolytic result [41] by measuring anxiety and emotion. In this study , we use this hole board test method to assess the new leads for anxiolytic activity compared to standard (reference) drug diazepam in animal model [42]. Every mouse evaluate by placing it separately in the center of the hole board apparatus and observed its activity for head dipping of the mouse is determined when both ears below the top of the hole [43]. The extract dose has anxiolytic activity determined by the reduction of number of head dipping with time spent after exposer of the METP dose compared with the control [44, 45]. The both METP 400 mg/kg and 200 mg/kg displayed significant decreases of number of head dipping (84 ± 2.65 and 100 ± 3.21) compared with control (1% tween-80 solution) 10 ml/kg whereas standard (Diazepam) 1 mg/kg showed (13 ± 1.73). From above discussion, we may can displayed another types anxiolytic ligand which will be effective if this result may analysis further. For the evaluation of leads compound of anxiolytic activity test method the elevated plus maze test and hole-board test were used. The presence of anxiolytic effect of METP due to the presence of flavonoid group [46].
Thrombolysis is a parameter which dissolve the blood clot already fromed in the blood vessel and protect from the various heart diseases through improving blood flow, especially infused an artificial enzyme such as streptokinase in dangerous situation associated with major pulmonary embolism, late arterial occlusion and in deep vein thrombosis [47, 48]. Activation of blood lysis factors such as plasmin,fibrinogen and fibrin and others enhances the breakdown of the blood clot to increase the blood flow [49, 50]. Generally, thrombolytic agent activates the plasminogen that forms plasmin which acts as a cleaved product to dissolve blood clots [51]. In this project we studied in vitro trombolytic experiment of METP, where METP exhibited siginifant percent of clot lysis (22.67 ± 1.96) applied in animal model compared with the control (4.66 ± 0.63), whereas standard drug manifested effective result (**P< 0.001). This effect may showed due to presence of flavonoids contents in the extract [52]. This significant result compared to negative control due to the presence of alkaloids and tannins [53].
CONCLUSION
In our present well-informed study stated that methanol extract of T. pauciflora is potetial source for the important phytochemicals having anxiolytic, antidepressant and thrombolytic outcome (Figure 4). According to study of the results and discussions of METP, this plant may contain effective herbal drug. Further advanced studies are required to carefully evaluate the mechanisms which responsible for this anxiolytic and antidepressants phenomenon being reported.
ACKNOWLEDGEMENTS
Authors are thankful to Department of Pharmacy, International Islamic University Chittagong, Chittagong, Bangladesh, for their research facilities and support. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. This work is conducted with the individual funding of all authors.
CONFLICTS OF INTERESTS
We, the authors, declare that we have no competing interests.
AUTHOR CONTRIBUTIONS
AH, MNI and AU together planned and designed the research. MNI arranged the whole facilities and co-supervised this research. AH conducted the entire laboratory works. MA, NB, NA, SAS and AH imparted in study design and interpreted the results putting efforts on statistical analysis with MRAM, SAJS and MNI participated in the manuscript draft and has thoroughly checked and revised the manuscript for necessary changes in format, grammar and English standard. All authors read and agreed on the final version of the manuscript.
References
- [1]Costello EJ, Egger H, Angold A. 10-year research update review: the epidemiology of child and adolescent psychiatric disorders: I. Methods and public health burden. Journal of the American Academy of Child & Adolescent Psychiatry. 2005;44:972-86.
- [2]Costello EJ, Foley DL, Angold A. 10-year research update review: the epidemiology of child and adolescent psychiatric disorders: II. Developmental epidemiology. Journal of the American Academy of Child & Adolescent Psychiatry. 2006;45:8-25.
- [3]Okasha A. Mental health in the Middle East: an Egyptian perspective. Clinical psychology review. 1999;19:917-33.
- [4]Baumann P. Pharmacokinetic-pharmacodynamic relationship of the selective serotonin reuptake inhibitors. Clinical Pharmacokinetics. 1996;31:444-69.
- [5]Lambert C, Berlin I, Lee T-L, Hee SW, Tan AS, Picard D, et al. A standardized transcutaneous electric acupoint stimulation for relieving tobacco urges in dependent smokers. Evidence-Based Complementary and Alternative Medicine. 2011;2011.
- [6]Celada P, Bortolozzi A, Artigas F. Serotonin 5-HT 1A receptors as targets for agents to treat psychiatric disorders: rationale and current status of research. CNS Drugs. 2013;27:703-16.
- [7]Kanba S. Although antidepressants and anxiolytics are frequently used together to treat depression in the acute phase, how effective is the concomitant use of these drugs? Journal of Psychiatry and Neuroscience. 2004;29:485-6.
- [8]Greenbaum DS, Mayle JE, Vanegeren LE, Jerome JA, Mayor JW, Greenbaum RB, et al. Effects of desipramine on irritable bowel syndrome compared with atropine and placebo. Digestive diseases and sciences. 1987;32:257-66.
- [9]Novokhatny V, Taylor K, Zimmerman T. Thrombolytic potency of acid‐stabilized plasmin: superiority over tissue‐type plasminogen activator in an in vitro model of catheter‐assisted thrombolysis. Journal of Thrombosis and Haemostasis. 2003;1:1034-41.
- [10]Banerjee A, Chisti Y, Banerjee U. Streptokinase—a clinically useful thrombolytic agent. Biotechnology advances. 2004;22:287-307.
- [11]Thapar N, Sanderson IR. Diarrhoea in children: an interface between developing and developed countries. The lancet. 2004;363:641-53.
- [12]Silveira D, de Melo AF, Magalhães P, Fonseca-Bazzo Y. Tabernaemontana species: Promising sources of new useful drugs. Studies in Natural Products Chemistry: Elsevier; 2017. p. 227-89.
- [13]Slinkard K, Singleton VL. Total phenol analysis: automation and comparison with manual methods. American journal of enology and viticulture. 1977;28:49-55.
- [14]Charrouf Z, Guillaume D. Ethnoeconomical, ethnomedical, and phytochemical study of Argania spinosa (L.) Skeels. Journal of Ethnopharmacology. 1999;67:7-14.
- [15]Costa APR, Vieira C, Bohner LO, Silva CF, da Silva Santos EC, De Lima TCM, et al. A proposal for refining the forced swim test in Swiss mice. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2013;45:150-5.
- [16]de Almeida RN, Motta SC, de Brito Faturi C, Catallani B, Leite JR. Anxiolytic-like effects of rose oil inhalation on the elevated plus-maze test in rats. Pharmacology Biochemistry and Behavior. 2004;77:361-4.
- [17]Rabbani M, Sajjadi S, Zarei H. Anxiolytic effects of Stachys lavandulifolia Vahl on the elevated plus-maze model of anxiety in mice. Journal of Ethnopharmacology. 2003;89:271-6.
- [18]Sonavane G, Sarveiya V, Kasture V, Kasture S. Anxiogenic activity of Myristica fragrans seeds. Pharmacology Biochemistry and Behavior. 2002;71:239-44.
- [19]Chopin P, Pellow S, File SE. The effects of yohimbine on exploratory and locomotor behaviour are attributable to its effects at noradrenaline and not at benzodiazepine receptors. Neuropharmacology. 1986;25:53-7.
- [20]Borsini F, Meli A. Is the forced swimming test a suitable model for revealing antidepressant activity? Psychopharmacology. 1988;94:147-60.
- [21]Detke MJ, Rickels M, Lucki I. Active behaviors in the rat forced swimming test differentially produced by serotonergic and noradrenergic antidepressants. Psychopharmacology. 1995;121:66-72.
- [22]Cryan JF, Mombereau C, Vassout A. The tail suspension test as a model for assessing antidepressant activity: review of pharmacological and genetic studies in mice. Neuroscience & Biobehavioral Reviews. 2005;29:571-625.
- [23]Karolewicz B, Paul IA. Group housing of mice increases immobility and antidepressant sensitivity in the forced swim and tail suspension tests. European Journal of Pharmacology. 2001;415:197-201.
- [24]Sikder MAA, Siddique AB, Hossian A, Miah MK, Kaisar MA, Rashid MA. Evaluation of thrombolytic activity of four Bangladeshi medicinal plants, as a possible renewable source for thrombolytic compounds. Journal of Pharmacy and Nutrition Sciences. 2011;1:4-8.
- [25]Uddin MMN, Shahriar M. Phytochemical screenings, thrombolytic activity and antimicrobial properties of the bark extracts of Averrhoa bilimbi. Journal of Applied Pharmaceutical Science. 2013;3:094-6.
- [26]Atanasov AG, Waltenberger B, Pferschy-Wenzig E-M, Linder T, Wawrosch C, Uhrin P, et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnology advances. 2015;33:1582-614.
- [27]Majid M, Khan MR, Shah NA, Haq IU, Farooq MA, Ullah S, et al. Studies on phytochemical, antioxidant, anti-inflammatory and analgesic activities of Euphorbia dracunculoides. BMC complementary and alternative medicine. 2015;15:349.
- [28]Wadood A, Ghufran M, Jamal SB, Naeem M, Khan A, Ghaffar R. Asnad. Phytochemical analysis of medicinal plants occurring in local area of Mardan Biochem Anal Biochem. 2013;2:1-4.
- [29]Zellweger R, Ayala A, DeMaso CM, Chaudry IH. Trauma-hemorrhage causes prolonged depression in cellular immunity. Shock. 1995;4:149-53.
- [30]Lauber C, Falcato L, Nordt C, Rössler W. Lay beliefs about causes of depression. Acta Psychiatrica Scandinavica. 2003;108:96-9.
- [31]Peng W-H, Lo K-L, Lee Y-H, Hung T-H, Lin Y-C. Berberine produces antidepressant-like effects in the forced swim test and in the tail suspension test in mice. Life Sciences. 2007;81:933-8.
- [32]Steru L, Chermat R, Thierry B, Simon P. The tail suspension test: a new method for screening antidepressants in mice. Psychopharmacology. 1985;85:367-70.
- [33]Page ME, Detke MJ, Dalvi A, Kirby LG, Lucki I. Serotonergic mediation of the effects of fluoxetine, but not desipramine, in the rat forced swimming test. Psychopharmacology. 1999;147:162-7.
- [34]Borsini F, Volterra G, Meli A. Does the behavioral “despair” test measure “despair”? Physiology & behavior. 1986;38:385-6.
- [35]Porsolt R, Bertin A, Jalfre M. Behavioral despair in mice: a primary screening test for antidepressants. Archives internationales de pharmacodynamie et de therapie. 1977;229:327.
- [36]Huffman JC, Smith FA, Blais MA, Januzzi JL, Fricchione GL. Anxiety, independent of depressive symptoms, is associated with in-hospital cardiac complications after acute myocardial infarction. Journal of psychosomatic research. 2008;65:557-63.
- [37]Hasanat A, Kabir MSH, Ansari MA, Chowdhury TA, Hossain MM, Islam MN, et al. Ficus cunia Buch.-Ham. ex Roxb.(leaves): An experimental evaluation of the cytotoxicity, thrombolytic, analgesic and neuropharmacological activities of its methanol extract. Journal of Basic and Clinical Physiology and Pharmacology. 2019;30.
- [38]Brown E, Hurd NS, McCall S, Ceremuga TE. Evaluation of the anxiolytic effects of chrysin, a Passiflora incarnata extract, in the laboratory rat. AANA journal. 2007;75.
- [39]Rickels K, Case WG, Downing RW, Winokur A. Long-term diazepam therapy and clinical outcome. Jama. 1983;250:767-71.
- [40]Bodnoff SR, Suranyi-Cadotte B, Aitken DH, Quirion R, Meaney MJ. The effects of chronic antidepressant treatment in an animal model of anxiety. Psychopharmacology. 1988;95:298-302.
- [41]Brown GR, Nemes C. The exploratory behaviour of rats in the hole-board apparatus: is head-dipping a valid measure of neophilia? Behavioural processes. 2008;78:442-8.
- [42]Crawley J, Davis L. Baseline exploratory activity predicts anxiolytic responsiveness to diazepam in five mouse strains. Brain research bulletin. 1982;8:609-12.
- [43]Calabrese EJ. An assessment of anxiolytic drug screening tests: hormetic dose responses predominate. Critical reviews in toxicology. 2008;38:489-542.
- [44]Nolan NA, Parkes M. The effects of benzodiazepines on the behaviour of mice on a hole-board. Psychopharmacologia. 1973;29:277-88.
- [45]Lourenço da Silva A, Elisabetsky E. Interference of propylene glycol with the hole-board test. Brazilian Journal of Medical and Biological Research. 2001;34:545-7.
- [46]Kaur R, Kaur S. Anxiolytic potential of methanol extract from Ageratum conyzoides Linn Leaves. Pharmacognosy Journal. 2015;7.
- [47]Brogden R, Speight T, Avery G. Streptokinase: a review of its clinical pharmacology, mechanism of action and therapeutic uses. Drugs. 1973;5:357-445.
- [48]Shifah F, Tareq AM, Sayeed MA, Islam MN, Emran TB, Ullah MA, et al. Antidiarrheal, cytotoxic and thrombolytic activities of methanolic extract of Hedychium coccineum leaves. measurement.11:12.
- [49]Taylor FB, MÜLLER-EBERHARD HJ. Factors influencing lysis of whole blood clots. Nature. 1967;216:1023-5.
- [50]Korninger C, Collen D. Studies on the specific fibrinolytic effect of human extrinsic (tissue-type) plasminogen activator in human blood and in various animal species in vitro. Thrombosis and haemostasis. 1981;46:561-5.
- [51]Ambrus CM, Back N, Ambrus JL. On the mechanism of thrombolysis by plasmin. Circulation research. 1962;10:161-5.
- [52]Gryglewski RJ, Korbut R, Robak J, Świȩs J. On the mechanism of antithrombotic action of flavonoids. Biochemical pharmacology. 1987;36:317-22.
- [53]Bhowmick R, Sarwar MS, RahmanDewan SM, Das A, Das B, NasirUddin MM, et al. In vivo analgesic, antipyretic, and anti-inflammatory potential in Swiss albino mice and in vitro thrombolytic activity of hydroalcoholic extract from Litsea glutinosa leaves. Biological Research. 2014;47:56.